Diabetes Mellitus Dr Wayne Riback Senior Medical Advisor

Diabetes Mellitus Dr Wayne Riback Senior Medical Advisor 1
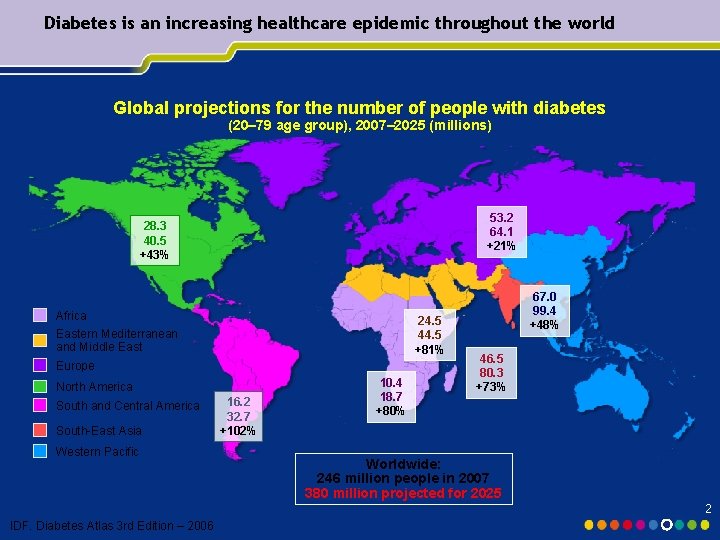
Diabetes is an increasing healthcare epidemic throughout the world Global projections for the number of people with diabetes (20– 79 age group), 2007– 2025 (millions) 53. 2 64. 1 +21% 28. 3 40. 5 +43% Africa 24. 5 44. 5 +81% Eastern Mediterranean and Middle East Europe North America South and Central America South-East Asia Western Pacific 16. 2 32. 7 +102% 10. 4 18. 7 +80% 67. 0 99. 4 +48% 46. 5 80. 3 +73% Worldwide: 246 million people in 2007 380 million projected for 2025 2 IDF. Diabetes Atlas 3 rd Edition – 2006
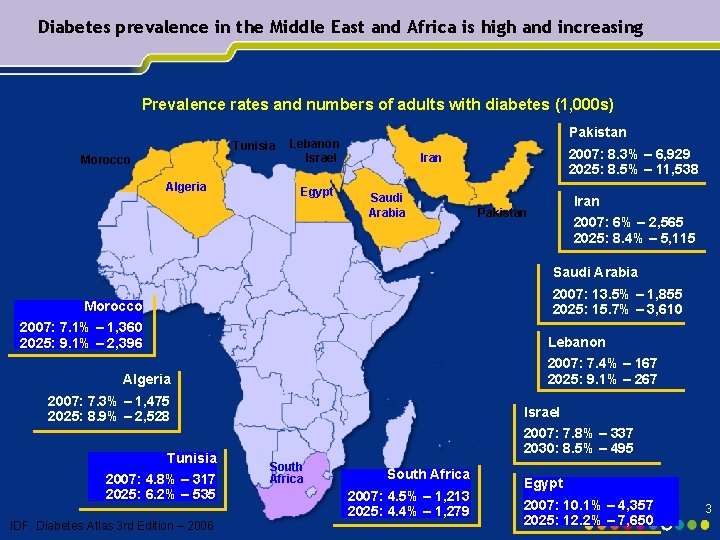
Diabetes prevalence in the Middle East and Africa is high and increasing Prevalence rates and numbers of adults with diabetes (1, 000 s) Tunisia Morocco Algeria Pakistan Lebanon Israel Egypt 2007: 8. 3% – 6, 929 2025: 8. 5% – 11, 538 Iran Saudi Arabia Iran Pakistan 2007: 6% – 2, 565 2025: 8. 4% – 5, 115 Saudi Arabia 2007: 13. 5% – 1, 855 2025: 15. 7% – 3, 610 Morocco 2007: 7. 1% – 1, 360 2025: 9. 1% – 2, 396 Lebanon 2007: 7. 4% – 167 2025: 9. 1% – 267 Algeria 2007: 7. 3% – 1, 475 2025: 8. 9% – 2, 528 Tunisia 2007: 4. 8% – 317 2025: 6. 2% – 535 IDF. Diabetes Atlas 3 rd Edition – 2006 Israel 2007: 7. 8% – 337 2030: 8. 5% – 495 South Africa 2007: 4. 5% – 1, 213 2025: 4. 4% – 1, 279 Egypt 2007: 10. 1% – 4, 357 2025: 12. 2% – 7, 650 3
