Diabetes and CV outcome its not only about

Diabetes and CV outcome, it’s not only about Hb. A 1 c 林口長庚心臟內科 吳家棟
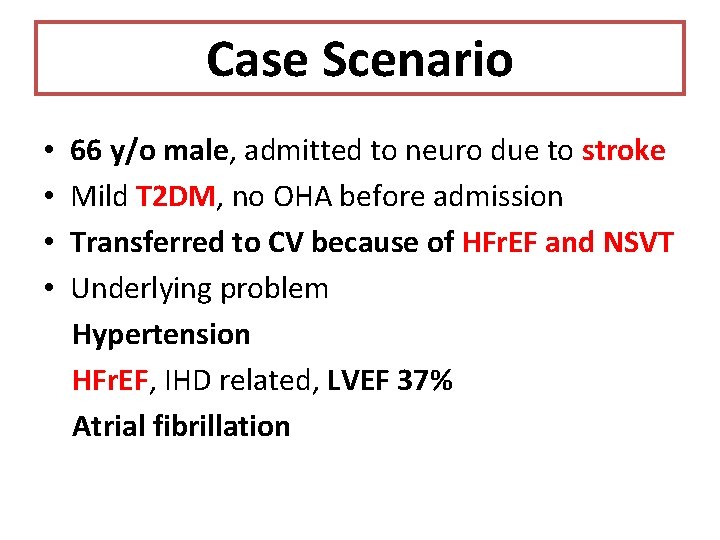
Case Scenario • • 66 y/o male, admitted to neuro due to stroke Mild T 2 DM, no OHA before admission Transferred to CV because of HFr. EF and NSVT Underlying problem Hypertension HFr. EF, IHD related, LVEF 37% Atrial fibrillation

Case Scenario • 2017/05/24 BNP ALT Creatinine Estimated GFR Hb. A 1 c 1176. 3 pg/m. L 15 U/L 1. 04 mg/d. L >60 m. L/min 6. 4 %

Case Scenario • Medication from neurology Rivaroxaban 10 mg/tab Metformin 500 mg/tab Atorvastatin 40 mg/tab(Lipitor, Pfizer) LORazepam 1 mg/tab Sennoside A+B calcium 12 mg/tab Amiodarone HCl 200 mg/tab 1 PC QD 1 PC BID 1 PC QD 0. 5 PC HS 2 PC HS 1 PC BID
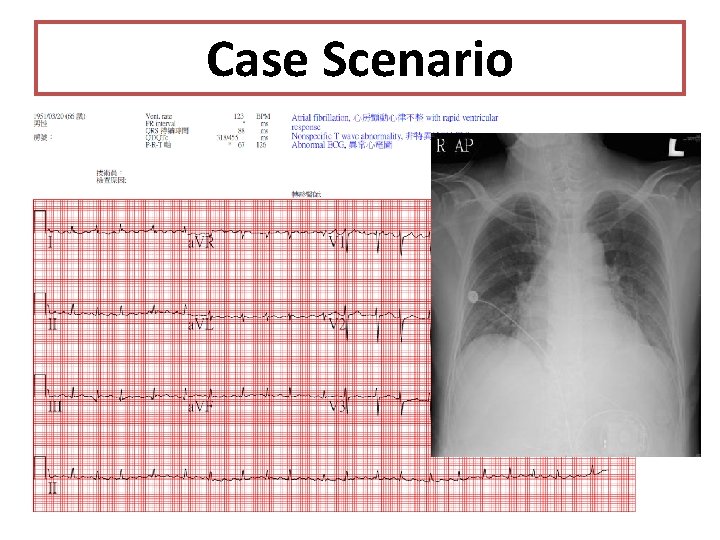
Case Scenario
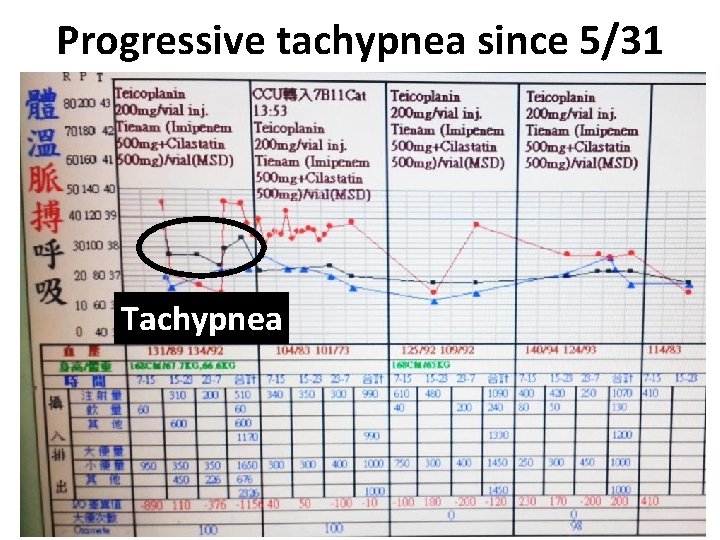
Progressive tachypnea since 5/31 Tachypnea
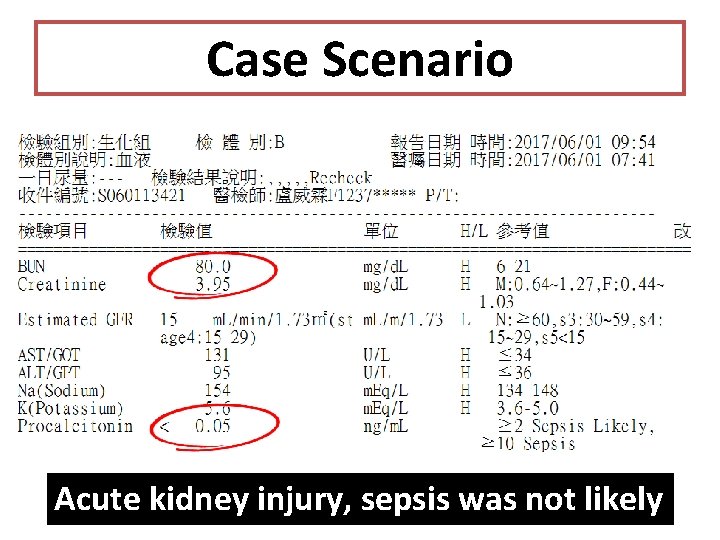
Case Scenario Acute kidney injury, sepsis was not likely
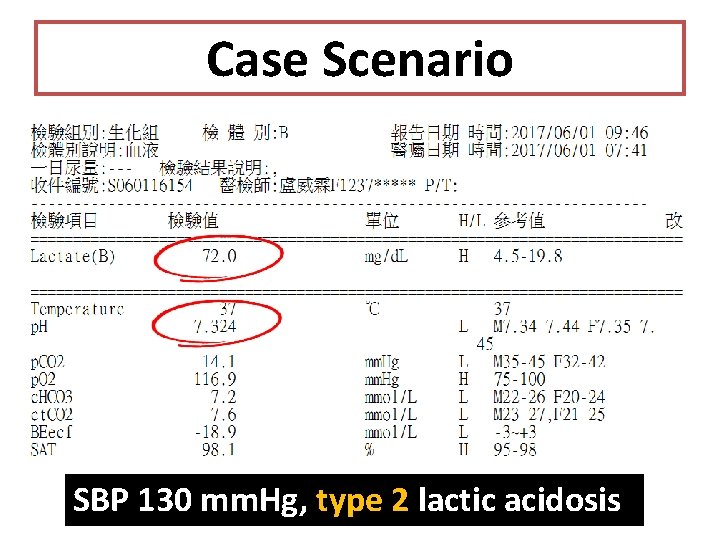
Case Scenario SBP 130 mm. Hg, type 2 lactic acidosis
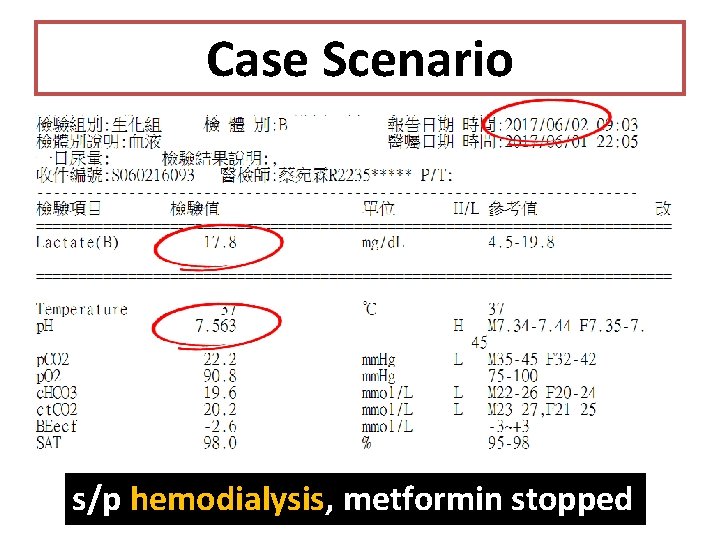
Case Scenario s/p hemodialysis, metformin stopped
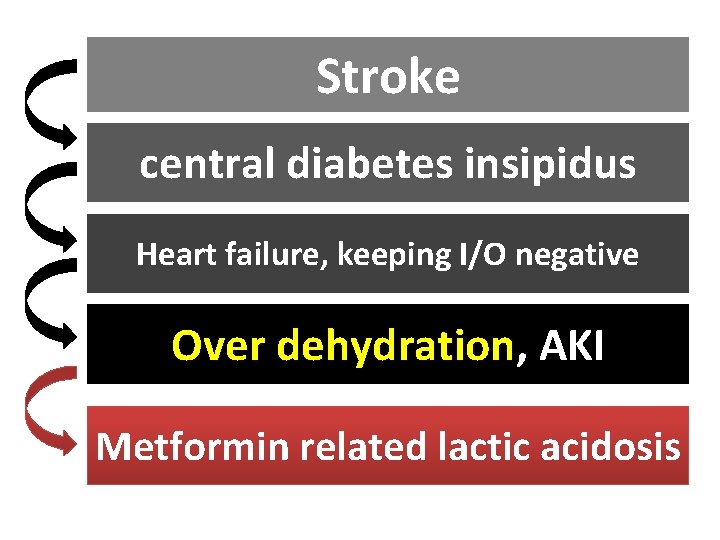
Stroke central diabetes insipidus Heart failure, keeping I/O negative Over dehydration, AKI Metformin related lactic acidosis

