Diabetes Mellitus an overview Diabetes is a disorder

Diabetes Mellitus an overview

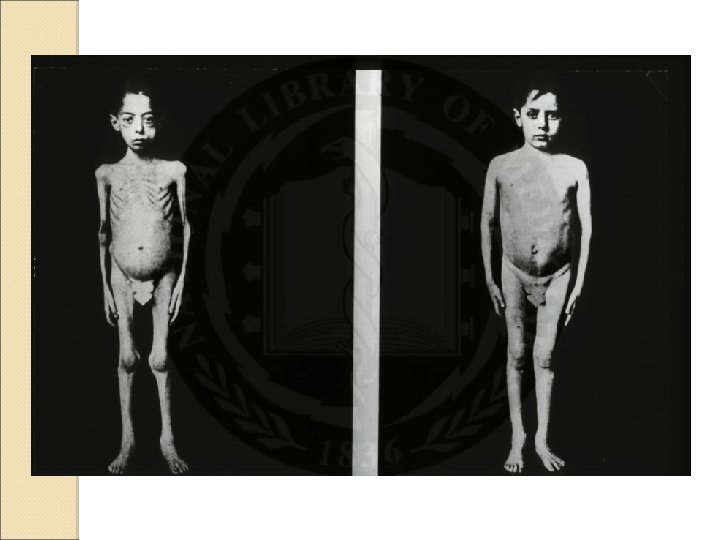
Diabetes is a disorder caused by the presence of too much glucose in the blood. A first depiction of this “sugar disease” was described in the “Ebers Papyrus”, a papyrus sold to the German Egyptologist Georg Moritz Ebers in 1872. It was said to have been found close to a mummy in the tomb of Thebes and appears to have been written between 3000 and 1500 BC.

History �Reference to diabetes was made 1550 BC. �In the 2 nd Century AD, Aretaeus gave an excellent description of diabetes. �Thomas Willis in the 17 th Century detected the sweet test of urine. �Mathew in the 18 th Century showed that the sugar in urine comes from the blood.

History �Minkowski and Von Mering discovered that disease of the pancreas is responsible for diabetes to develop in the 19 th century. �In the 19 th century treatment of diabetes was confined to food regulation which reduced urination but did not prevent wasting and complications.

History �In the second half of the 19 th Century, Paul Langerhans, a German student, identified clusters of cells within the pancreas responsible for the production on glucose lowering substance. “islets of Langerhans”. �Insulin: in Latin insula= island. So the name was coined before the hormone was discovered.

History �Banting and Best “a student” worked in Mc. Leod's labs in Toronto. �In 1921 they made the exocrine cells atrophy by ligation of the pancreatic duct. �They made aqueous extracts of the remaining tissue keeping it cold and filtered it. �The extract was injected into a diabetic dog on 30 July 1921.

History They convinced themselves that they had discovered the active pancreatic hormone which normalizes the blood sugar.

History The first person to be treated with insulin was Leonard Thompson (19081935). The first injection was in 11 January 1922

History: Noble Prize 1923 Banting Mc. Leod Best Collip

Definition of diabetes �A syndrome of chronic hyperglycaemia with other metabolic abnormalities together with micro and macro-vascular complications.

What is wrong with diabetes �Insulin deficiency resistance Hyperglycaemia

Classification of diabetes �Type 1 DM �Type 2 DM �IFG: impaired fasting glycaemia �IGT: impaired glucose tolerance �GDM: Gestational diabetes mellitus �Secondary DM.

Criteria of diagnosis �FBS > 126. MGS% �PP > 180 MGS% normal: �FBS - 80 – 100 MGS% �PP - 80 – 140 MGS%

T 1 DM �Usually in young age �Characterized by absolute insulin deficiency. �Increased catabolism and liability to ketosis. �Stormy presentation. �must be treated with insulin.

T 2 DM �Usually in older age. �Relative insulin deficiency. �Increased insulin resistance. �Can be treated with OHA or insulin. �Slow onset, less likely to develop ketosis. �May present with complications.

