DIABETES CASE PRESENTATIONS 3 rd Chronic complications 1

DIABETES CASE PRESENTATIONS 3 rd – Chronic complications

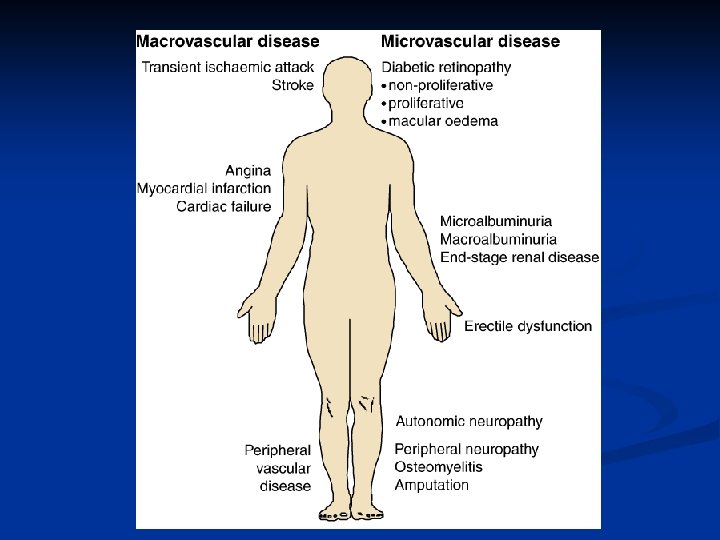
1. Diabetic retinopathy n Early subclinical abnormalities of the retinal vessels: n thickening of the basement membrane n loss of pericytes (the contractile cells that control vessel calibre and flow) n increased blood flow n increased capillary permeability (leakage)

Simplified grading of diabetic retinopathy n Background (Level 1) n n n Preproliferative (Level 2) n n n Venous beading Venous loop or reduplication Intraretinal microvascular abnormalities (IRMA) Multiple deep, round haemorrhages Cotton-wool spots Proliferative (Level 3) n n n Microaneurysms Retinal haemorrhages any exudates (hard exudates) New vessels on disc (NVD) New vessels elsewhere (NVE) Preretinal or vitreous hemorrhage Preretinal fibrosis tractional retinal detatchment Maculopathy n n n Exudate within a disc area (DA) of fovea Retinal thickening within DA of fovea Microaneurysm or hemorrhage within DA of fovea

Screening for diabetic retinopathy n Regular examination of the eye in the diabetic patient is essential n n yearly examinations for those with no retinopathy n n n visual acuity measurement examination of the fundus through dilated pupils digital fundus photography from puberty / 5 yrs after diagnosis for T 1 DM from diagnosis for T 2 DM 6 -monthly for those with background retinopathy

Treatment for DR Strict glycaemic control n Strict blood pressure control n ACE inhibitors n Early detection n Prompt treatment n n n Panretinal photocoagulation Education

2. Diabetic nephropathy n n Pathological changes: glomerulus + tubular interstitium Glomerulus: n at diagnosis of DM - enlarged because of the increased capillary surface area n subsequently, glomerular enlargement is caused by basement membrane thickening and (usually) expansion of the mesangium n Tubular interstitium: n total kidney volume is also increased, mainly through expansion of tubular tissue n basement membrane thickening n atrophy n interstitial fibrosis n arteriosclerosis