We fight for the best, and prepare for the worst !
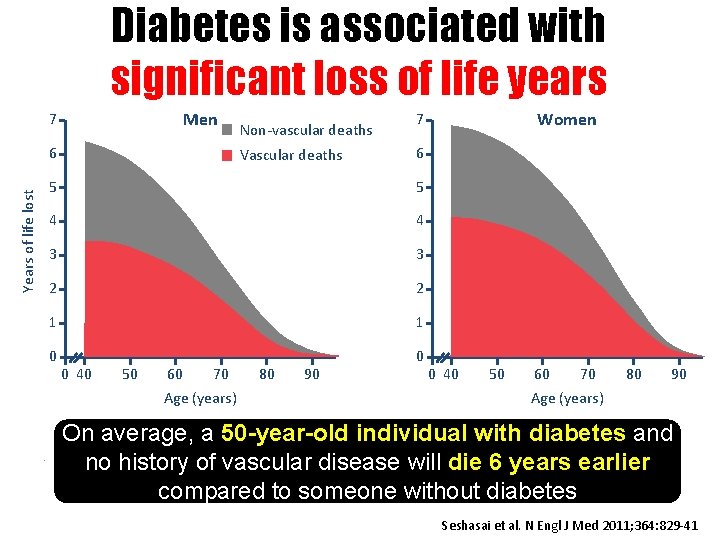
Diabetes is associated with significant loss of life years Men 7 Years of life lost 6 Vascular deaths 6 5 4 4 3 3 2 2 1 1 0 0 0 40 50 60 70 80 90 Women 7 5 Age (years) . Non-vascular deaths 0 40 50 60 70 80 90 Age (years) On average, a 50 -year-old individual with diabetes and no history of vascular disease will die 6 years earlier compared to someone without diabetes Seshasai et al. N Engl J Med 2011; 364: 829 -41
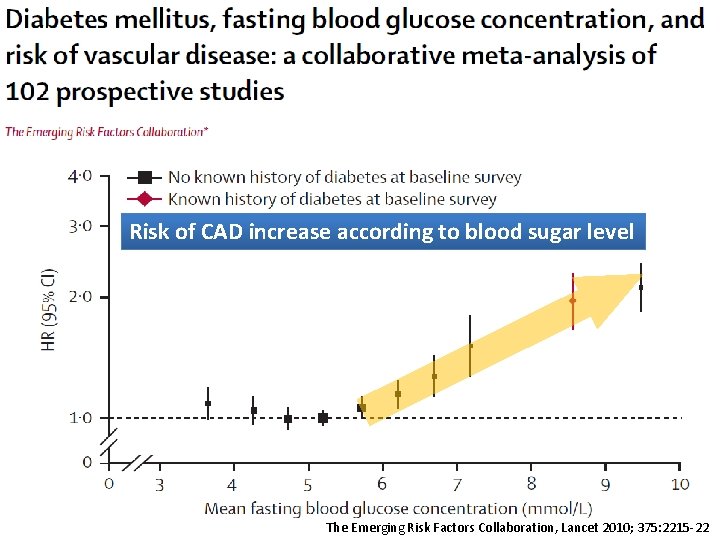
Risk of CAD increase according to blood sugar level The Emerging Risk Factors Collaboration, Lancet 2010; 375: 2215 -22
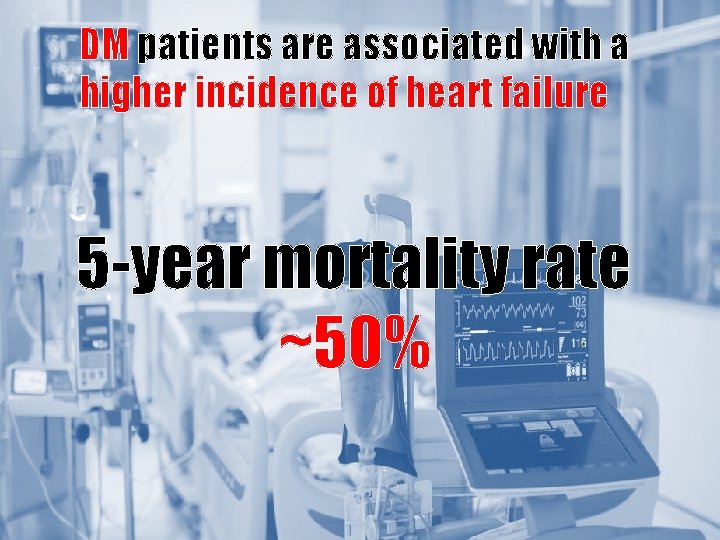
DM patients are associated with a higher incidence of heart failure Patient with diabetes have a 2 -5 fold increased risk of CHF. 5 -year mortality rate ~50% Age (years) Hb. A 1 c (%) 1, Nichols GA, Diabetes Care 2004; 27 (8); 1879 -84 2. Richard EG, Lancet 2015; 385; 2107 -17

CV outcome trial on T 1 DM patients

From 1983 to 1993 and funded by the National Institute of Diabetes and Digestive and Kidney Diseases 1, 441 volunteers, ages 13 to 39, with type 1 diabetes Intensive care (Hb. A 1 c close to normal) vs standard care DCCT N Engl J Med 1993; 329: 977– 986 EDIC N Engl J Med 2000; 342: 381– 389
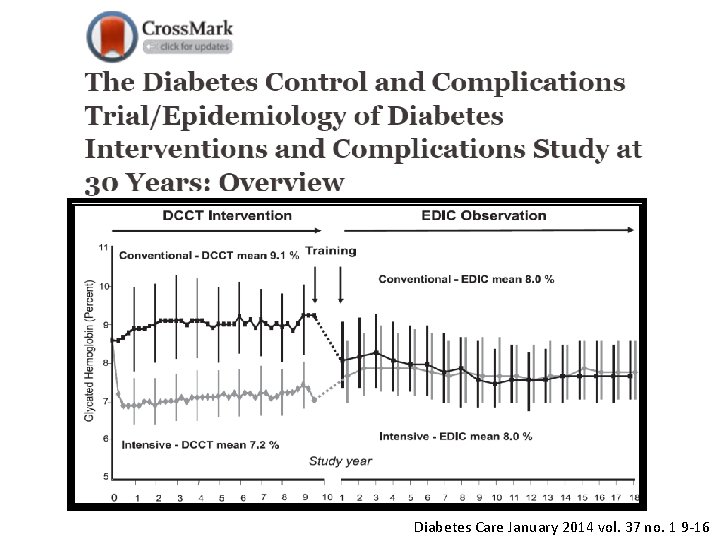
Diabetes Care January 2014 vol. 37 no. 1 9 -16
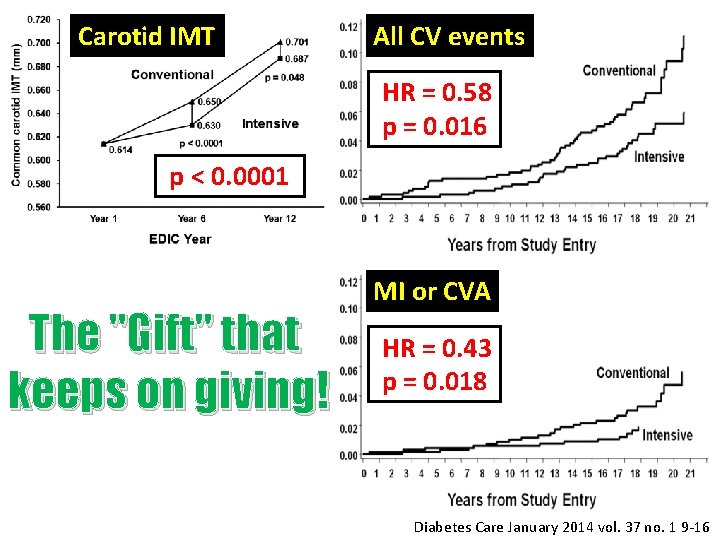
Carotid IMT All CV events HR = 0. 58 p = 0. 016 p < 0. 0001 The "Gift" that keeps on giving! MI or CVA HR = 0. 43 p = 0. 018 Diabetes Care January 2014 vol. 37 no. 1 9 -16