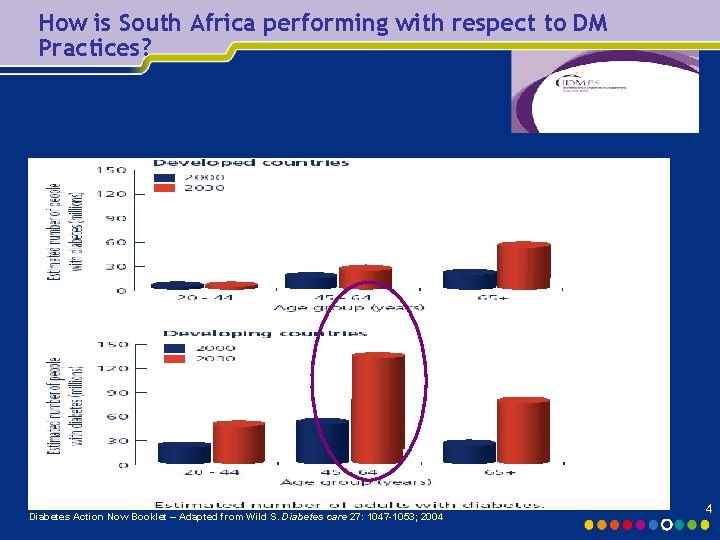
How is South Africa performing with respect to DM Practices? Diabetes Action Now Booklet – Adapted from Wild S. Diabetes care 27: 1047 -1053; 2004 4
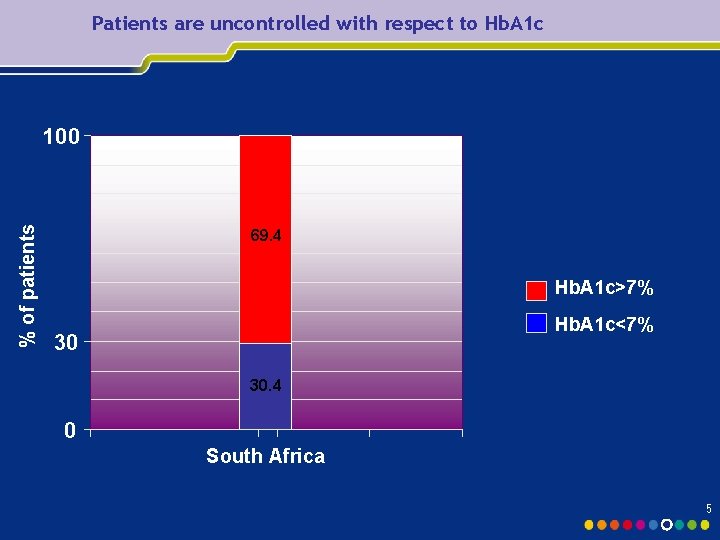
Patients are uncontrolled with respect to Hb. A 1 c % of patients 100 69. 4 Hb. A 1 c>7% Hb. A 1 c<7% 30 30. 4 0 South Africa 5
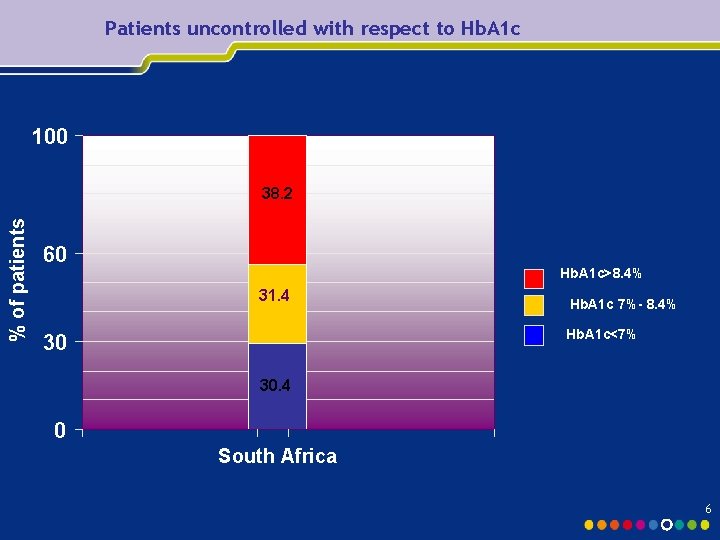
Patients uncontrolled with respect to Hb. A 1 c 100 % of patients 38. 2 60 Hb. A 1 c>8. 4% 31. 4 Hb. A 1 c 7%- 8. 4% Hb. A 1 c<7% 30 30. 4 0 South Africa 6
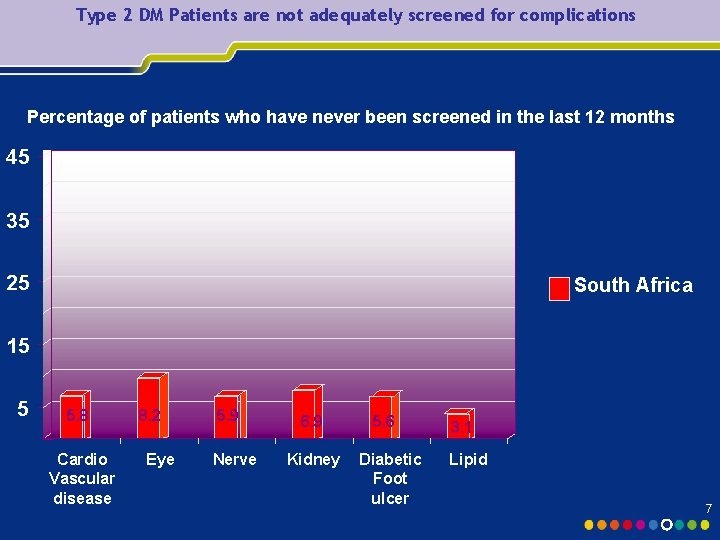
Type 2 DM Patients are not adequately screened for complications Percentage of patients who have never been screened in the last 12 months 45 35 25 South Africa 15 5 5. 8 Cardio Vascular disease 8. 2 Eye 5. 9 Nerve 6. 9 Kidney 5. 6 Diabetic Foot ulcer 3. 1 Lipid 7
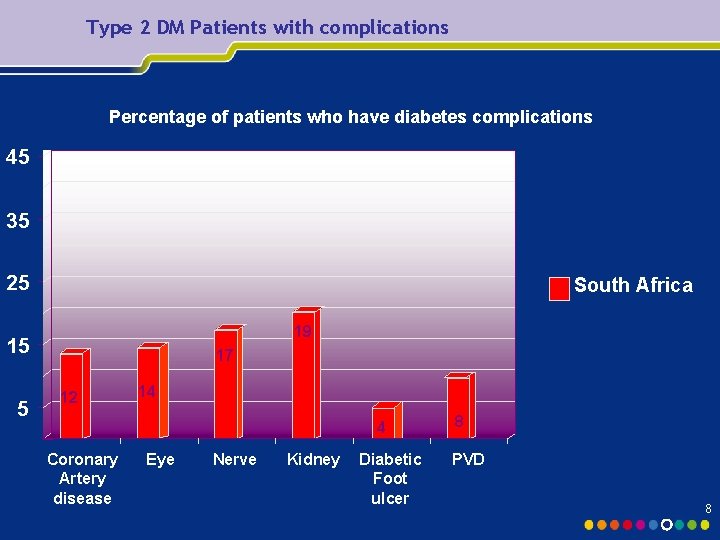
Type 2 DM Patients with complications Percentage of patients who have diabetes complications 45 35 25 South Africa 19 15 5 17 12 14 4 Coronary Artery disease Eye Nerve Kidney Diabetic Foot ulcer 8 PVD 8
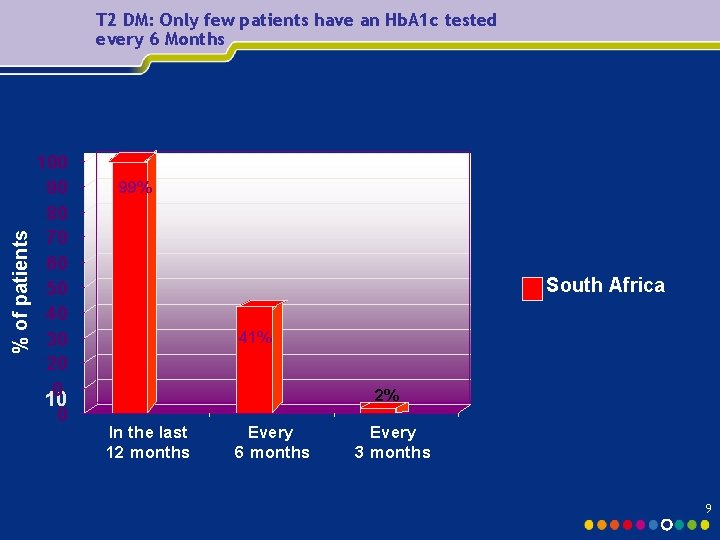
% of patients T 2 DM: Only few patients have an Hb. A 1 c tested every 6 Months 100 90 80 70 60 50 40 30 20 0 10 0 99% South Africa 41% 2% In the last 12 months Every 6 months Every 3 months 9