n Glomerular filtration rate (GFR): early in diabetes – increased because of the increased filtration area n later declines in parallel with mesangial expansion and the resultant glomerular occlusion n n The natural history of diabetic nephropathy is marked by increasing loss of protein (mostly albumin) in the urine
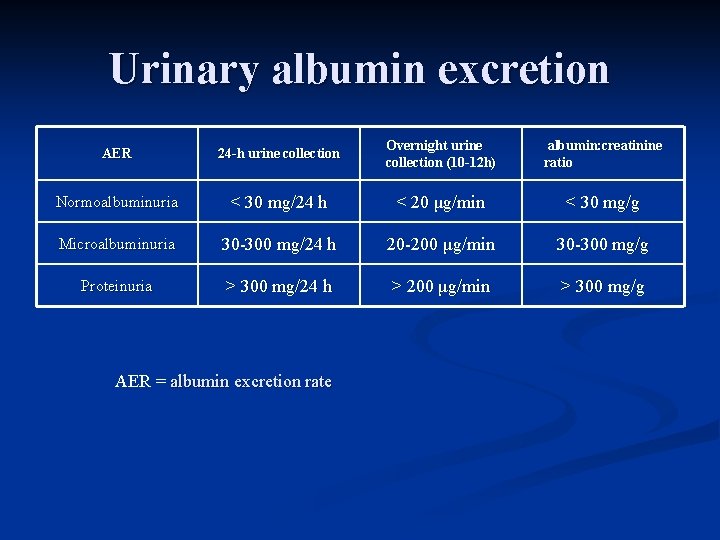
Urinary albumin excretion AER 24 -h urine collection Overnight urine collection (10 -12 h) albumin: creatinine ratio Normoalbuminuria < 30 mg/24 h < 20 μg/min < 30 mg/g Microalbuminuria 30 -300 mg/24 h 20 -200 μg/min 30 -300 mg/g Proteinuria > 300 mg/24 h > 200 μg/min > 300 mg/g AER = albumin excretion rate
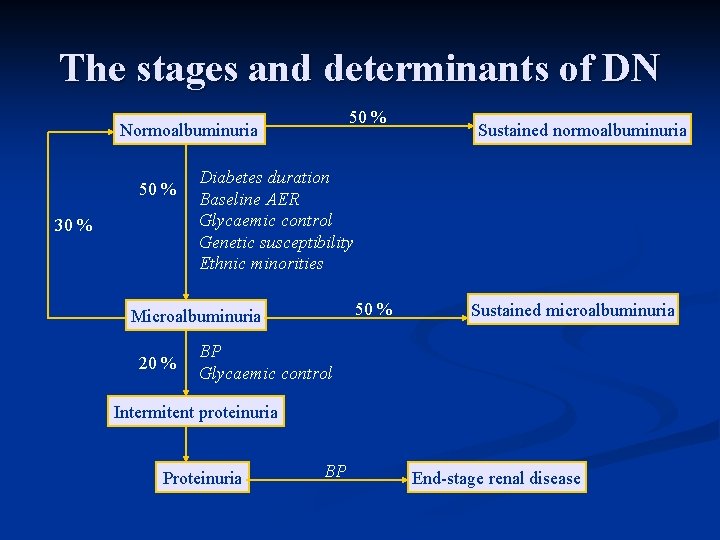
The stages and determinants of DN 50 % Normoalbuminuria 50 % 30 % Diabetes duration Baseline AER Glycaemic control Genetic susceptibility Ethnic minorities 50 % Microalbuminuria 20 % Sustained normoalbuminuria Sustained microalbuminuria BP Glycaemic control Intermitent proteinuria Proteinuria BP End-stage renal disease

Mogensen classification (1) Stage I (renal hyperfunction / hypertrophy) n n n n from diagnosis of DM good glycemic control → reversible GFR by 20 -50 % (>150 ml/min/1, 73 m 2) possible microalbuminuria (transitory!) size of kidneys (ultrasonography) and of glomerules (biopsy) Electronic microscope: normal basal membrane normal BP

Mogensen classification (2) Stage II – asymptomatic, normoalbuminuria n n n n first 5 yrs from diagnosis of DM 5 -7 yrs from onset of disease: 30 -50 % → stage III biopsy: thickening of glomerular basal membrane, mesangium expansion GFR still ↑ (by 20 -50 %) normal urine albumin excretion possible microalbuminuria (transitory!) normal BP

Mogensen classification (3) Stage III (early diabetic nephropathy) n 6 -15 yrs from diagnosis of DM n blood glucose and BP comtrol → stops/decreases progression n persistent microalbuminuria (30 -300 mg/24 hrs) n GFR still , but decreases by 3 -5 ml/min/yr n BP normal or mildly ( by 3 mm. Hg/yr)

Mogensen classification (4) Stage IV (clinical diabetic nephropathy) n n 15 -25 yrs after onset of DM proteinuria (albuminuria > 300 mg/24 hrs) GFR progressively (by 8 -12 ml/min/yr) 3 sub-classes: n n early (GFR > 130 ml/min) intermediate (GFR < 100 ml/min) advanced (GFR < 70 ml/min) BP (by 5 mm. Hg/yr)

Mogensen classification (5) Stage V (end-stage renal failure) n n n 25 -30 yrs after onset of DM variable proteinuria urine albumin excretion < 10 g/24 hrs GFR < 10 ml/min BP constantly Microscope: important glomerular modifications

Screening for diabetic nephropathy Annual determining of microalbuminuria: n n Type 1 DM: n from puberty n 5 yrs after diagnosis Type 2 DM : n n from diagnosis How? n minimum 3 samples in 3 -6 months n avoid conditions that can produce transient urinary albumin excretions n persistent microalbuminuria = 2 out of 3 results of 30 -300 mg/24 hrs, 20 -200 µg/min or urine albumin/creatinine = 30 -300 mg/g

Tratament of diabetic nephropathy Objectives: n Good glycemic control n Treat high BP n Diet n Treat dyslipidemia n Prevent / treat related comorbidities n Treat renal anemia n Prevent / treat renal bone disease

Treating hypertension in patients with diabetic nephropathy n Non-pharmacologically: n n n n exercise weight loss low-sodium diet low-protein diet smoking cessation avoid alcohol coffee Drugs: n n n ACE inhibitors ARBs diuretics Calcium blockers beta-blockers -adrenergic blockers

3. Diabetic neuropathy n Acute reversible n n hyperglycemic neuropathy Persistent n Symmetrical n Distal symmetrical neuropathy (chronic sensory and autonomic polyneuropathy) n Acute painful neuropathy n Focal and multifocal n Mononeuropathies (diabetic amyotrophy)