How about intensive sugar control on T 2 DM patients

Major CVOT on T 2 DM before 2010 2009 NEJM VADT 2008 NEJM ADVANCE 2008 NEJM ACCORD
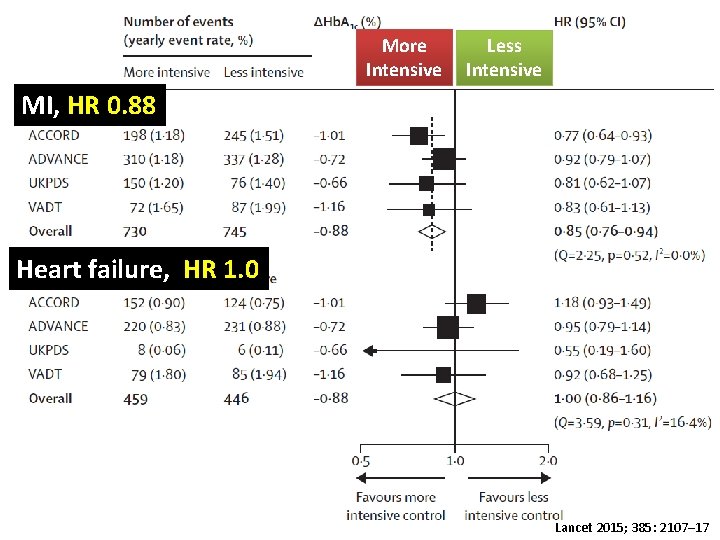
More Intensive Less Intensive MI, HR 0. 88 Heart failure, HR 1. 0 Lancet 2015; 385: 2107– 17
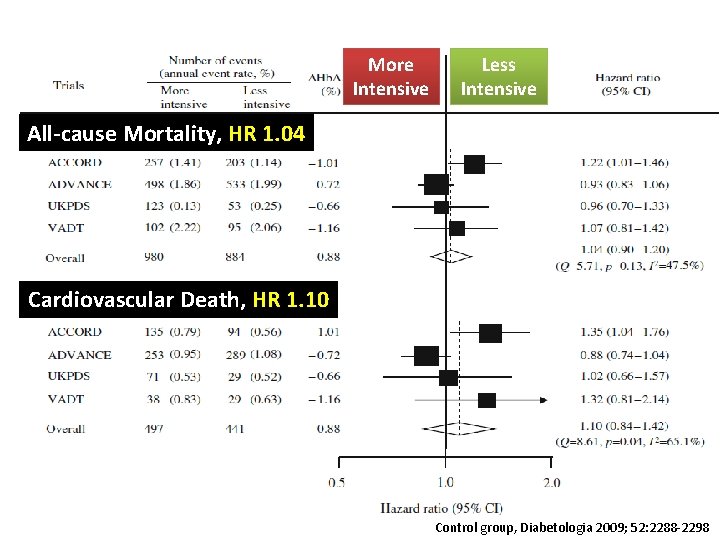
More Intensive Less Intensive All-cause Mortality, HR 1. 04 Cardiovascular Death, HR 1. 10 Control group, Diabetologia 2009; 52: 2288 -2298
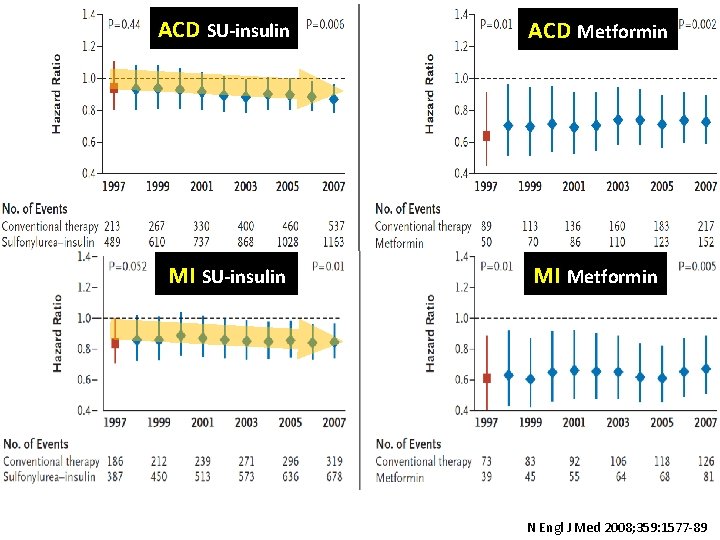
ACD SU-insulin ACD Metformin MI SU-insulin MI Metformin N Engl J Med 2008; 359: 1577 -89
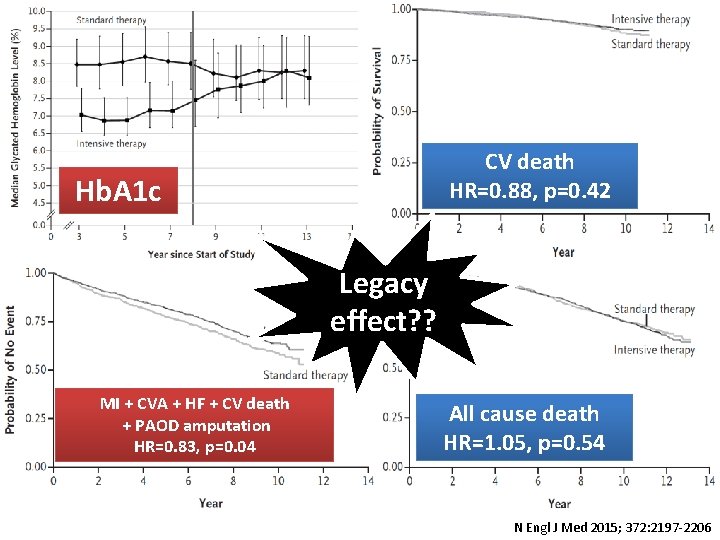
CV death HR=0. 88, p=0. 42 Hb. A 1 c Legacy effect? ? MI + CVA + HF + CV death + PAOD amputation HR=0. 83, p=0. 04 All cause death HR=1. 05, p=0. 54 N Engl J Med 2015; 372: 2197 -2206

Blood sugar control on high risk patients

Case Scenario • • 82 y/o female Type 2 DM Hypertension Hyperlipidemia AV block s/p DDDR pacemaker Heart failure, LVEF 34% to 47% Tl-201: Mixed myocardial infarction with ischemia in septal segment of LV
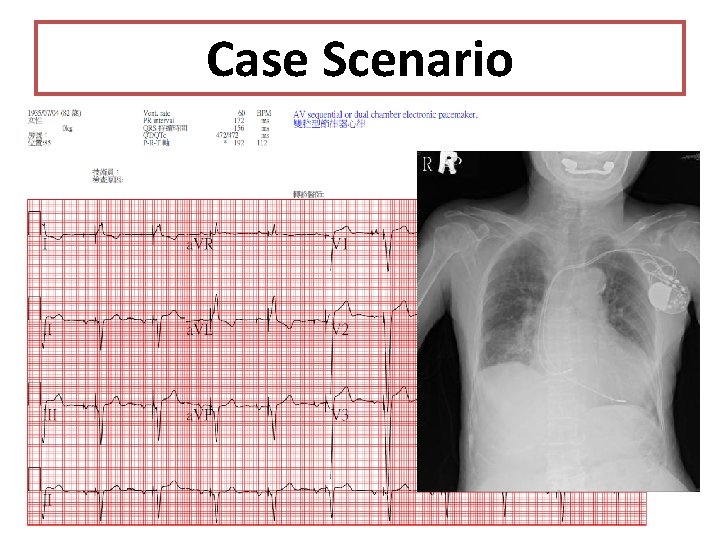
Case Scenario

Admitted due to pulmonary edema What’s best 1 st line OHA for her T 2 DM ? 1. Metformin 2. Sulfonylurea 3. DPP-4 inhibitor 4. SGLT 2 inhibitor 5. Pioglitazone