MODY Maturity onset diabetes of the youth �A special type of diabetes similar to type 2 diabetes but develop in young age groups. �Increased prevalence worldwide. �Associated with increased childhood obesity.

Diabetes related to drugs �Glucocorticoids �Diazoxide. �Thiazides. �Phyention �Pentamidine

GDM Gestational diabetes mellitus �Diabetes discovered for the first time during pregnancy. �Every pregnant lady should be screened. �Usually disappears after labor. �Increased risk to develop T 2 DM later in life.
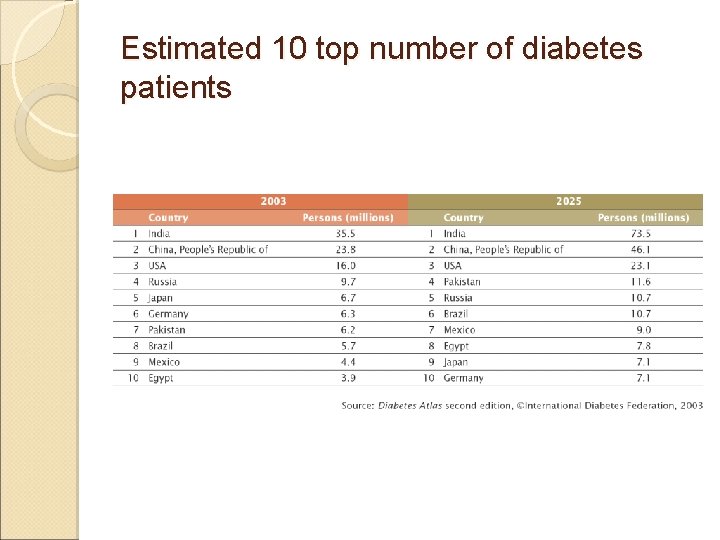
Estimated 10 top number of diabetes patients

Diagnosis � How to diagnose diabetes: 1. 2. 3. 4. Signs and symptoms Blood glucose test OGTT Hb. A 1 c

Diagnosis �Most people are diagnosed with diabetes when they are suspected to have symptoms of polyurea, polydepsia, fatigue, loss of weight. �This is confirmed by fasting or PP blood glucose. �In case of doubt OGTT may be done. �Urine testing should not be used in diagnosis.

Diagnosis �Peers and medical ‘advisors’ should be aware of the following: �T 1 DM & T 2 DM are two distinct diseases. �T 1 DM is stormy at presentation, delay in diagnosis can be disastrous. �Among the presentations of T 1 DM could be some non-specific symptoms like vomiting, abdominal pain….

Diagnosis �T 2 DM may present with late symptoms, like numpness, disturbed vision, generalized oedema. �Patients with hypertension, dyslipidaemia, MI and family history of diabetes are very likely to develop T 2 DM.

Pathophysiology of T 1 DM Absence of insulin secretion Failure to use glucose as a fuel Hyperglycaemia & using fat Ketosis

Pathophysiology of T 1 DM � Possible contributing factors: 1. 2. 3. 4. Autoimmune disease. HLA typing Viruses chemicals

Pathophysiology of T 2 DM Insulin resistance hyperinsulinaemia Relative hypoinsulinaemia Hyperglycaemia, dyslipidaemia, atherosclerosis, HTN

Pathophysiology of T 2 DM � Causes of insulin resistance: 1. 2. 3. 4. 5. Hereditary. Decreased glucose transporters. Decreased insulin receptors Post receptor mechanisms Chemical mediators e. g. TNFα

Pathophysiology of T 2 DM �Loss of first phase of insulin secretion. �Delayed insulin release.
Insulin
Insulin