The burden of diabetes on healthcare systems l Diabetes accounts for between 5% and 10% of any nation’s health budget l Three times the healthcare resources are being spent on treating diabetes complications compared with that spent on controlling diabetes before the onset of complications Jonsson B. Diabetologia 2002; 45: S 5–S 12 Status of diabetes management CODE-2: cost of type 2 diabetes in Europe 10

Pathogenesis of type 2 diabetes mellitus 11
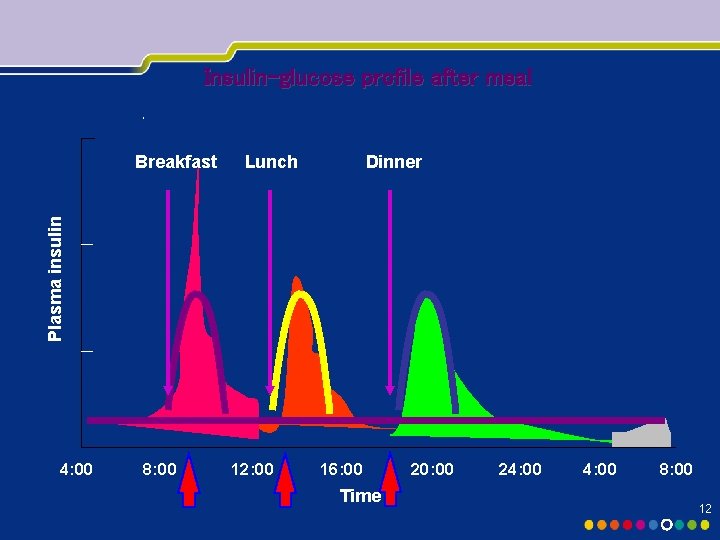
Insulin-glucose profile after meal Lunch Dinner Plasma insulin Breakfast 4: 00 8: 00 12: 00 16: 00 Time 20: 00 24: 00 8: 00 12