Management of DM, with concern about CV outcome and HF Thiazolidinedione (TZD)
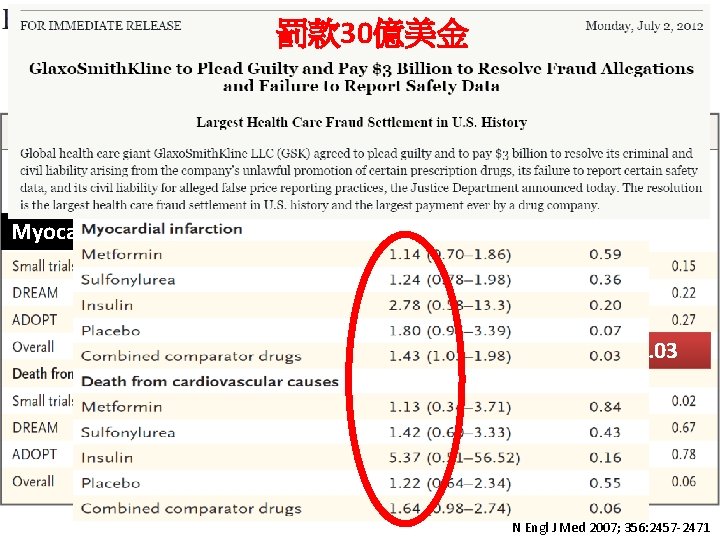
罰款 30億美金 Myocardial infarction OR 1. 43, P = 0. 03 N Engl J Med 2007; 356: 2457 -2471
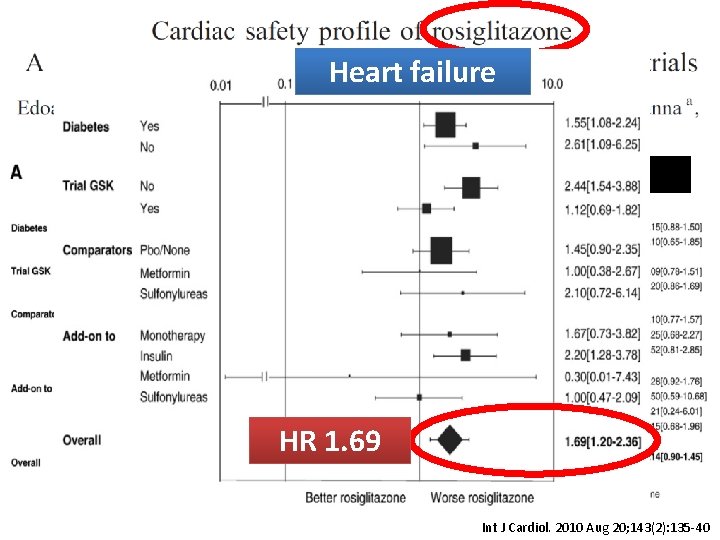
Heart failure Coronary event, HR = 1. 09 MI, HR = 1. 14 HR 1. 69 Int J Cardiol. 2010 Aug 20; 143(2): 135 -40
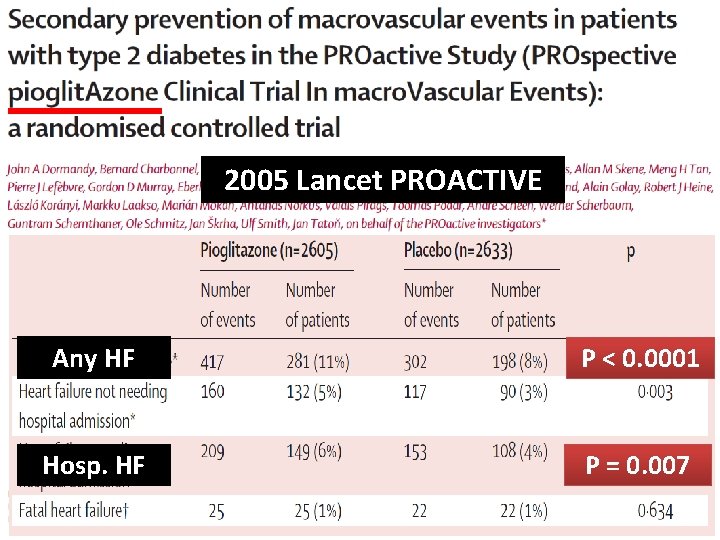
2005 Lancet PROACTIVE Any HF P < 0. 0001 HR 0. 9, p=0. 09 Hosp. HF Death from any cause, non-fatal P =amputation, 0. 007 MI, stroke, ACS, leg coronary or leg revascularisation Lancet 2005; 366: 1279– 89
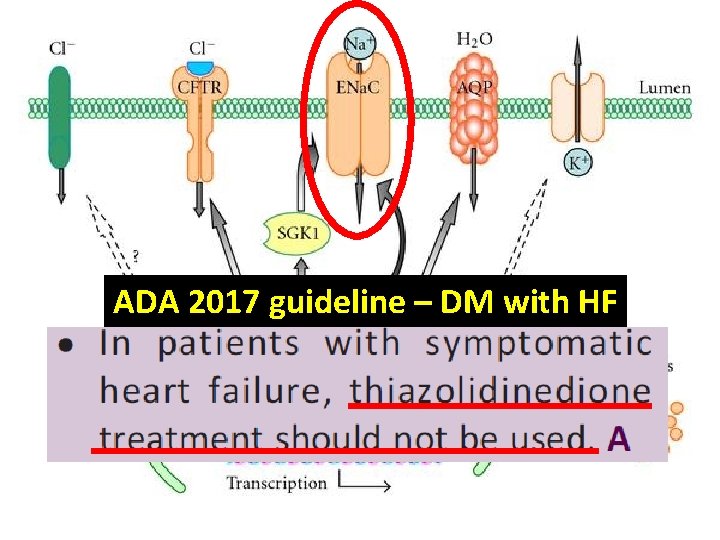
ADA 2017 guideline – DM with HF

Management of DM, with concern about CV outcome and HF Metformin and SU

ADA 2017 guideline – DM with HF HR 0. 80 Circ Heart Fail. 2013 May; 6(3): 395 -402
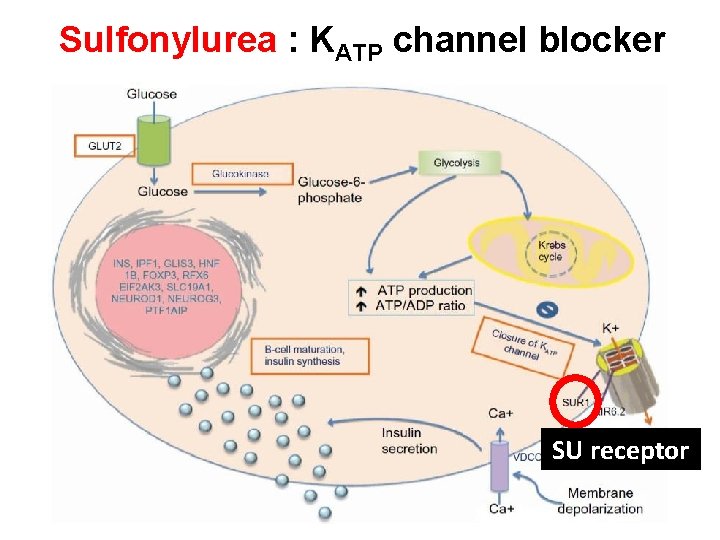
Sulfonylurea : KATP channel blocker SU receptor
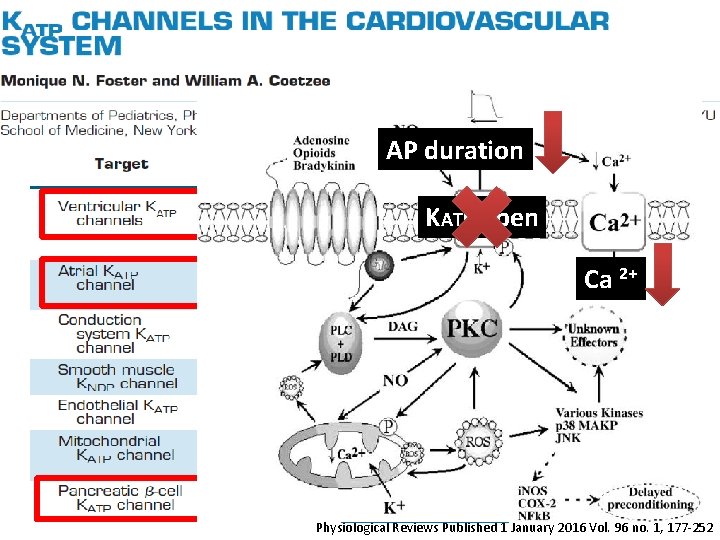
AP duration KATP open Ca 2+ Physiological Reviews Published 1 January 2016 Vol. 96 no. 1, 177 -252

SU ADA 2017 guideline – SU No comment Pioglitazone Metformin HR 1. 17 SU HR 1. 3 BMC Cardiovascular Disorders 201414: 129

Management of DM, with concern about CV outcome and HF DPP-4 inhibitors
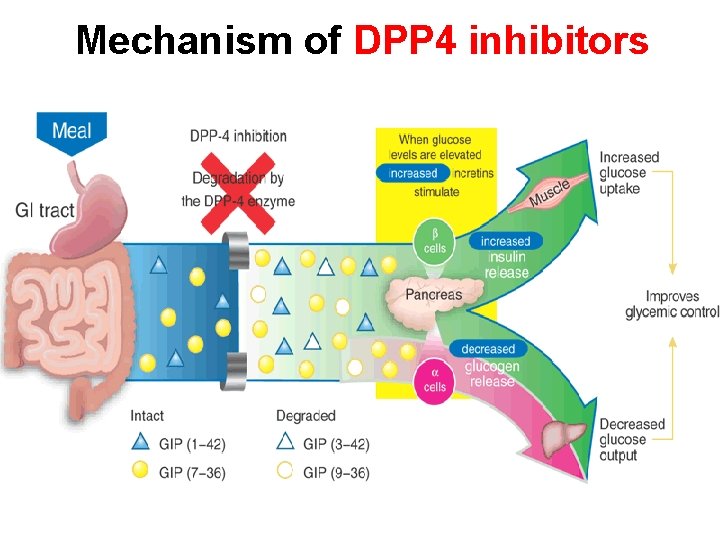
Mechanism of DPP 4 inhibitors
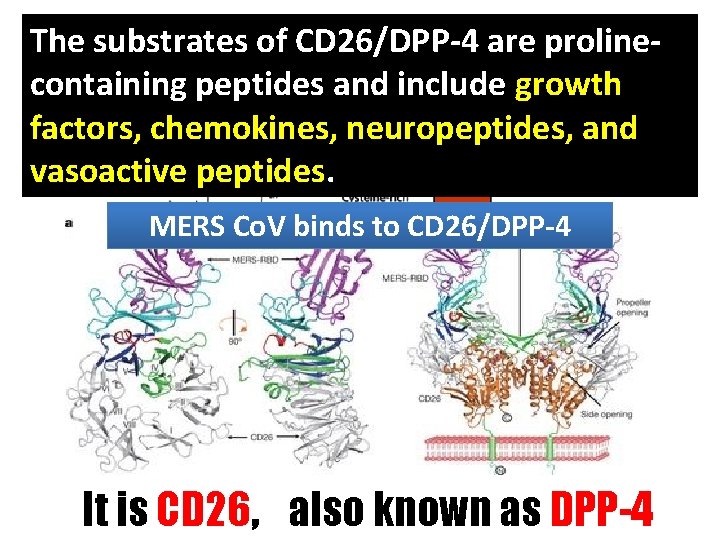
The substrates of CD 26/DPP-4 are prolinecontaining peptides and include growth factors, chemokines, neuropeptides, and vasoactive peptides. MERS Co. V binds to CD 26/DPP-4 It is CD 26, also known as DPP-4