Insulin � Action of insulin: 1. On glucose metabolism 2. On amino acid metabolism 3. On lipid metabolism
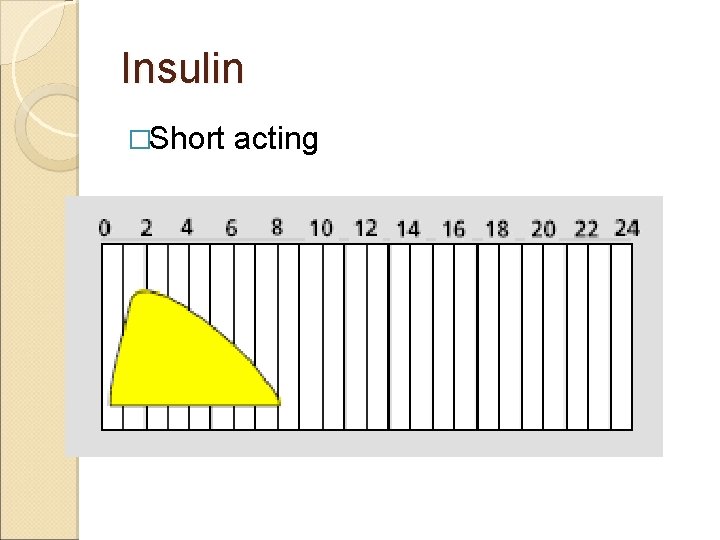
Insulin �Short acting
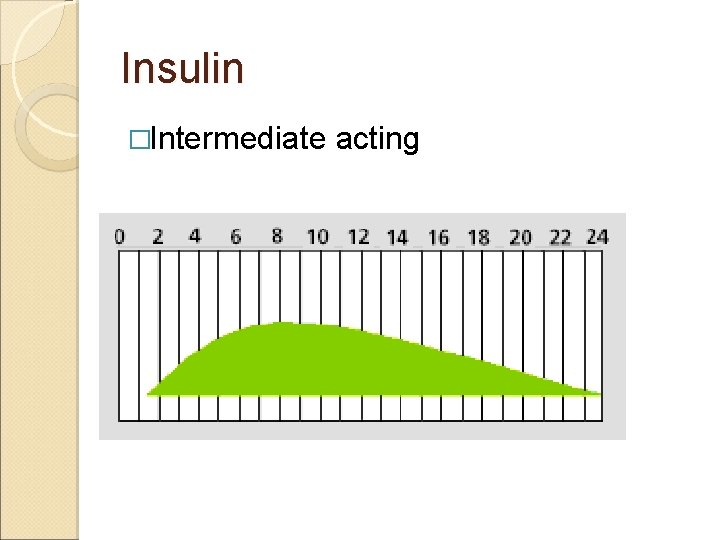
Insulin �Intermediate acting
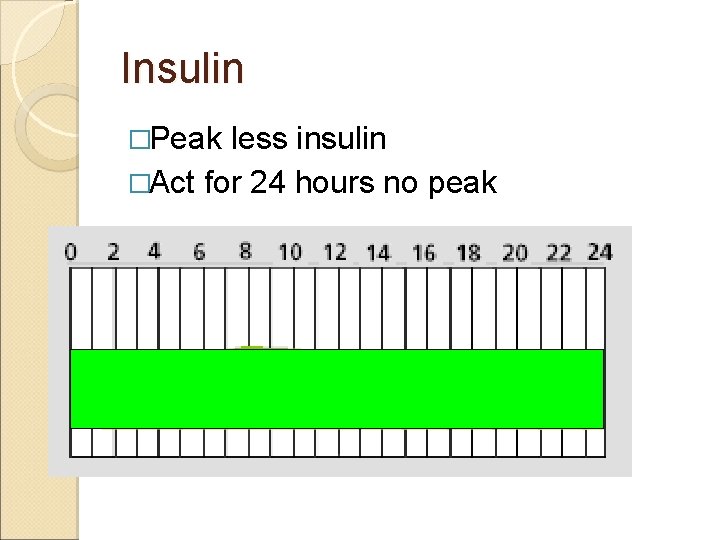
Insulin �Peak less insulin �Act for 24 hours no peak
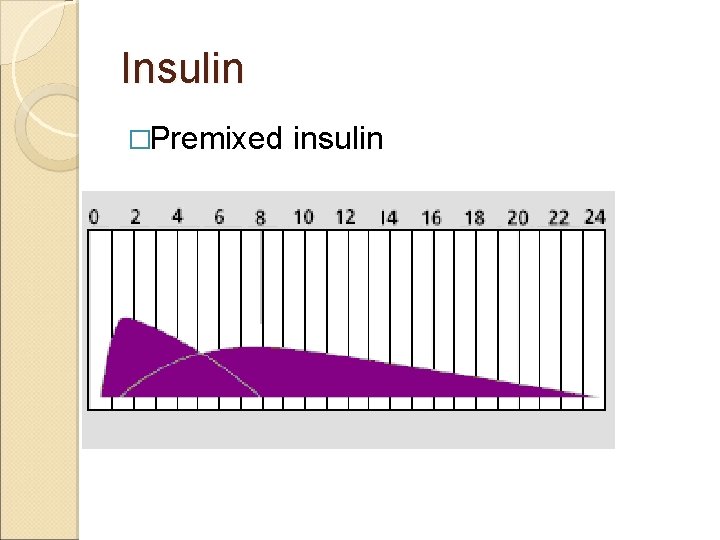
Insulin �Premixed insulin
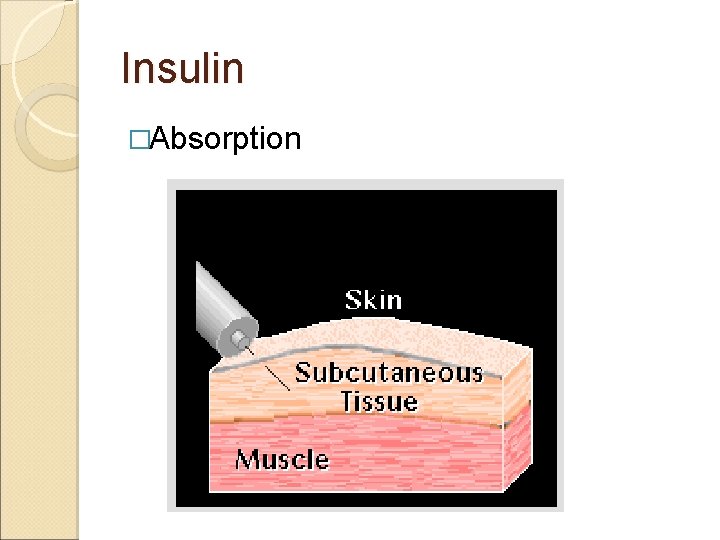
Insulin �Absorption