Type 2 diabetes mellitus (T 2 DM) requires progressive therapy l T 2 DM is a progressive disease characterised by increased insulin resistance and decreasing pancreatic β-cell function 1 l At diagnosis, patients may have already lost approximately 50% of β-cell function 2 l An ideal treatment regimen for T 2 DM should provide: § Continuity of care as the disease progresses § Flexibility to adapt to individual needs 1. Bergenstal RM. In: Textbook of Diabetes Mellitus, 3 rd edition: John Wiley & Sons; 2004: p 995― 1015. 2. Holman RR. Diabetes Res Clin Pract 1998; 40(suppl 1): S 21– 5. 13
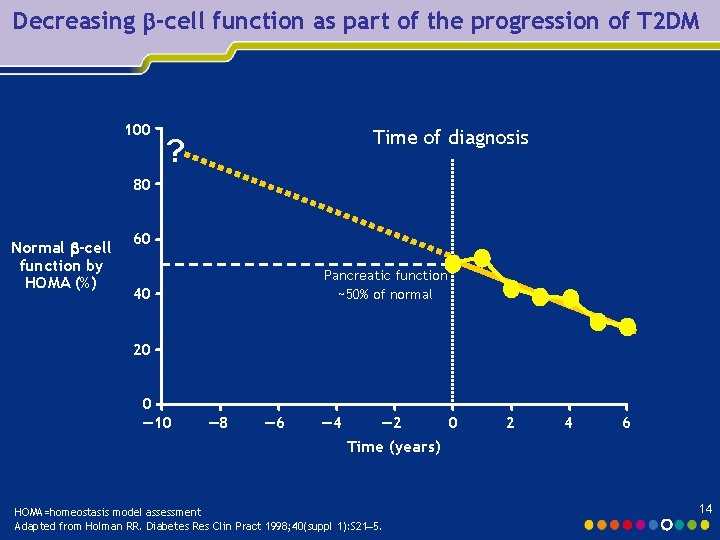
Decreasing -cell function as part of the progression of T 2 DM 100 Time of diagnosis ? 80 Normal -cell function by HOMA (%) 60 Pancreatic function ~50% of normal 40 20 0 ― 10 ― 8 ― 6 ― 4 ― 2 0 Time (years) HOMA=homeostasis model assessment Adapted from Holman RR. Diabetes Res Clin Pract 1998; 40(suppl 1): S 21– 5. 2 4 6 14
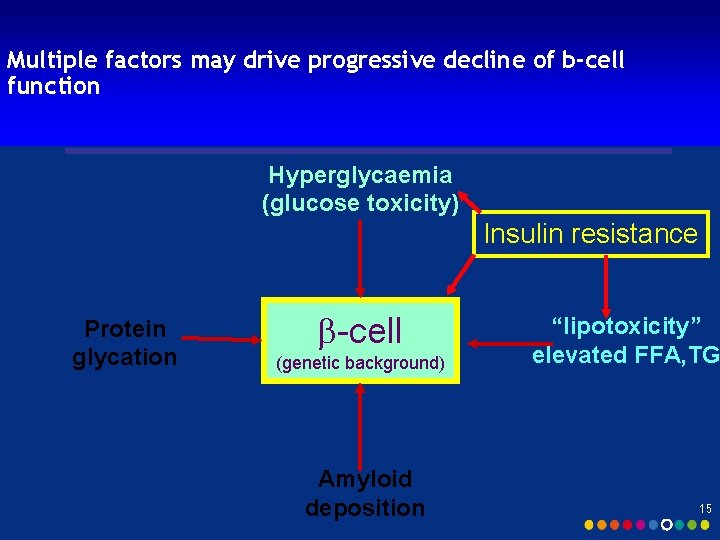
Multiple factors may drive progressive decline of b-cell function Hyperglycaemia (glucose toxicity) Insulin resistance Protein glycation b-cell (genetic background) Amyloid deposition “lipotoxicity” elevated FFA, TG 15
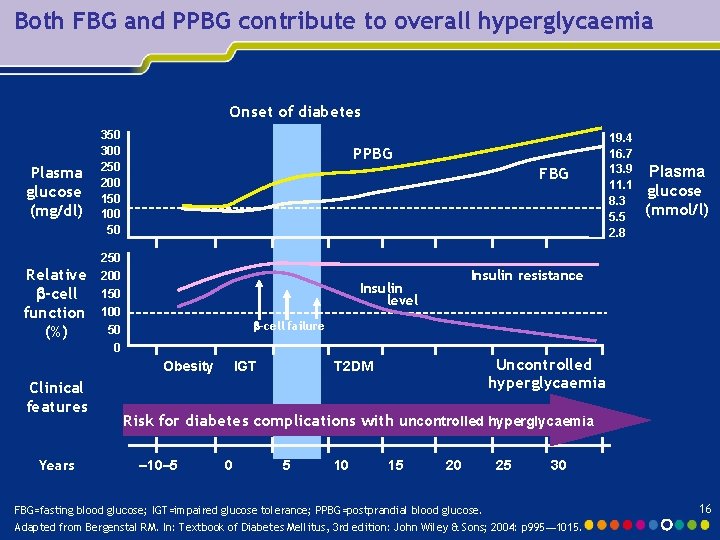
Both FBG and PPBG contribute to overall hyperglycaemia Onset of diabetes Plasma glucose (mg/dl) Relative -cell function (%) 350 300 250 200 150 100 50 PPBG FBG 250 200 150 -cell failure Obesity Clinical features Years Plasma glucose (mmol/l) Insulin resistance Insulin level 100 50 0 19. 4 16. 7 13. 9 11. 1 8. 3 5. 5 2. 8 IGT Uncontrolled hyperglycaemia T 2 DM Risk for diabetes complications with uncontrolled hyperglycaemia – 10– 5 0 5 10 15 20 25 30 FBG=fasting blood glucose; IGT=impaired glucose tolerance; PPBG=postprandial blood glucose. Adapted from Bergenstal RM. In: Textbook of Diabetes Mellitus, 3 rd edition: John Wiley & Sons; 2004: p 995 ― 1015. 16
17
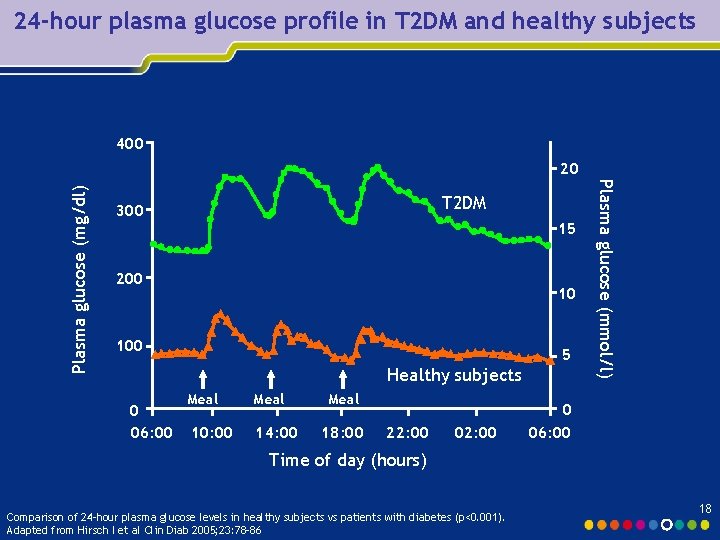
24 -hour plasma glucose profile in T 2 DM and healthy subjects 400 T 2 DM 300 15 200 10 100 Healthy subjects 0 06: 00 Meal 10: 00 14: 00 Meal 18: 00 22: 00 02: 00 5 Plasma glucose (mmol/l) Plasma glucose (mg/dl) 20 0 06: 00 Time of day (hours) Comparison of 24 -hour plasma glucose levels in healthy subjects vs patients with diabetes (p<0. 001). Adapted from Hirsch I et al Clin Diab 2005; 23: 78 -86 18
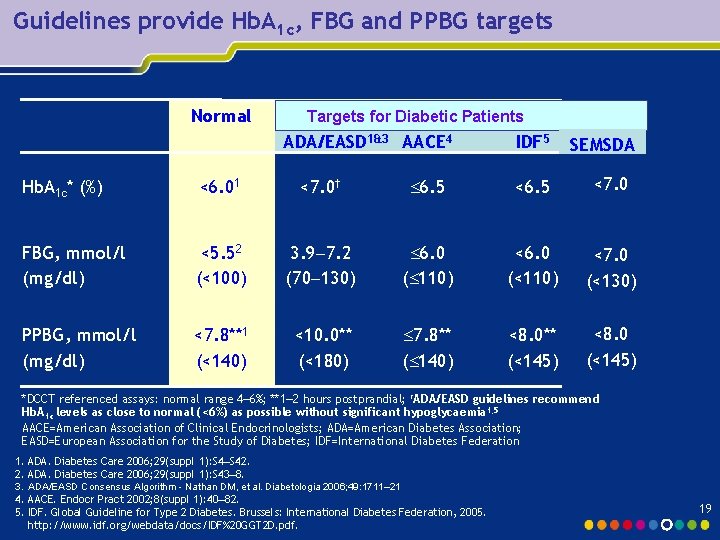
Guidelines provide Hb. A 1 c, FBG and PPBG targets Normal Targets for Diabetic Patients ADA/EASD 1&3 AACE 4 IDF 5 SEMSDA Hb. A 1 c* (%) <6. 01 <7. 0† 6. 5 <7. 0 FBG, mmol/l (mg/dl) <5. 52 (<100) 3. 9 7. 2 (70 130) 6. 0 ( 110) <6. 0 (<110) <7. 0 (<130) PPBG, mmol/l (mg/dl) <7. 8**1 (<140) <10. 0** (<180) 7. 8** ( 140) <8. 0** (<145) <8. 0 (<145) *DCCT referenced assays: normal range 4– 6%; **1– 2 hours postprandial; †ADA/EASD guidelines recommend Hb. A 1 c levels as close to normal (<6%) as possible without significant hypoglycaemia 1, 5 AACE=American Association of Clinical Endocrinologists; ADA=American Diabetes Association; EASD=European Association for the Study of Diabetes; IDF=International Diabetes Federation 1. ADA. Diabetes Care 2006; 29(suppl 1): S 4–S 42. 2. ADA. Diabetes Care 2006; 29(suppl 1): S 43– 8. 3. ADA/EASD Consensus Algorithm - Nathan DM, et al. Diabetologia 2006; 49: 1711– 21 4. AACE. Endocr Pract 2002; 8(suppl 1): 40– 82. 5. IDF. Global Guideline for Type 2 Diabetes. Brussels: International Diabetes Federation, 2005. http: //www. idf. org/webdata/docs/IDF%20 GGT 2 D. pdf. 19