What happened after Avandia event: FDA issued a guidance to ascertain CV safety – Dec 2008 Guidance for industry Diabetes Mellitus – Evaluating Cardiovascular Risk in New Antidiabetic Therapies to Treat Type 2 Diabetes U. S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDFR) December 2008 Clinical/Medical Key message: All development programs for T 2 DM should rule out "unacceptable increase in CV risk" FDA guidance to ascertain CV safety "Important CV events" (core major adverse cardiac events)1 should be analyzed • To rule out an 80% increase in risk at point of registration • To rule out a 30% increase in risk to remain on the market High-risk population to be included in the program • Advanced disease (e. g. recent event or long duration of diabetes) • Elderly • Renally impaired Long term data required (≥ 2 years) Prospective adjudication of CV events by an independent committee 1 Cardiac death, myocardial infarction, revascularization http: //www. fda. gov/downloads/Drugs/Guidance. Compliance. Regulatory. Information/Guidances/ucm 071627. pdf

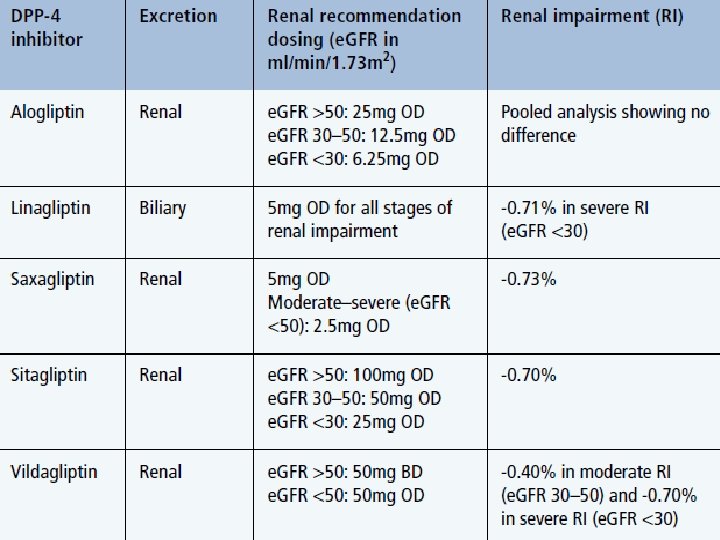
Comparison of DPP-4 i CV outcome trials Hb. A 1 c Range, Duration of Treatment (as part of usual care) % Alogliptin 6. 5– EXAMINE 1 11. 0 R Placebo SAVORTIMI 532 6. 5– 12. 0 TECOS 3– 5 6. 5– 8. 0 Saxagliptin R Placebo Sitagliptin R Placebo Primary End point CV death, Nonfatal MI, or Nonfatal stroke CV death, Nonfatal MI, Nonfatal stroke, or UA admission 1. Safety trial for high risk patients 2. Different inclusion criteria 3. HF is not primary end-point Randomization Year 1 Year 2 Year 3 Median Duration of Followup EXAMINE = Examination of Cardiovascular Outcomes: Alogliptin vs Standard of Care in Patients With Type 2 Diabetes Mellitus and Acute Coronary Syndrome; SAVOR-TIMI 53 = Saxagliptin Assessment of Vascular Outcomes Recorded in Patients With Diabetes Mellitus Trial-Thrombolysis in Myocardial Infarction 53; TECOS = Trial Evaluating Cardiovascular Outcomes With Sitagliptin. CV = cardiovascular; MI = myocardial infarction; UA = unstable angina 1. White WB et al. N Engl J Med. 2013; 369: 1327– 1335. 2. Scirica BM et al. N Engl J Med 2013; 369: 1317– 1326. 3. Green JB et al. Am Heart J. 2013; 166: 983– 989. e 7. 4. Bethel MA et al. Diabetes Obes Metab. 2015; 10. 1111/dom. 12441. 5. Green JB et al. N Engl J Med 2015; 373: 232 -242 Jul , 16 2015
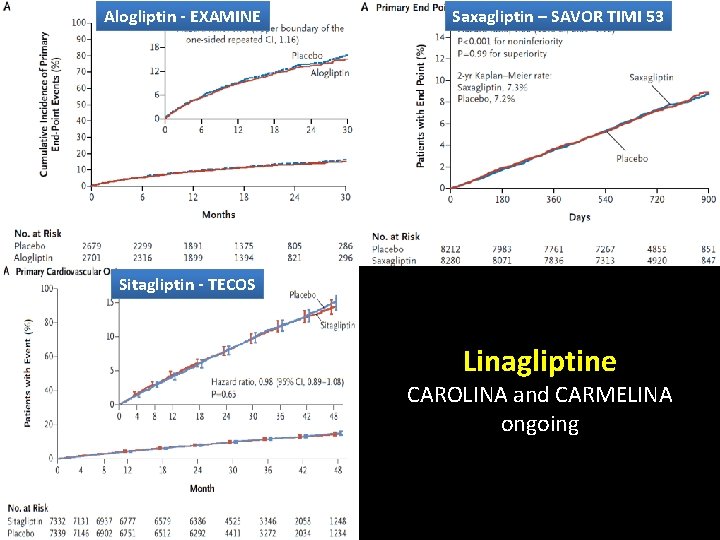
Alogliptin - EXAMINE Saxagliptin – SAVOR TIMI 53 Comparison of DPP-4 i CV outcome trials Hb. A 1 c Range, Duration of Treatment (as part of usual care) % Alogliptin 6. 5– EXAMINE 1 11. 0 R Placebo SAVORTIMI 532 6. 5– 12. 0 TECOS 3– 5 6. 5– 8. 0 Saxagliptin R Sitagliptin - TECOS Placebo Sitagliptin R Randomization Primary End point CV death, Nonfatal MI, or Nonfatal stroke CV death, Nonfatal MI, Nonfatal stroke, or UA admission Linagliptine Placebo CAROLINA CARMELINA Year and 3 Median Duration of Followongoing up Year 1 Year 2 EXAMINE = Examination of Cardiovascular Outcomes: Alogliptin vs Standard of Care in Patients With Type 2 Diabetes Mellitus and Acute Coronary Syndrome; SAVOR-TIMI 53 = Saxagliptin Assessment of Vascular Outcomes Recorded in Patients With Diabetes Mellitus Trial-Thrombolysis in Myocardial Infarction 53; TECOS = Trial Evaluating Cardiovascular Outcomes With Sitagliptin. CV = cardiovascular; MI = myocardial infarction; UA = unstable angina 1. White WB et al. N Engl J Med. 2013; 369: 1327– 1335. 2. Scirica BM et al. N Engl J Med 2013; 369: 1317– 1326. 3. Green JB et al. Am Heart J. 2013; 166: 983– 989. e 7. 4. Bethel MA et al. Diabetes Obes Metab. 2015; 10. 1111/dom. 12441. 5. Green JB et al. N Engl J Med 2015; 373: 232 -242 Jul , 16 2015
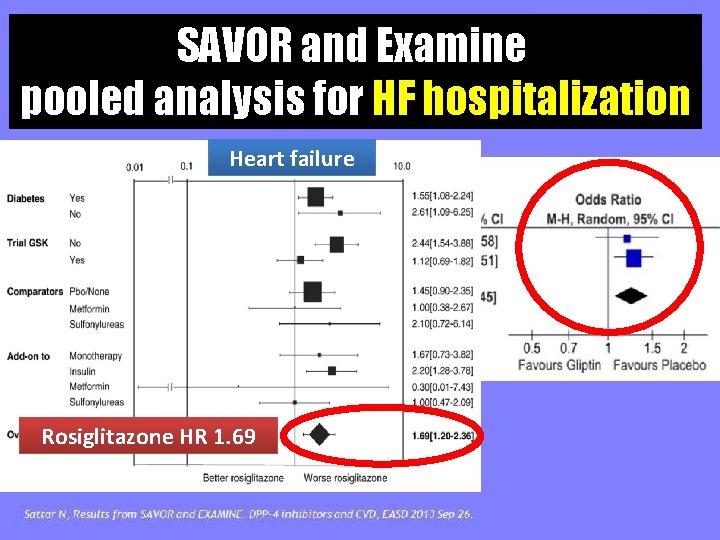
SAVOR and Examine pooled analysis for HF hospitalization Heart failure Rosiglitazone HR 1. 69