Insulin � Variation of absorption: 1. 2. 3. 4. 5. Type Dose Site of preparation Temperature. circulation
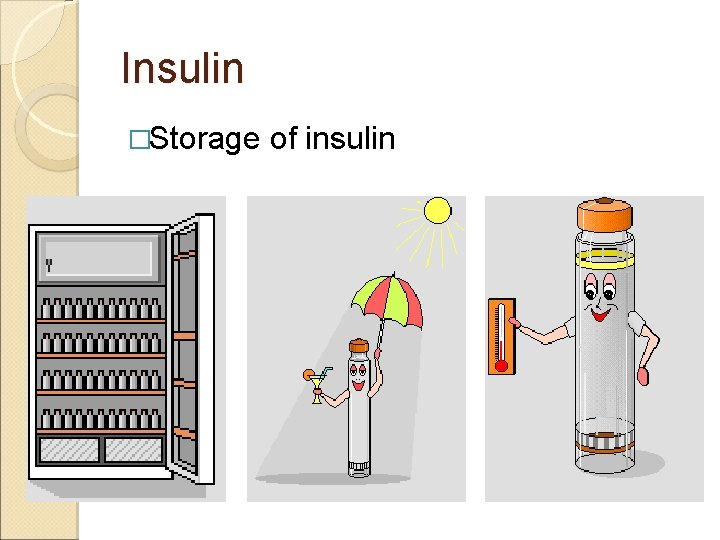
Insulin �Storage of insulin
Insulin �injection
insulin �injection:
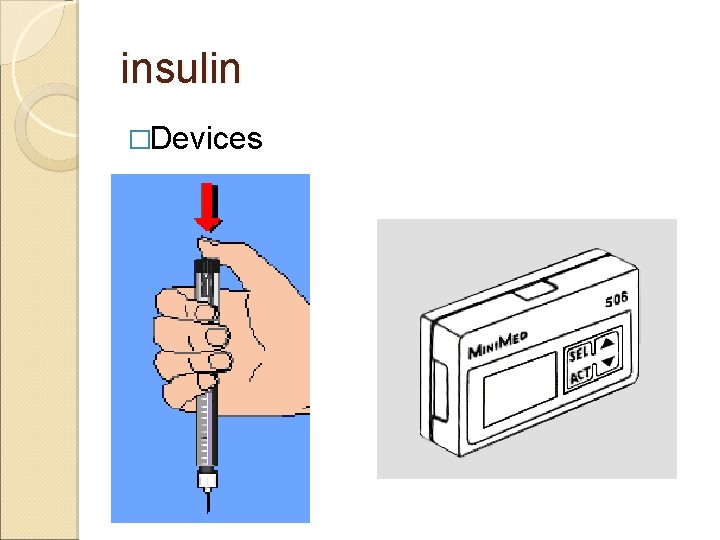
insulin �Devices

