DIABETES MELLITUS BEYOND INSULIN AND GLUCAGON Seminar Presentation

DIABETES MELLITUS: BEYOND INSULIN AND GLUCAGON Seminar Presentation by: Luqman A. OYEKUNLE (Intern Med. Lab. Scientist) January, 2012

OUTLINE v. INTRODUCTION v. DIAGNOSIS v. HISTORICAL PERSPECTIVE v. HORMONAL ROLES v. HORMONAL ESTIMATION v. RECOMMENDATIONS v. REFERENCES

INTRODUCTION DIABETES MELLITUS (DM) va metabolic disorder characterized by elevated plasma glucose concentration (>7. 0 mmol/L) v. Basic types include: Type-1 DM Type-2 DM Gestational DM (Alkofide, 2010)

TYPE-1 DM v v an autoimmune disease the immune system attacks and destroys the insulin-producing beta cells in the pancreas v v little or no insulin is produced also called Non-Insulin dependent Diabetes mellitus (NIDDM) v daily administration of insulin required for management (Alkofide, 2010)

TYPE-2 DM vmost common form of diabetes vaccounts for about 90 -95% cases of diabetes vmost often associated with older age, obesity, family history of diabetes, previous history of gestational diabetes, physical inactivity, and certain ethnicities vinsulin resistance: the pancreas usually produces enough insulin, but for unknown reasons the body cannot use the insulin effectively (Alkofide, 2010)

TYPE-2 DM (metabolic syndrome) RISK FACTOR Abdominal obesity DEFINING LEVEL Waist circumference men >102 cm (> 40 in) women > 88 cm (>35 in) Triglycerides <150 mg/dl (8. 3 mmol/L) High density lipoprotein men <40 mg/dl (2. 2 mmol/L) women <50 mg/dl (2. 8 mmol/L) Blood pressure 130/85 mm. Hg Fasting glucose 110 mg/dl Five Components of the Metabolic Syndrome (Individuals having at least three components meet the criteria for diagnosis)

Diagnosis OF DM Criteria for the diagnosis of diabetes (Type 1 or Type 2) v FPG ≥ 126 mg/dl (7. 0 mmol/l) OR v Symptoms of hyperglycemia & a random plasma glucose ≥ 200 mg/dl (11. 1 mmol/l) OR v 2 -h plasma glucose ≥ 200 mg/dl (11. 1 mmol/l) (Alkofide, 2010)

TYPE-2 DM vsymptoms of type 2 DM develop gradually, not as sudden as Type-1 diabetes v Symptoms: fatigue, frequent urination, increased thirst and hunger, weight loss, blurred vision, and slow healing of wounds or sores v. Some people have no symptoms

Other types of diabetes Other types include those caused by: v genetic defects of the β-cell. e. g. neonatal diabetes mellitus (NDM) v diseases of the pancreas or conditions that damage the pancreas. e. g. pancreatitis, cystic fibrosis v medications that reduce insulin action, such as glucocorticoids, or chemicals that destroy beta cells v Infections. e. g. congenital rubella, cytomegalovirus v genetic syndromes associated with diabetes. e. g. Down syndrome and Prader-Willi syndrome (Alkofide, 2010)

HISTORICAL PERSPECTIVE (GLUCOSE HOMEOSTASIS) v 1920 s: understanding of diabetes, as a metabolic disease, evolved significantly since the discovery of insulin v. Subsequently, diabetes was viewed as a mono-hormonal disorder characterized by absolute or relative insulin deficiency (Hirsch, 1999, Buse et al. , 2001)

HISTORICAL PERSPECTIVE (GLUCOSE HOMEOSTASIS) v 1950 s: glucagon was characterized as a major stimulus of hepatic glucose production v This discovery led to a better understanding of the interplay between insulin and glucagon, thus leading to a bi-hormonal definition of diabetes (Lefebvre, 1995)

HISTORICAL PERSPECTIVE (GLUCOSE HOMEOSTASIS) v 1987: the discovery of a second β-cell hormone, amylin, was first reported. v Amylin was determined to have a role that complemented that of insulin, and, like insulin, was found to be deficient in people with diabetes. Hence, glucose homeostasis viewed as involving multiple pancreatic hormones (Moore and Cooper, 1991)

HISTORICAL PERSPECTIVE (GLUCOSE HOMEOSTASIS) v. Mid 1970 s: several gut hormones were identified, an incretin hormone, glucagon-like peptide-1 (GLP-1), was recognized as another important contributor to the maintenance of glucose homeostasis (Cooper et. al. , 2007, Naslund et al. , 2009)