TECOS : Heart failure hospitalization • Hospitalization for heart failure ITT Population Sitagliptin N=7, 332 Placebo N=7, 339 Hospitalization for heart failure, n (%); rate per 100 patient-years 228 (3. 1); 1. 07 229 (3. 1); 1. 09 HR (95% CI) P-value 1. 00 (0. 83, 1. 20) 0. 98 • Composite outcome of hospitalization for heart failure or CV death ITT Population Sitagliptin N=7, 332 Placebo N=7, 339 Composite outcome of hospitalization for heart failure or CV death, n (%); rate per 100 patient-years 538 (7. 3); 2. 54 525 (7. 2); 2. 50 HR (95% CI) P-value 1. 02 (0. 90– 1. 15) 0. 74 N Engl J Med 2015; 373: 232 -242
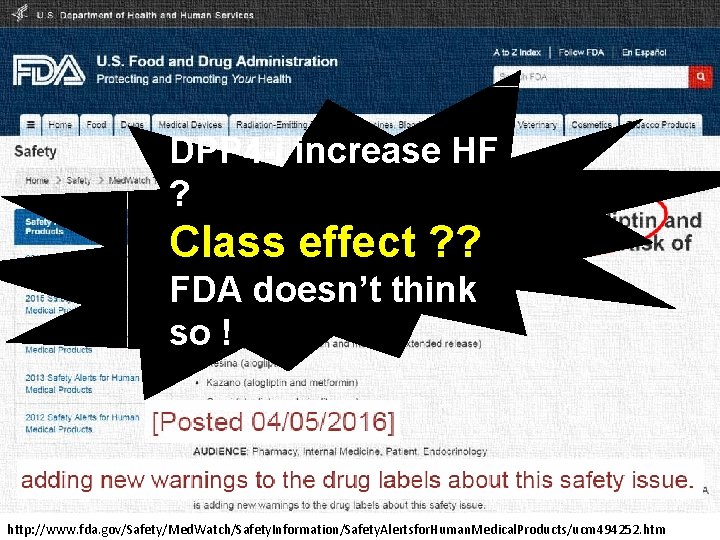
DPP 4 -i increase HF ? Class effect ? ? FDA doesn’t think so ! http: //www. fda. gov/Safety/Med. Watch/Safety. Information/Safety. Alertsfor. Human. Medical. Products/ucm 494252. htm
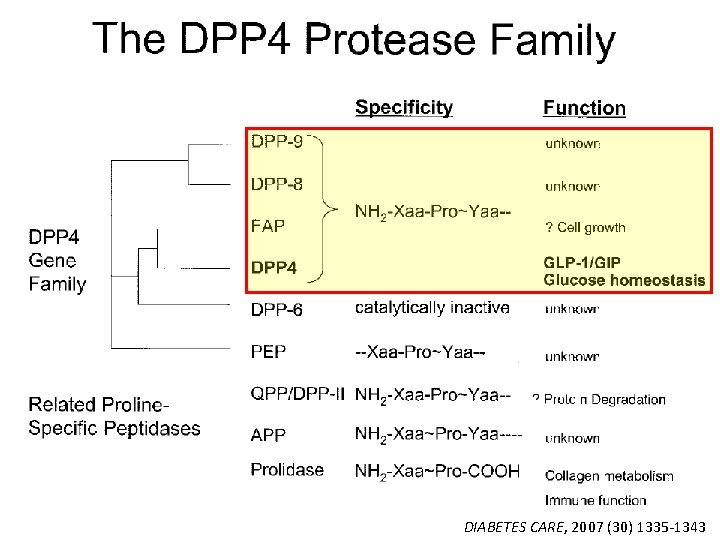
DIABETES CARE, 2007 (30) 1335 -1343
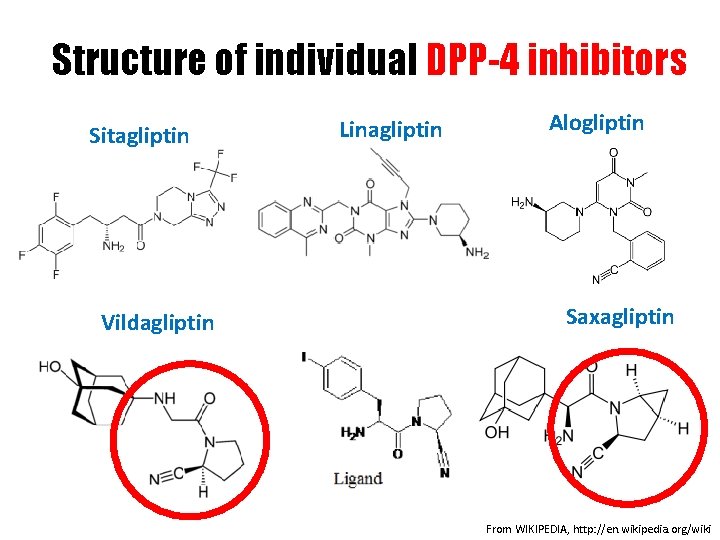
Structure of individual DPP-4 inhibitors Sitagliptin Vildagliptin Linagliptin Alogliptin Saxagliptin From WIKIPEDIA, http: //en. wikipedia. org/wiki
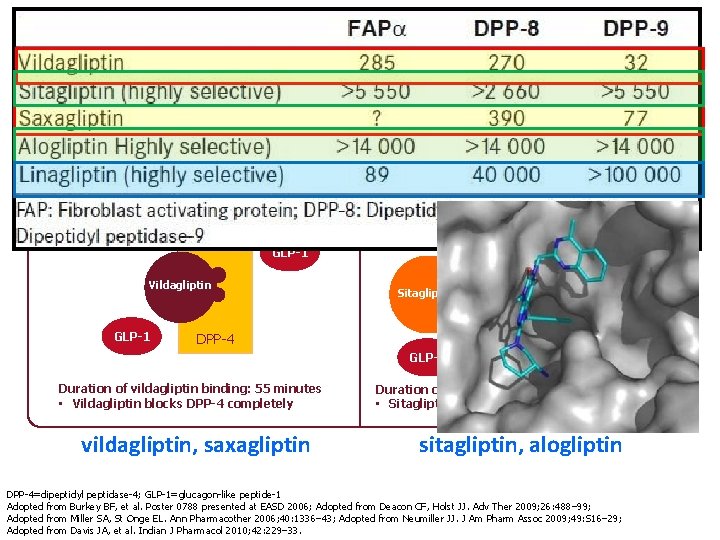
Substrate-like binding vs Competitive binding Substrate-like enzyme blocker GLP-1 Competitive enzyme inhibitor GLP-1 Vildagliptin GLP-1 Sitagliptin DPP-4 GLP-1 Duration of vildagliptin binding: 55 minutes • Vildagliptin blocks DPP-4 completely vildagliptin, saxagliptin Duration of sitagliptin interaction: <30 seconds • Sitagliptin slows the rate of DPP-4 inactivation sitagliptin, alogliptin DPP-4=dipeptidyl peptidase-4; GLP-1=glucagon-like peptide-1 Adopted from Burkey BF, et al. Poster 0788 presented at EASD 2006; Adopted from Deacon CF, Holst JJ. Adv Ther 2009; 26: 488– 99; Adopted from Miller SA, St Onge EL. Ann Pharmacother 2006; 40: 1336– 43; Adopted from Neumiller JJ. J Am Pharm Assoc 2009; 49: S 16– 29; Adopted from Davis JA, et al. Indian J Pharmacol 2010; 42: 229– 33.
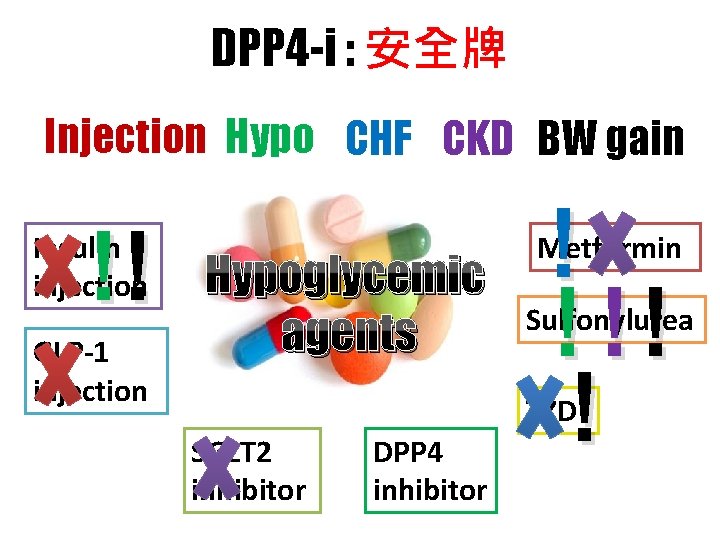
DPP 4 -i : 安全牌 Injection Hypo CHF CKD BW gain !! Insulin injection GLP-1 injection Hypoglycemic agents ! ! !! ! Metformin Sulfonylurea TZD SGLT 2 inhibitor DPP 4 inhibitor

Management of DM, with concern about CV outcome and HF SGLT-2 inhibitors
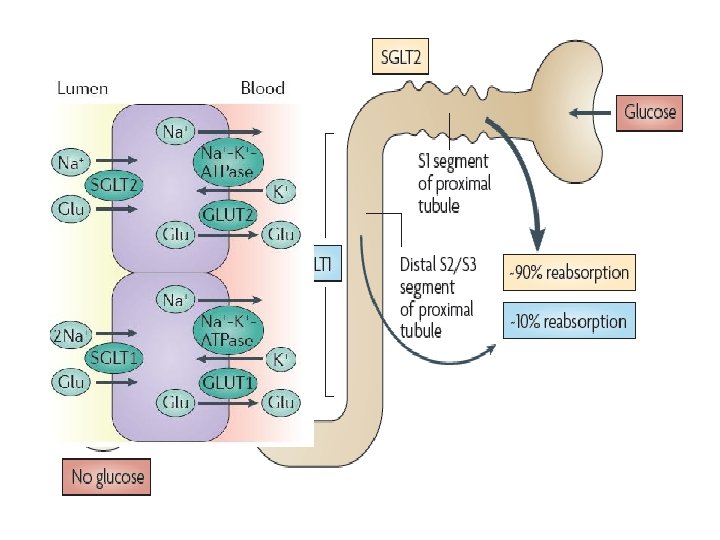

Phlorizin is a naturally occurring flavonoid produced in some plants. It is found primarily in Malus (apple) species. In 1886, Von Mering observed that dogs given doses of phlorizin above 1. 0 g developed glucosuria. Joseph von Mering (1849– 1908) en. wikipedia. org

排糖, 減重, 降血壓 三個願望 一次實現 Glycaemic control Blood pressure reduction Weight loss FORXIGA: • Acts independently of insulin mechanisms to reduce Hb. A 1 c via the kidney • Urinary excretion of ~70 g glucose/day with FORXIGA corresponds to loss of 280 Kcal/day*1 (1 g glucose = ~4 Kcal) • Works regardless of β-cell function • 1 lb of body fat equates to ~3500 calories 2 • Has a low propensity for hypoglycaemia • FORXIGA can result in loss of 1 lb of body fat in under 2 weeks • 1. FORXIGA®. Summary of product characteristics, 2014; 2. Calories per hour. com. Available at: http: //www. caloriesperhour. com/tutorial_pound. php. Last accessed August 2014; 3. Bailey CJ, et al. Lancet 2010; 375: 2223– 33. . • FORXIGA increases diuresis and is associated with significant reductions in systolic blood pressure