Insulin � Side effect: 1. 2. 3. 4. 5. Hypoglycaemia Atrophy Hypertrophy Sensitivity Weight gain

Diet � Rules: 1. 2. 3. 4. Balanced meal Maintain body weight Adequate nutrition Regular meal time.

Diet

OHA

OHA � Sulphonylureas: 1. 2. 3. 4. Mode of action Side effect Differences Use

OHA � Metformin: 1. 2. 3. 4. Action When to use Side effects Warning.

OHA � Acarbose 1. Action 2. Effect 3. Side effect use

OHA � Non Sulphonylureas insulin secreatgauges: 1. Repaglinide 2. Natiglinide.

OHA � Insulin sensitizers: 1. 2. 3. 4. Mode of action Effect Side effect use

Sulfonylureas e. g. Chlorpropamide, Glyburide �Mechanism ◦ Increase insulin secretion by pancreas �Advantages ◦ Well established, Decrease microvascular risk, Convenient dosing �Disadvantages ◦ Hypoglycemia, Weight gain �FDA Approval for combination therapy ◦ Metformin, TZD, acarbose Adapted from SE Inzucchi, JAMA 2002; 287: 360 -372.

Non-SU Secretagogues e. g. Nateglinide, Repaglinide �Mechanism ◦ Increase insulin secretion by pancreas �Advantages ◦ Targets post-prandial glycemia �Disadvantages ◦ TID dosing, No long-term data �FDA Approval for combination therapy ◦ Metformin Adapted from SE Inzucchi, JAMA 2002; 287: 360 -372.

Biguanides e. g. Metformin �Mechanism ◦ Decrease hepatic glucose production �Advantages ◦ Well established, Weight loss, No hypoglycemia, Decrease micro & macrovascular risk, Convenient dosing, [Also prevents diabetes] �Disadvantages ◦ GI distress, Lactic acidosis, Contraindications �FDA Approval for combination therapy ◦ Insulin, SU and non-SU secretagogues, TZD Adapted from SE Inzucchi, JAMA 2002; 287: 360 -372.