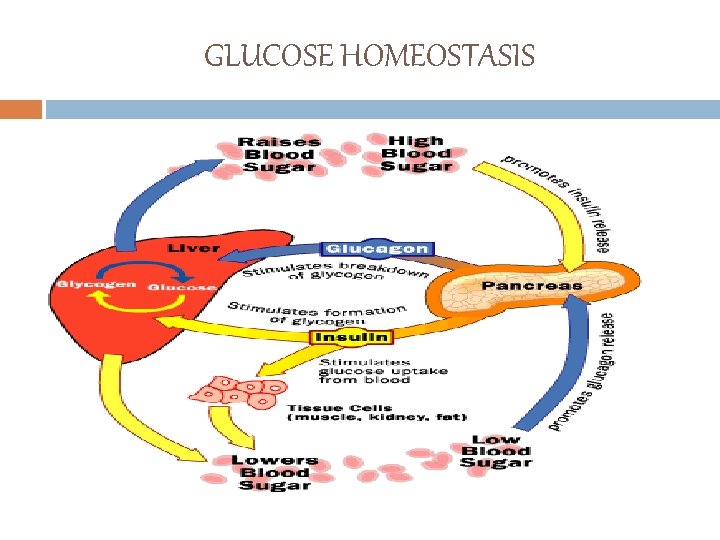
GLUCOSE HOMEOSTASIS v. Plasma glucose concentration is a function of the rate of glucose entering the circulation (glucose appearance) balanced by the rate of glucose removal from the circulation (glucose disappearance) v. Sources of circulating glucose v. Intestinal absorption (during the fed state) v. Hepatic processes ( Glycogenolysis and Gluconeogenesis) (Cryer, 1992)
GLUCOSE HOMEOSTASIS
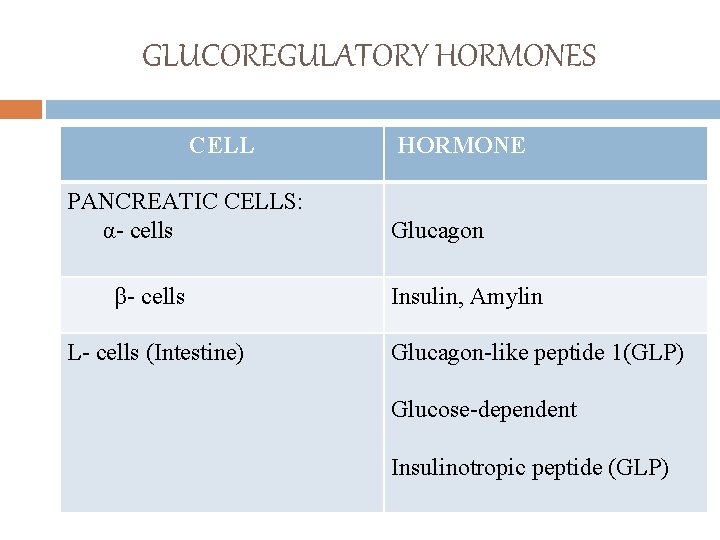
GLUCOREGULATORY HORMONES CELL PANCREATIC CELLS: α- cells β- cells L- cells (Intestine) HORMONE Glucagon Insulin, Amylin Glucagon-like peptide 1(GLP) Glucose-dependent Insulinotropic peptide (GLP)
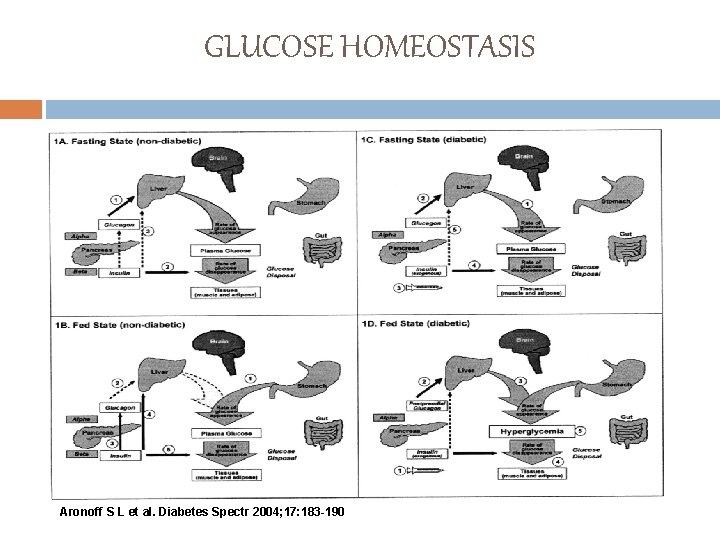
GLUCOSE HOMEOSTASIS Aronoff S L et al. Diabetes Spectr 2004; 17: 183 -190