2015 NEJM EMPA-REG N Engl J Med 2015; 373: 2117 -2128
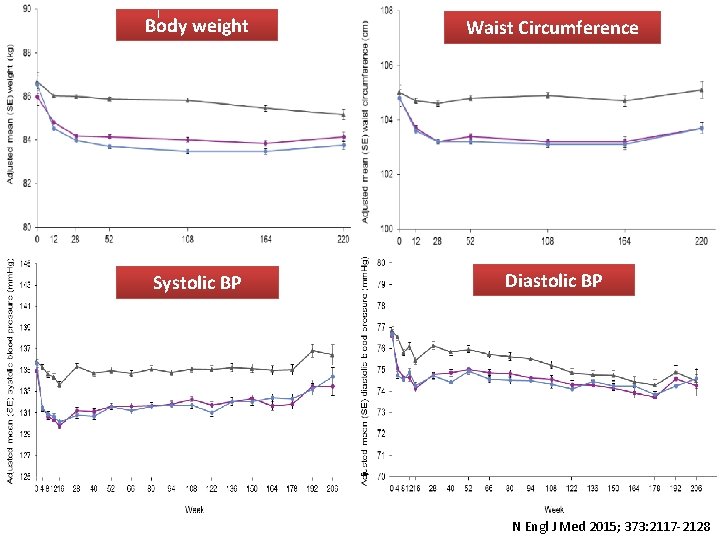
Body weight Waist Change of Hb. A 1 c comparing to. Circumference placebo -0. 54 % in 10 mg group -0. 60% in 25 mg group Adjusted mean (SE) Hb. A 1 c (%) 9. 0 8. 5 Placebo 8. 0 Empagliflozin 10 mg Empagliflozin 25 mg 7. 5 Diastolic BP Systolic BP 7. 0 6. 5 6. 0 0 12 28 40 52 66 80 94 108 122 136 150 164 178 192 206 Week Placebo Empagliflozin 10 mg 2294 2272 2296 2272 Empagliflozin 25 mg 2296 2280 2188 2133 2113 2063 2218 2150 2155 2108 2212 2150 2115 2008 1967 2072 2058 1741 1456 1241 1109 962 705 420 151 1805 1520 1297 1164 1006 749 488 170 2080 2044 1842 1540 1327 1190 1043 795 498 195 N Engl J Med 2015; 373: 2117 -2128
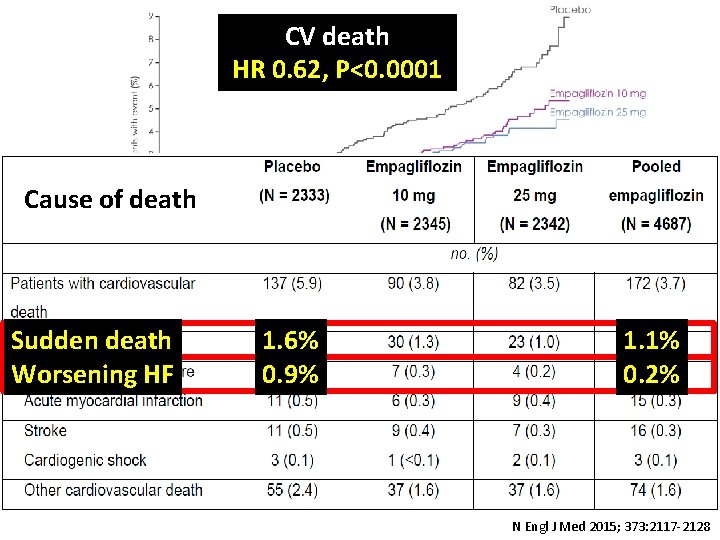
CV death 3 -point MACE Primary outcome: HR 0. 62, P<0. 0001 HR 0. 86 (95. 02% CI 0. 74, 0. 99) p=0. 0382 Cause of death Sudden death Worsening HF 1. 6% 0. 9% 1. 1% 0. 2% N Engl J Med 2015; 373: 2117 -2128

2017 NEJM CANVAS N Engl J Med 2017; 377: 644 -657
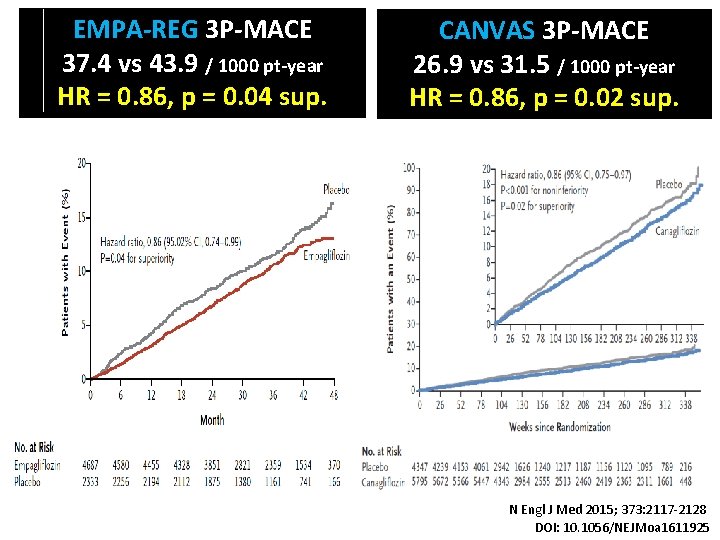
EMPA-REG 3 P-MACE 37. 4 vs 43. 9 / 1000 pt-year HR = 0. 86, p = 0. 04 sup. CANVAS 3 P-MACE 26. 9 vs 31. 5 / 1000 pt-year HR = 0. 86, p = 0. 02 sup. N Engl J Med 2015; 373: 2117 -2128 DOI: 10. 1056/NEJMoa 1611925
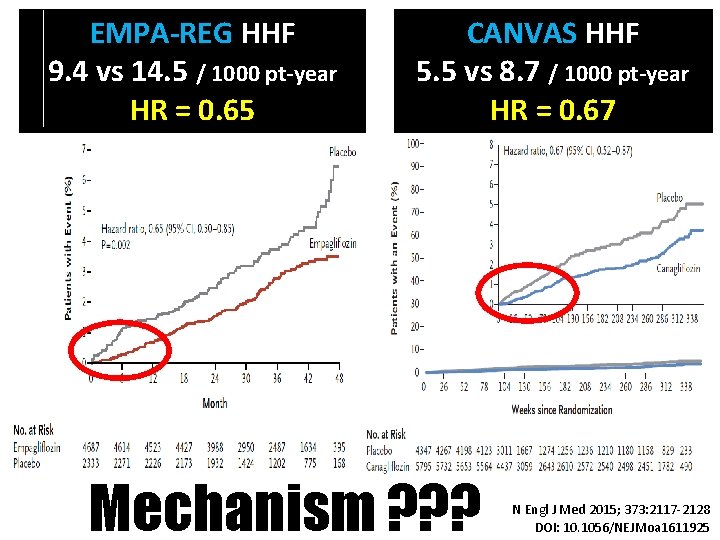
EMPA-REG HHF 9. 4 vs 14. 5 / 1000 pt-year HR = 0. 65 CANVAS HHF 5. 5 vs 8. 7 / 1000 pt-year HR = 0. 67 Mechanism ? ? ? N Engl J Med 2015; 373: 2117 -2128 DOI: 10. 1056/NEJMoa 1611925
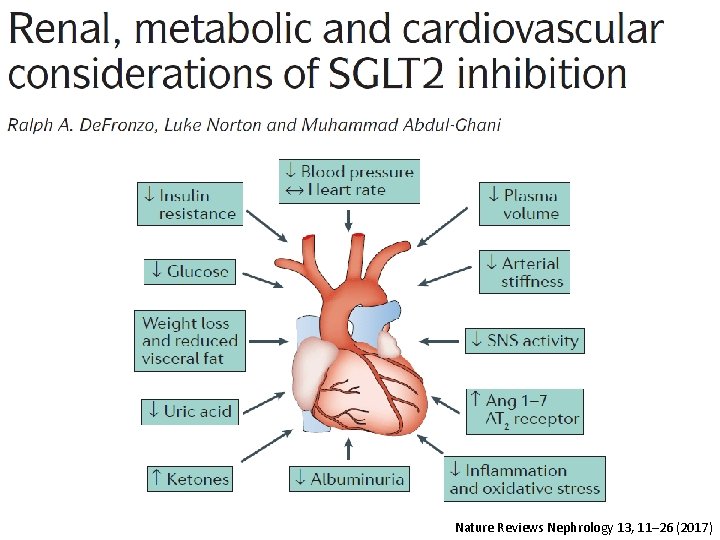
Nature Reviews Nephrology 13, 11– 26 (2017)