Alpha-Glucosidase Inhibitors e. g. Acarbose, Miglitol �Mechanism ◦ Decrease gut carbohydrate absorption �Advantages ◦ Targets post-prandial hyperglycemia, No systemic absorption, [Also prevents diabetes] �Disadvantages ◦ GI distress, TID dosing, No long-term data �FDA Approval for combination therapy ◦ Sulfonylureas Adapted from SE Inzucchi, JAMA 2002; 287: 360 -372.

Thiazolidindiones e. g. Pioglitazone, Rosiglitazone �Mechanism ◦ Increase peripheral glucose disposal �Advantages ◦ Physiologically “correct, ” Convenient dosing, [Also prevents diabetes] �Disadvantages ◦ Liver toxicity, Liver monitoring, Weight gain, Edema, No long-term data �FDA Approval for combination therapy ◦ Insulin, sulfonylurea, metformin Adapted from SE Inzucchi, JAMA 2002; 287: 360 -372.

Acute complications of Diabetes Mellitus

Hypoglycaemia

Hypoglycaemia �Most common complication of diabetes ◦ 100% of Type 1 patients affected ◦ ~ 10%/year severe (requiring assistance) ◦ much less common in Type 2 �Multiple causes: ◦ exercise/activity ◦ reduced food intake ◦ delayed meal drug overdose alcohol use

Symptoms of Hypoglycemia Adrenergic Neuroglycopenic tachycardia palpitations sweating tremor hunger dizziness confusion sleepiness coma seizure

Hypoglycemia Symptoms and Signs • Sweating, tremors, pounding heart beats. • Pallor, cold sweat, irritability • May develop coma. 63

Prevention of Hypoglycemia �Consistent meal times, appropriate to drug regimen �Consistent carbohydrate intake, or matched to drug dose �Adjustments for extra exercise ◦ extra food, e. g. 15 gm carb/30 min ◦ reduce drug, e. g. prior dose by 20 -30% �Accurate drug dosing �Blood glucose monitoring

Treatment of Hypoglycemia �Oral carbohydrate: ◦ 10 -15 gms, repeat after 15 minutes if needed ◦ glucose tabs preferred; food acts slower, adds unneeded calories (fat, protein) �IV Glucose ◦ 20 -50 cc of D 50 �Glucagon ◦ 1 mg IM

Hyperosmolar Hyperglycemic Nonketotic Syndrome

Hyperosmolar Hyperglycemic Nonketotic Syndrome Clinical presentation Severe hyperglycemia (BG > 600) No or minimal ketosis Hyperosmolarity Profound dehydration Altered mental status

Causes of HHNS �Drugs: glucocorticoids, diuretics �Acute stressors: infection, burns, CVA, MI, gastroenteritis �Other chronic disease: renal, heart, old stroke �Procedures: surgery

Prevention of HHNS �Awareness of the syndrome �Maintenance of adequate hydration �Control of blood glucose during acute stress with insulin

DIABETIC KETOACIDOSIS

Diabetic Ketoacidosis �An acute, life threatening metabolic acidosis complicating IDDM and some cases of NIDDM with intercurrent illness (infection or surgery) �Usually coupled with an increase in glucagon concentration with two metabolic consequences: ◦ 1) Maximal gluconeogenesis with impaired peripheral utilization of glucose ◦ 2) Activation of the ketogenic process and development of metabolic acidosis.

Diabetic Ketoacidosis �Usually seen in Type 1 DM, but CAN OCCUR in Type 2 �Often with acute stress, such as infection, MI, etc. �Recurrent DKA almost always related to omission of insulin, psychosocial problems �Preventive measures same as for HHNS

Clinical Presentation � Anorexia, N/V, along with polydepsia and polyuria for about 24 hrs. followed by stupor (or coma). � Abdominal pain and tenderness could be present (remember DDx of acute abdomen). � Kussmaul breathing with fruity odor “acetone” � Sings of dehydration ( HR, postural BP, etc. ) � Normal or low temperature: NB. : if fever is present it suggests infection while leukocytosis alone is not because DKA per se can cause fever.