GLUCOSE HOMEOSTASIS (Bi-hormonal model) Non-diabetic individuals v. Fasting state: plasma glucose derived from gluconeogenesis under the influence of glucagon Insulin’s role is minimal due to low secretion v. Fed state: plasma glucose derived from ingestion of nutrients Glucagon secretion suppressed through endogenous insulin secretion

GLUCOSE HOMEOSTASIS (Bi-hormonal model) Diabetic individuals (fasting state) v plasma glucose derived from glycogenolysis and gluconeogenesis, under the control of glucagon v. Exogenous insulin influences the rate of glucose disappearance, though, not properly regulated due to its deficiency in portal circulation

GLUCOSE HOMEOSTASIS (Bi-hormonal model) Diabetic individuals (Fed state) vexogenous insulin is ineffective in suppressing glucagon secretion, resulting in elevated hepatic glucose production Hence, the net effect is postprandial hyperglycaemia

β- cell pancreatic hormones INSULIN v. Composed of two polypeptide chains of 51 amino acids va major anabolic hormone secreted in response to increased blood glucose concentration vexerts its actions by binding to specific receptors on many body cells e. g. fat, liver and muscle cells v. Modes of actions (postprandial control): Signalling of insulin-sensitive cells Glycogenesis Inhibits glucagon secretion

β- cell pancreatic hormones AMYLIN v. Isolated from pancreatic amyloid deposits in the islets of langerhans v. First reported in literature in 1987 va 37 -amino acid peptide van Neuroendocrine hormone, cosecreted and coexpressed with insulin by pancreatic –cells in response to nutrient stimuli vhealthy individuals: amylin concentration= 4 -8 pmol/l but rises as high as 25 pmol/l postprandially (Moore and Cooper, 1991)

β- cell pancreatic hormones AMYLIN v works with insulin to help coordinate the rate of appearance and disappearance of glucose in circulation v Complements the effects of insulin on circulating glucose concentrations through 2 main mechanisms: v Suppresses post-prandial glucagon secretion v Slows the rate of gastric emptying (Pehling, 1984)
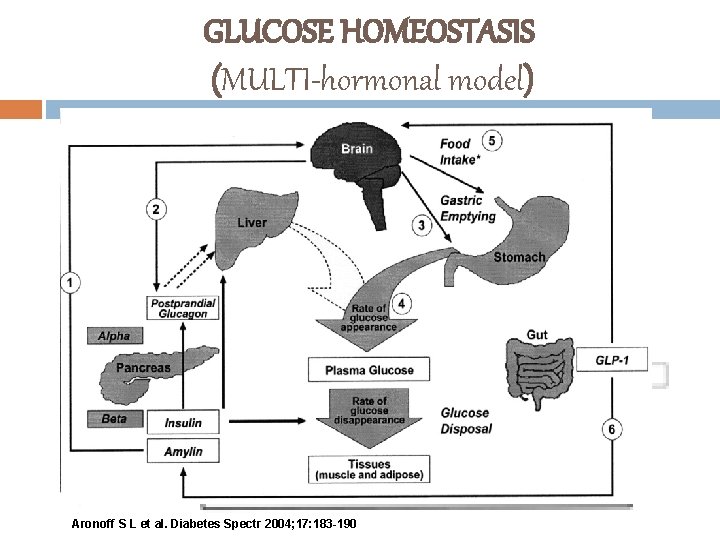
GLUCOSE HOMEOSTASIS (MULTI-hormonal model) Aronoff S L et al. Diabetes Spectr 2004; 17: 183 -190

α-CELL HORMONE: GLUCAGON va key catabolic hormone consisting of 29 amino acids, secreted from pancreatic α-cells v described by Roger Unger in the 1950 s, glucagon was characterized as opposing the effects of insulin. 38 vplays a major role in sustaining plasma glucose during fasting conditions by stimulating hepatic glucose production (Unger, 1971)

INCRETIN HORMONES (GLP-1 & gip) vhormones synthesized and secreted by the L-cells of the intestine (mainly in the ileum and colon) v dominant ones for glucose homeostasis are GIP and GLP-1. v The intricacies of glucose homeostasis become clearer when considering the role of gut peptides v late 1960 s: Perley and Kipnis and others demonstrated that ingested food caused a more potent release of insulin than glucose infused intravenously v This effect, termed the “incretin effect, ” suggested that signals from the gut are important in the hormonal regulation of glucose disappearance (Perley and Kipnis , 1987)

INCRETIN HORMONES (GLP-1 And gip) GIP (glucose-dependent insulinotropic peptide) v stimulates insulin secretion and regulates fat metabolism, but does not inhibit glucagon secretion or gastric emptying (Perley and Kipnis , 1987) v levels are normal or slightly elevated in people with type 2 diabetes (Yip and Wolfe, 2000) v. While GIP is a more potent incretin hormone, GLP-1 is secreted in greater concentrations and is more physiologically relevant in humans (Vilsboll et al. , 2001)