SEMSDA Guidelines 2009 20
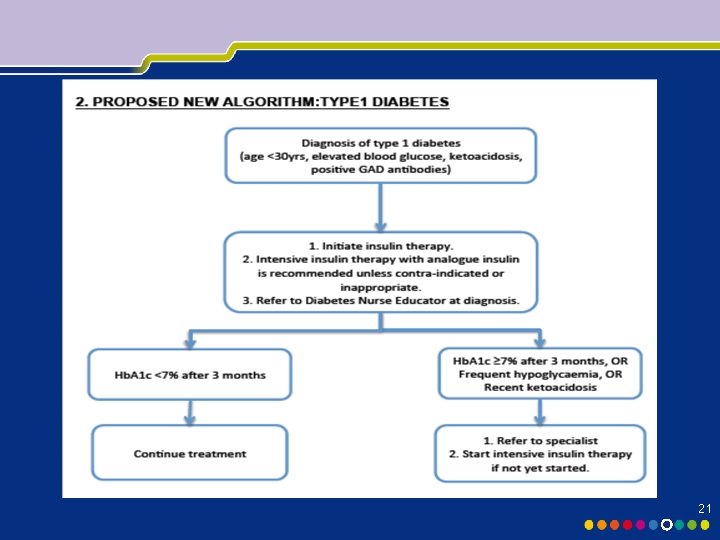
21
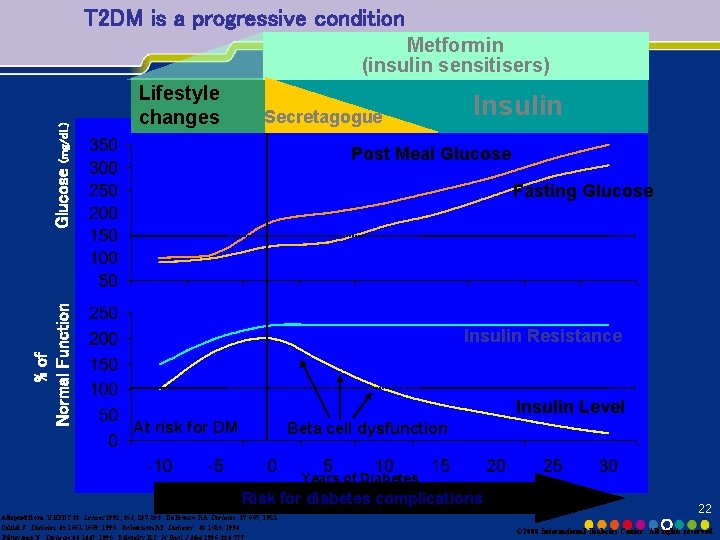
T 2 DM is a progressive condition Lifestyle changes Secretagogue Insulin Post Meal Glucose % of Normal Function Glucose (mg/d. L) Metformin (insulin sensitisers) Fasting Glucose Insulin Resistance Insulin Level At risk for DM Beta cell dysfunction Years of Diabetes Risk for diabetes complications Adapted from: UKPDS 33: Lancet 1998; 352, 837 -853 De. Fronzo RA. Diabetes. 37: 667, 1988. Saltiel J. Diabetes. 45: 1661 -1669, 1996. Robertson RP. Diabetes. 43: 1085, 1994. 22 © 2000 International Diabetes Center. All rights reserved

Insulin The most powerful agent we have to control glucose 23
Balancing Good Glycemic Control with a Low Risk of Hypoglycemia… Glycemic control Hypoglycemia 24

Imagine consulting room on Monday with the following: 59 years old male working as a financial manager – sedentary work most of the times during the week – works long hours. Over week-ends he enjoys hiking Type 2 diabetes diagnosed 12 years ago He has hypertension, dyslipidaemia. 25

Case study (continued) Current medication: § § § Glimepiride 4 mg/day Metformin 1 g bd Irbesartan 300 mg/day Rosuvastatin 10 mg/day Disprin CV 1/day 26

Case study (continued) § § Body mass index = 30 Waist circumference = 104. 5 cm BP = 120/74 mm Hg No diabetes complications § § § Hb. A 1 c = 8. 9% FBG of 9 -10 mmol/l PPBG up to 15 mmol/l 27