Has to be treated in Hospital Always refer to Endocrinologist • Insulin: is a prerequisite for recovery • IVF: the usual fluid deficit is 3 -5 L • Potassium: replacement is always necessary • Bicarbonate:

Acute Complications of Diabetes SUMMARY: �Acute complications can be prevented or greatly reduced �Prevention depends on effective patient education

Chronic complications of Diabetes Mellitus

Causes of Death Among People With Diabetes Cause % of Deaths Ischemic heart disease 40 Other heart disease 15 Diabetes (acute complications) 13 Cancer 13 Cerebrovascular disease 10 Pneumonia/influenza 4 All other causes 5 Geiss LS et al. In: Diabetes in America. 2 nd ed. 1995: 233 -257.
Complications of Diabetes: Long term ◦ Macrovascular �Ischaemic heart disease – heart attacks; stroke �Peripheral vascular disease – gangrene, amputations ◦ Microvascular �EYE – retinopathy - blindness �NERVE - neuropathy (peripheral and autonomic) �KIDNEY – nephropathy; dialysis ◦ Infections

Magnitude of Problem �Diabetic retinopathy: most common cause of blindness before age 65 �Nephropathy: most common cause of ESRD �Neuropathy: most common cause of non-traumatic amputations � 2 -3 fold increase in cardiovascular disease

Microvascular Complications �Diabetic retinopathy �background retinopathy �macular edema �proliferative retinopathy �Diabetic nephropathy �Diabetic neuropathy �distal symmetrical polyneuropathy �mononeuropathy (peripheral, cranial nerves) �autonomic neuropathy

chronic complications* population based - Egyptians • prevalence known D – retinopathy – nephrop. – neuropathy – foot ulcers • 41. 5 6. 7 21. 9 0. 8 associations ret; nephr; *microvasc + neuropathic; new D % 15. 7 6. 8 13. 6 0. 8 neuro : glucose n: 1451

Retinopathy and Blindness in Diabetes Patients ◦ It is estimated that retinopathy affects 80%-97% of patients with diabetes of ³ 15 years’ duration ◦ Diabetes is the leading cause of new cases of blindness in adults* ◦ Diabetic retinopathy accounts for the majority of these cases ◦ Minimum cost of blindness for working-age adult is estimated at $12, 769 per year *Blindness is defined as visual acuity £ 20/200 Klein R, Klein BEK. In: Diabetes in America. 2 nd ed. 1995: 293 -338.

Diabetic Retinopathy �Background retinopathy ◦ present in 90% of patients after 10 years ◦ asymptomatic ◦ red dots (microaneurysms) ◦ dot, blot, and flame shaped hemorrhages ◦ hard waxy exudates of lipid and protein ◦ best detected by dilated eye exam or photos
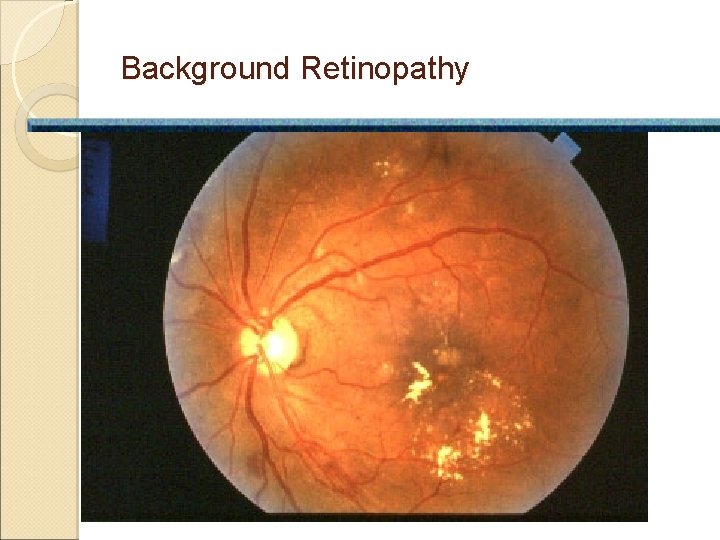
Background Retinopathy