INCRETIN HORMONES (GLP-1 And gip) GLP-1(Glucagon-like peptide 1) v also stimulates glucose-dependent insulin secretion vsignificantly reduced postprandially in people with type 2 diabetes or impaired glucose tolerance vstimulates insulin secretion with increased plasma glucose concentrations (Yip and Wolfe, 2000, Lugari et al. , 2002)

ESTIMATION OF GUCOREGULATORY HORMONES v the aforementioned hormones can be measured by 2 possible methods: v. Radioimmunoassay method (RIA) v. Enzyme-linked immunossorbent assay method (ELISA) (Percy et al. , 2006)

RECOMMENDATIONS v. The fasting plasma glucose of all individuals, health-care providing personnels inclussive, should be determined to rule out diabetes v. All pregnant women should be screened for diabetes at 15 -20 weeks of gestation v. Other glucoregulatory hormones should be taken into consideration in the management of diabetes

REFERENCES Alkofide H: Pathophisology of diabetes, 2010 Buse JB, Weyer C, Maggs DG: Amylin replacement with pramlintide in type 1 and type 2 diabetes: a physiological approach to overcome barriers with insulin therapy. Clinical Diabetes 20: 137 – 144, 2002

REFERENCES Cooper GJS, Willis AC, Clark A, Turner RD, Sim RB, Reid KB: Purification and characterization of a peptide from amyloid-rich pancreas of type 2 diabetic patients. Proc Natl Acad Sci U S A 84: 8628 – 8632, 1987 Cryer PE: Glucose homeostasis and hypoglycaemia. In William's Textbook of Endocrinology. Wilson JD, Foster DW, Eds. Philadelphia, Pa. , W. B. Saunders Company, 1992, p. 1223 – 1253

REFERENCES Hirsch IB: Type 1 diabetes mellitus and the use of flexible insulin regimens. Am Fam Physician 60: 2343 – 2352, 2355– 2356, 1999 Koda JE, Fineman M, Rink TJ, Dailey GE, Muchmore DB, Linarelli LG: Amylin concentrations and glucose control. Lancet 339: 1179 – 1180, 1992 Lefebvre PJ: Glucagon and its family revisited. Diabetes Care 18 : 715– 730, 1995

REFERENCES Lugari R, Dei Cas A, Ugolotti D, Finardi L, Barilli AL, Ognibene C, Luciani A, Zandomeneghi R, Gnudi A: Evidence for early impairment of glucagonlike peptide 1 -induced insulin secretion in human type 2 (non insulindependent) diabetes. Horm Metab Res 34: 150 – 154, 2002 Moore CX, Cooper GJS: Co-secretion of amylin and insulin from cultured islet beta-cells: modulation by nutrient secretagogues, islet hormones and hypoglycaemic agents. Biochem Biophys Res Commun 179: 1 – 9, 1991

REFERENCES Naslund E, Bogefors J, Skogar S, Gryback P, Jacobsson H, Holst JJ, Hellstrom PM: GLP-1 slows solid gastric emptying and inhibits insulin, glucagon, and PYY release in humans. Am J Physiol 277: R 910 –R 916, 1999 Ogawa A, Harris V, Mc. Corkle SK, Unger RH, Luskey KL: Amylin secretion from the rat pancreas and its selective loss after streptozotocin treatment. J Clin Invest 85: 973 – 976, 1990

REFERENCES Pehling G, Tessari P, Gerich JE, Haymond MW, Service FJ, Rizza RA: Abnormal meal carbohydrate disposition in insulin-dependent diabetes: relative contributions of endogenous glucose production and initial splanchnic uptake and effect of intensive insulin therapy. J Clin Invest 74 : 985– 991, 1984 Percy AJ, Tramor DA, Rittenhouse J, Phelps Julie and Koda JE: Development of sensitive immunoassays to detect amylin and amylin-like peptides in unextracted plasma Clinical chemistry 42: 4 576 -585 (2006)

REFERENCES Perley MJ, Kipnis DM: Plasma insulin responses to oral and intravenous glucose: studies in normal and diabetic subjects. J Clin Invest 46 : 1954– 1962, 1967 Unger RH: Glucagon physiology and pathophysiology. N Engl J Med 285 : 443– 449, 1971

REFERENCES Vilsboll T, Krarup T, Deacon CF, Madsbad S, Holst JJ: Reduced postprandial concentrations of intact biologically active glucagon-like peptide 1 in type 2 diabetic patients. Diabetes 50: 609 – 613, 2001 Yip RG, Wolfe MM: GIP biology and fat metabolism. Life Sci 66 : 91– 103, 2000

THANKS FOR LISTENING
- Slides: 39