What will be prescribed? More than 200 combinations of insulin and oral agents are possible: § Intensified insulin – basal-bolus or MDI § Basal-plus § Conventional – premix § Basal-oral § Basal – Glargine, Detemir, NPH § Bolus – Glulisine, Lispro, Aspart, Human Regular, § Premix- Regular or Rapidacting bolus component § Different % combinations 28
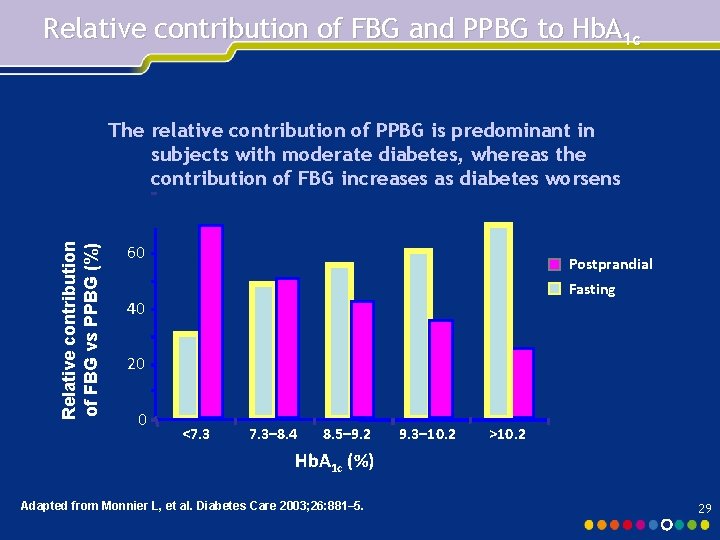
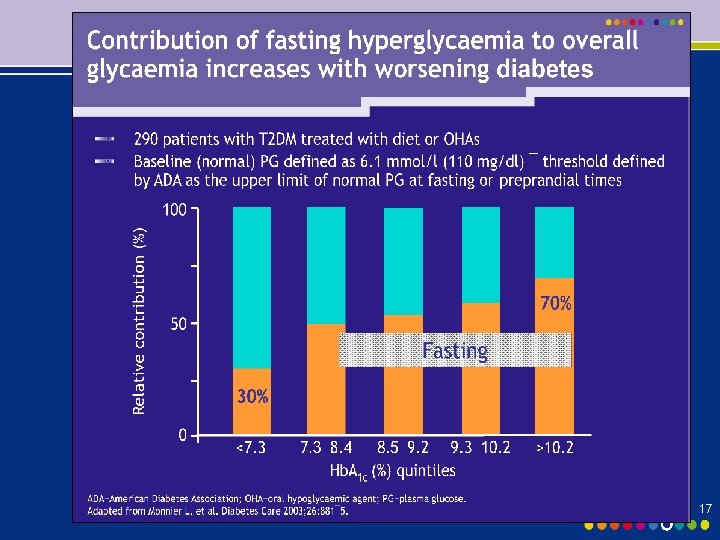
Relative contribution of FBG and PPBG to Hb. A 1 c Relative contribution of FBG vs PPBG (%) The relative contribution of PPBG is predominant in subjects with moderate diabetes, whereas the contribution of FBG increases as diabetes worsens 60 Postprandial Fasting 40 20 0 <7. 3– 8. 4 8. 5– 9. 2 9. 3– 10. 2 >10. 2 Hb. A 1 c (%) Adapted from Monnier L, et al. Diabetes Care 2003; 26: 881– 5. 29
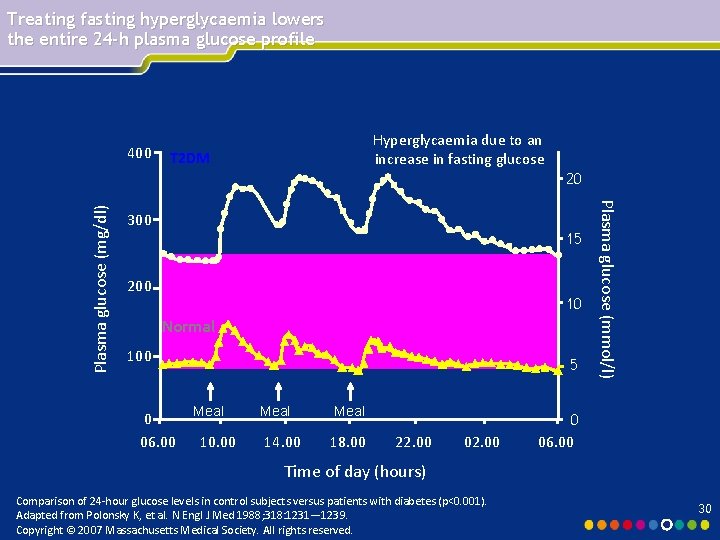
Treating fasting hyperglycaemia lowers the entire 24 -h plasma glucose profile 400 Hyperglycaemia due to an increase in fasting glucose T 2 DM 300 15 200 10 Normal 100 0 06. 00 5 Meal 10. 00 Meal 14. 00 Meal 18. 00 22. 00 02. 00 Plasma glucose (mmol/l) Plasma glucose (mg/dl) 20 0 06. 00 Time of day (hours) Comparison of 24 -hour glucose levels in control subjects versus patients with diabetes (p<0. 001). Adapted from Polonsky K, et al. N Engl J Med 1988; 318: 1231― 1239. Copyright © 2007 Massachusetts Medical Society. All rights reserved. 30

Case study l § § § Max OAD Hb. A 1 c = 8. 9% FBG of 9 -10 mmol/l PPBG up to 15 mmol/l Hb 32

More physiologic insulin replacement = Basal – Bolus/ Basal-Plus 33
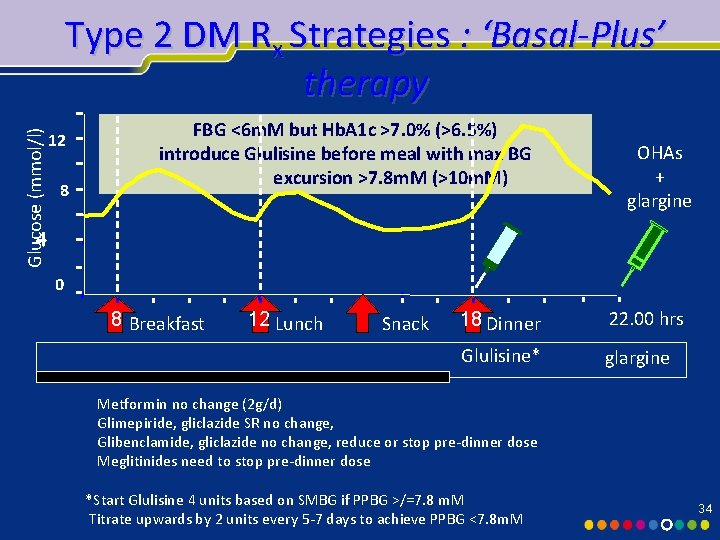
Glucose (mmol/l) Type 2 DM Rx Strategies : ‘Basal-Plus’ therapy 12 8 FBG <6 m. M but Hb. A 1 c >7. 0% (>6. 5%) introduce Glulisine before meal with max BG excursion >7. 8 m. M (>10 m. M) OHAs + glargine 4 0 8 Breakfast 12 Lunch Snack 18 Dinner 22. 00 hrs Glulisine* glargine Metformin no change (2 g/d) Glimepiride, gliclazide SR no change, Glibenclamide, gliclazide no change, reduce or stop pre-dinner dose Meglitinides need to stop pre-dinner dose *Start Glulisine 4 units based on SMBG if PPBG >/=7. 8 m. M Titrate upwards by 2 units every 5 -7 days to achieve PPBG <7. 8 m. M 34