Diabetic Retinopathy �Macular edema ◦ sight threatening edema of the macula ◦ usually reduces visual acuity early ◦ can only be diagnosed by ophthalmologic exam ◦ focal photocoagulation reduces risk of blindness by 50%
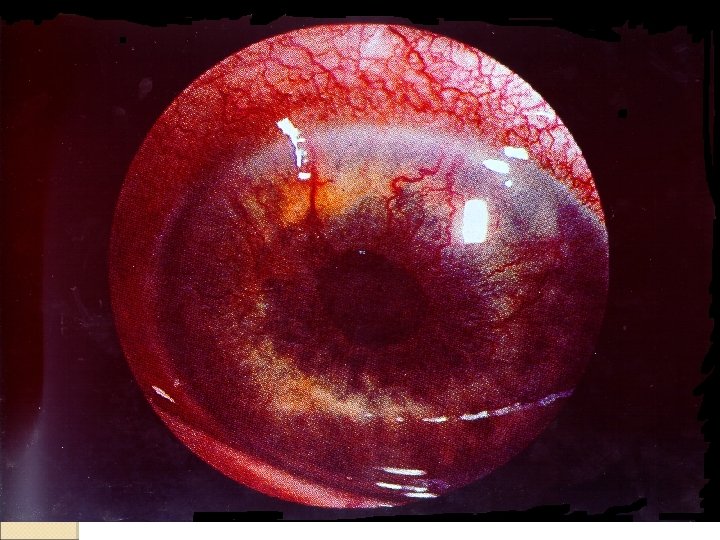

Diabetic Retinopathy �Proliferative retinopathy ◦ growth of small, fragile blood vessels that may bleed (vitreous hemorrhage) ◦ associated with growth of fibrous tissue that may cause retinal detachment ◦ may occur on the optic disk or elsewhere ◦ high risk of blindness (50% in 3 years) ◦ hypertension, isometric exercise, high contact sports may increase risk of bleeding
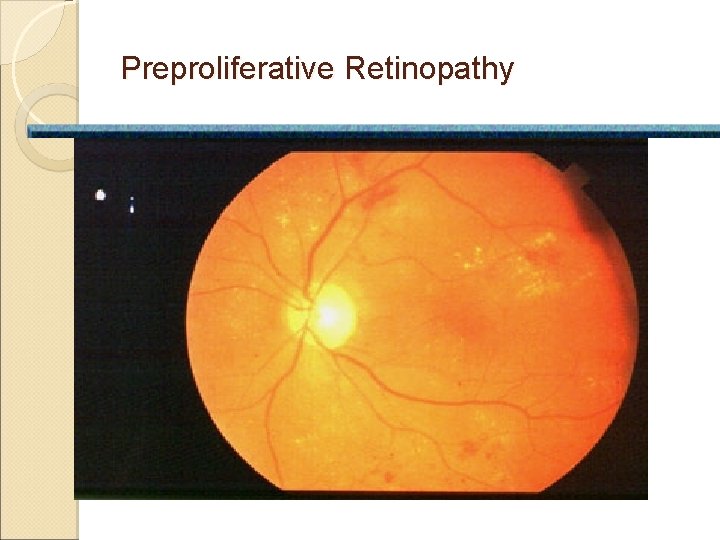
Preproliferative Retinopathy

Kidney Disease in Diabetes Patients ◦ 27, 851 new cases of ESRD in diabetes patients in 1995 � 40% of all new cases in the US ◦ Nearly 99, 000 diabetes patients required dialysis or kidney transplantation that year ◦ Annual cost of ESRD: �$45, 000 in diabetic patients ages 45 -64 National Diabetes Fact Sheet. November 1, 1997: 1 -8. U. S. Renal Data System, USRDS 1997 Annual Data Report.
- Slides: 87