Symptoms in distal symmetrical neuropathy Asymptomatic in some n Numbness n Altered sensation n paraesthesiae n allodynia n n Pain
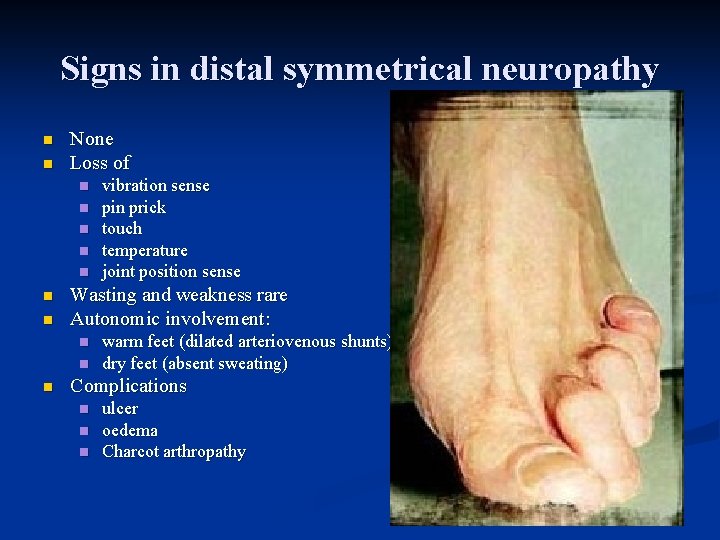
Signs in distal symmetrical neuropathy n n None Loss of n n n n Wasting and weakness rare Autonomic involvement: n n n vibration sense pin prick touch temperature joint position sense warm feet (dilated arteriovenous shunts) dry feet (absent sweating) Complications n n n ulcer oedema Charcot arthropathy
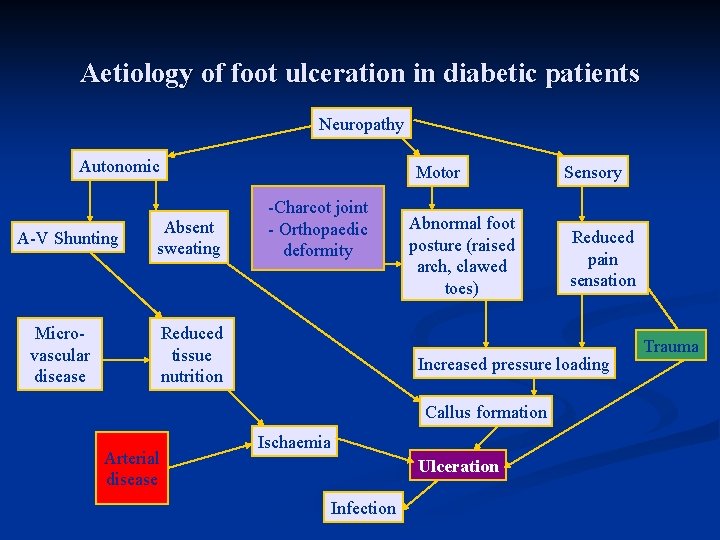
Aetiology of foot ulceration in diabetic patients Neuropathy Autonomic A-V Shunting Motor Absent sweating Microvascular disease -Charcot joint - Orthopaedic deformity Reduced tissue nutrition Abnormal foot posture (raised arch, clawed toes) Reduced pain sensation Increased pressure loading Callus formation Arterial disease Sensory Ischaemia Ulceration Infection Trauma
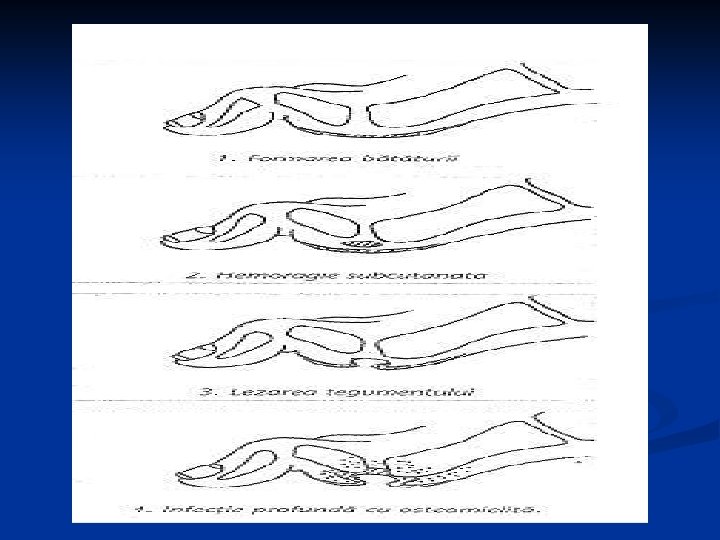

Charcot arthropathy severe neuropathy n long-standing diabetes n initiating event = injury, causing bone fracture n gross deformity n cubic foot n acute Charcot foot n
- Slides: 24