Insulin treatment options in type 2 diabetes Prandial / Intensified Insulin Therapy Conventional Insulin Therapy 2 -3 injections Short acting insulin or rapid insulin mix of regular analog prandially insulin / rapidacting insulin + analog and long-acting insulin Basal insulin therapy l. Long-acting insulin +/- oral agents Basal Plus therapy long-acting insulin + rapid acting analog with 1 -2 meals +/- oral agents 35
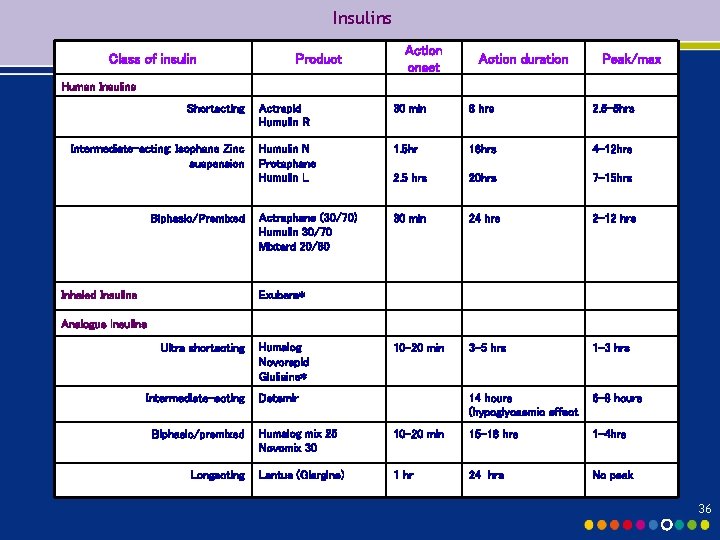
Insulins Class of insulin Product Action onset Action duration Peak/max Human Insulins Shortacting Intermediate-acting: Isophane Zinc suspension Biphasic/Premixed Inhaled Insulins Actrapid Humulin R 30 min 8 hrs 2. 5 -5 hrs Humulin N Protaphane Humulin L 1. 5 hr 16 hrs 4 -12 hrs 2. 5 hrs 20 hrs 7 -15 hrs Actraphane (30/70) Humulin 30/70 Mixtard 20/80 30 min 24 hrs 2 -12 hrs 10 -20 min 3 -5 hrs 1 -3 hrs 14 hours (hypoglycaemic effect 6 -8 hours Exubera* Analogue insulins Ultra shortacting Intermediate-acting Biphasic/premixed Longacting Humalog Novorapid Glulisine* Detemir Humalog mix 25 Novomix 30 10 -20 min 15 -18 hrs 1 -4 hrs Lantus (Glargine) 1 hr 24 hrs No peak 36
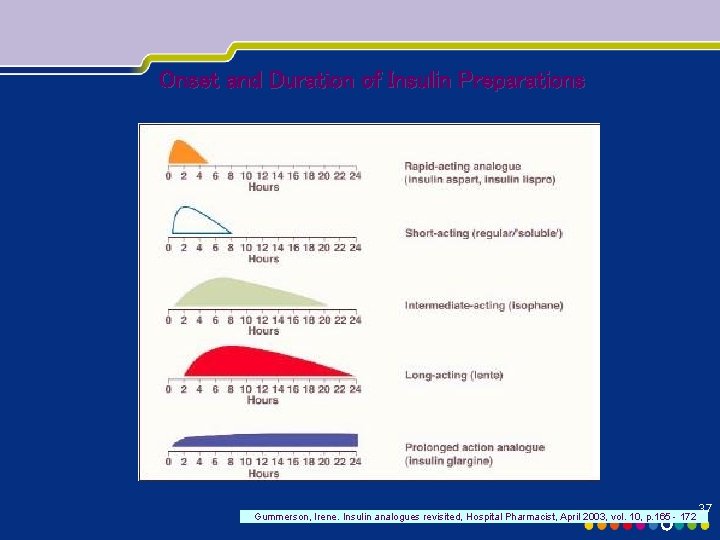
Onset and Duration of Insulin Preparations Gummerson, Irene. Insulin analogues revisited, Hospital Pharmacist, April 2003, vol. 10, p. 165 - 172 37

The Ideal Basal Insulin … Ø Mimics normal pancreatic basal insulin secretion Ø Long-lasting effect Ø Smooth peakless profile Ø Reproducible and predictable effects Ø Reduced nocturnal hypoglycemia Ø Once-daily administration for convenience Ø Pharmacodynamic effects similar to insulin pump 38
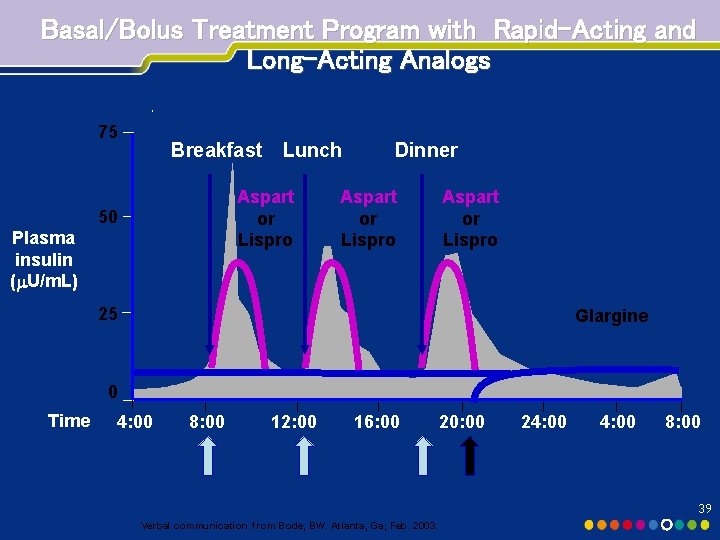
Basal/Bolus Treatment Program with Rapid-Acting and Long-Acting Analogs 75 Breakfast Lunch Aspart or Lispro 50 Plasma insulin ( U/m. L) Dinner Aspart or Lispro 25 Glargine 0 Time 4: 00 8: 00 12: 00 16: 00 20: 00 24: 00 8: 00 39 Verbal communication from Bode, BW. Atlanta, Ga; Feb. 2003.

Current Strategies for Improving the Therapeutic Potential of GLP-1 l Agents that mimic the actions of GLP-1 (incretin mimetics) § DPP-IV–resistant GLP-1 derivatives (dipeptidyl peptidase) § Examples: GLP-1 analogues, albumin bound GLP-1 § Novel peptides that mimic some of the glucoregulatory actions of GLP-1 § Exenatide - Victoza (Novo. Nordisk) - Syncria (GSK – Phase II) l Agents that prolong the activity of endogenous GLP-1 § DPP-IV Inhibitors Drucker DJ, et al. Diabetes Care. 2003; 26: 2929 -2940. ; Baggio LL, et al. Diabetes. 2004; 53: 2492 -2500. 40
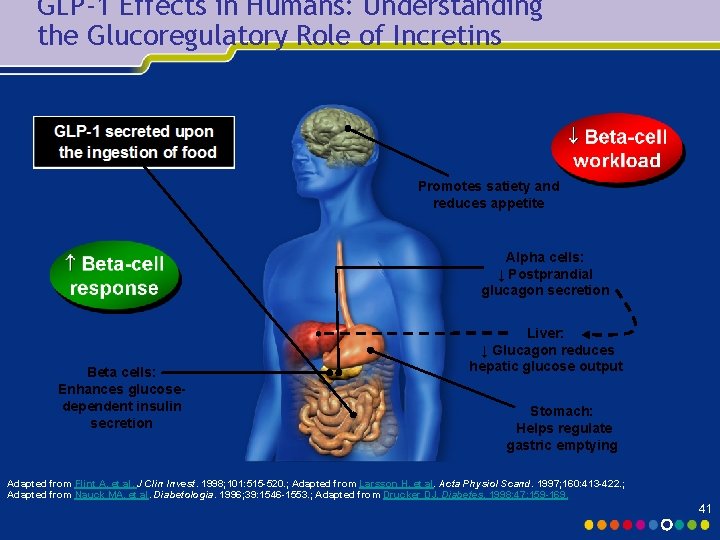
GLP-1 Effects in Humans: Understanding the Glucoregulatory Role of Incretins Promotes satiety and reduces appetite Alpha cells: ↓ Postprandial glucagon secretion Beta cells: Enhances glucosedependent insulin secretion Liver: ↓ Glucagon reduces hepatic glucose output Stomach: Helps regulate gastric emptying Adapted from Flint A, et al. J Clin Invest. 1998; 101: 515 -520. ; Adapted from Larsson H, et al. Acta Physiol Scand. 1997; 160: 413 -422. ; Adapted from Nauck MA, et al. Diabetologia. 1996; 39: 1546 -1553. ; Adapted from Drucker DJ. Diabetes. 1998; 47: 159 -169. 41
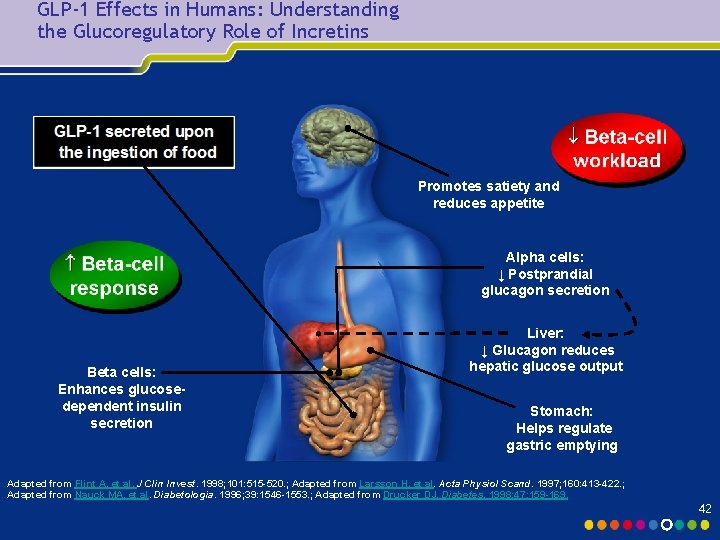
GLP-1 Effects in Humans: Understanding the Glucoregulatory Role of Incretins Promotes satiety and reduces appetite Alpha cells: ↓ Postprandial glucagon secretion Beta cells: Enhances glucosedependent insulin secretion Liver: ↓ Glucagon reduces hepatic glucose output Stomach: Helps regulate gastric emptying Adapted from Flint A, et al. J Clin Invest. 1998; 101: 515 -520. ; Adapted from Larsson H, et al. Acta Physiol Scand. 1997; 160: 413 -422. ; Adapted from Nauck MA, et al. Diabetologia. 1996; 39: 1546 -1553. ; Adapted from Drucker DJ. Diabetes. 1998; 47: 159 -169. 42
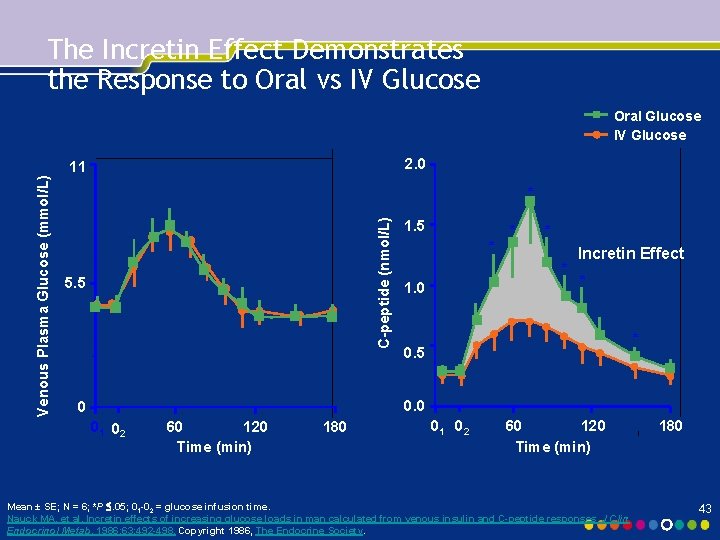
The Incretin Effect Demonstrates the Response to Oral vs IV Glucose 2. 0 11 * C-peptide (nmol/L) Venous Plasma Glucose (mmol/L) Oral Glucose IV Glucose 5. 5 1. 5 * * 1. 0 Incretin Effect * * 0. 5 0. 0 0 01 02 60 120 Time (min) 180 01 02 60 120 Time (min) Mean ± SE; N = 6; *P . 05; 01 -02 = glucose infusion time. Nauck MA, et al. Incretin effects of increasing glucose loads in man calculated from venous insulin and C-peptide responses. J Clin Endocrinol Metab. 1986; 63: 492 -498. Copyright 1986, The Endocrine Society. 180 43
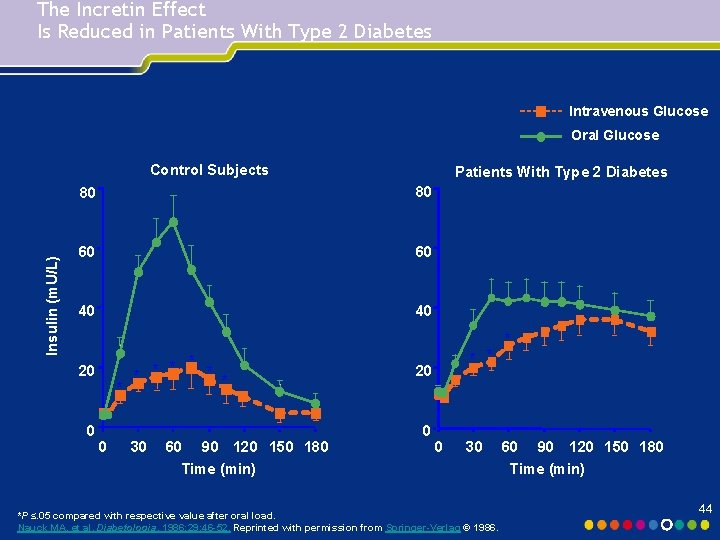
The Incretin Effect Is Reduced in Patients With Type 2 Diabetes Intravenous Glucose Oral Glucose Insulin (m. U/L) Control Subjects Patients With Type 2 Diabetes 80 80 60 60 40 40 * * * * 20 0 * * 20 * 0 0 30 60 90 120 150 180 Time (min) 0 30 *P ≤. 05 compared with respective value after oral load. Nauck MA, et al. Diabetologia. 1986; 29: 46 -52. Reprinted with permission from Springer-Verlag © 1986. 60 90 120 150 180 Time (min) 44
- Slides: 43