SGLT 2 -i and kidney Friend of foe ?
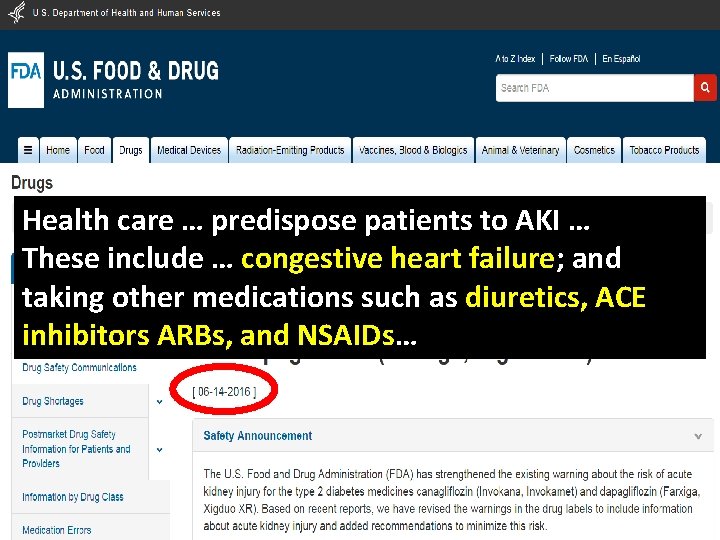
Health care … predispose patients to AKI … These include … congestive heart failure; and taking other medications such as diuretics, ACE inhibitors ARBs, and NSAIDs…

Is glycosuria per se nephrotoxic ?
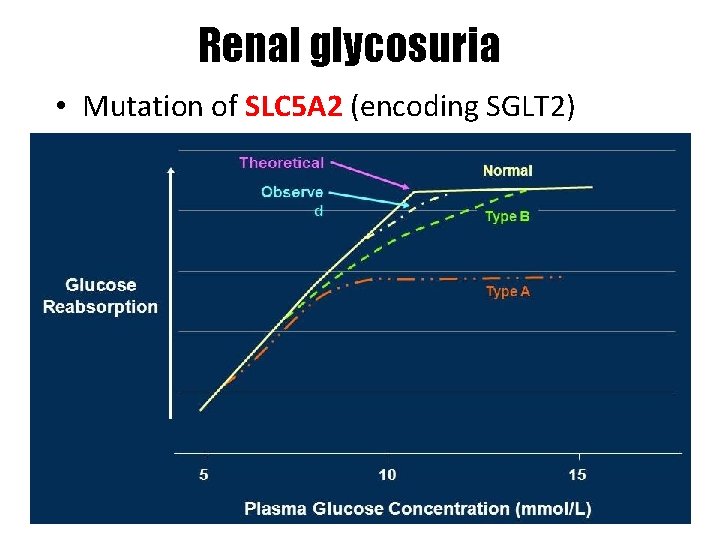
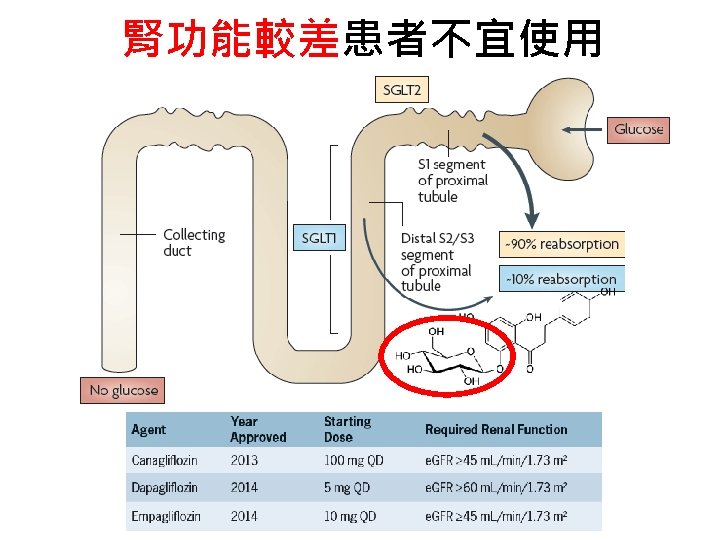
Renal glycosuria • • Mutation of SLC 5 A 2 (encoding SGLT 2) AR or AD Type A (low threshold, reduced Tm. G) Type B (low threshold, normal Tm. G) (Tm. G: tubular maximum for glucose)


This article was published on June 14, 2016, at NEJM. org. N Engl J Med 2016; 375: 323 -34
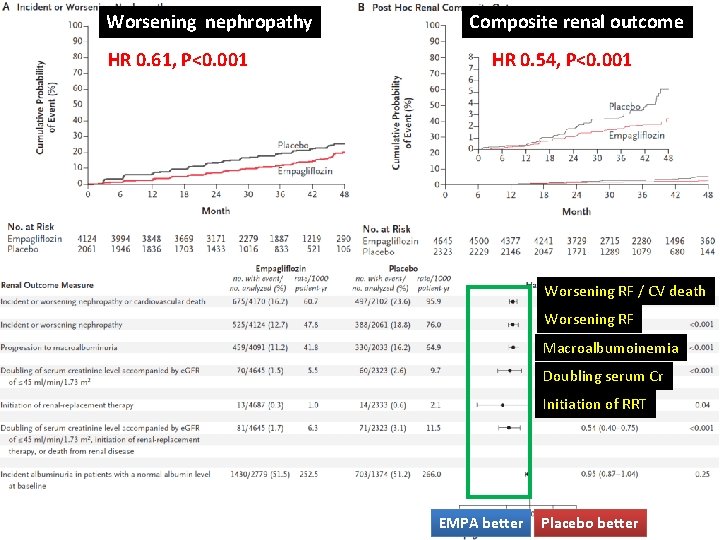
Worsening nephropathy HR 0. 61, P<0. 001 Composite renal outcome HR 0. 54, P<0. 001 Worsening RF / CV death Worsening RF Macroalbumoinemia Doubling serum Cr Initiation of RRT EMPA better NPlacebo better Engl J Med 2016; 375: 323 -34
N Engl J Med 2017; 377: 644 -657
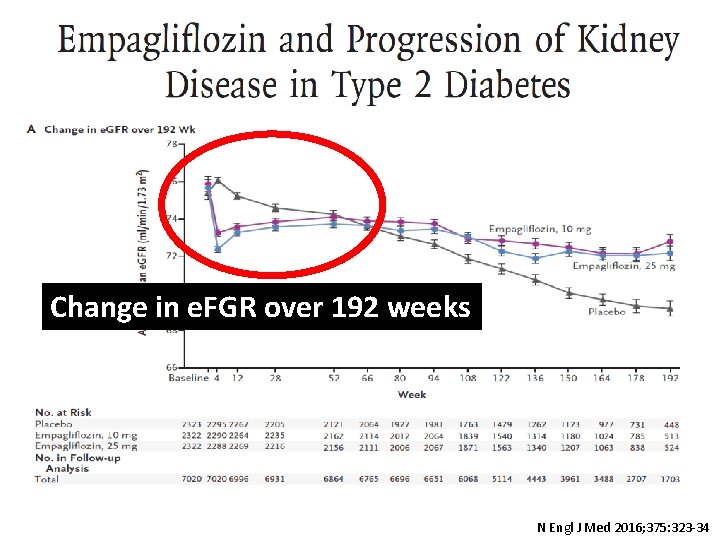
Change in e. FGR over 192 weeks N Engl J Med 2016; 375: 323 -34
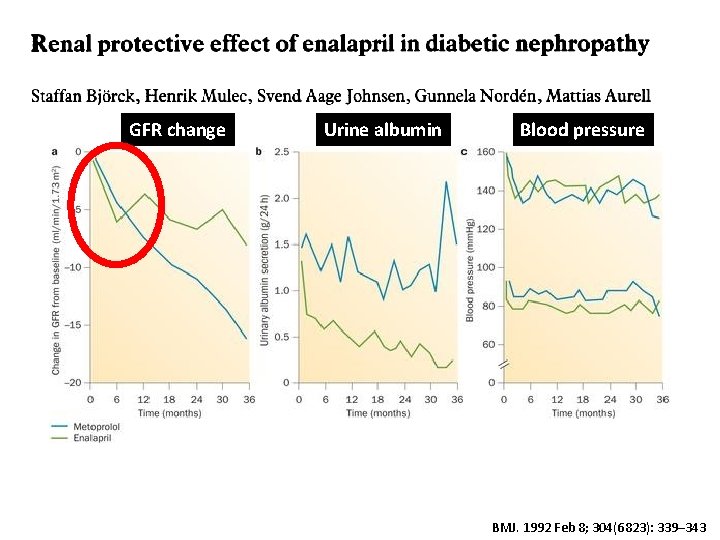
GFR change Urine albumin Blood pressure BMJ. 1992 Feb 8; 304(6823): 339– 343
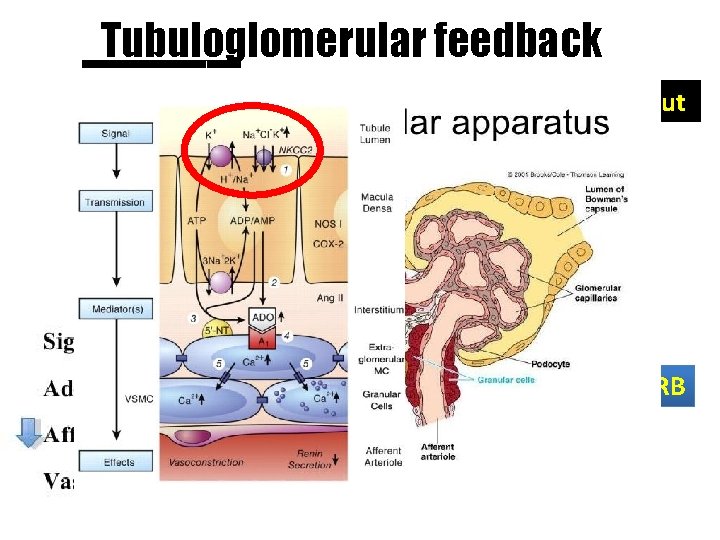
Tubuloglomerular feedback Bleeding, Dehydration Low cardiac output SGLT-2 inhibitor DM, SGLT increase ACEI / ARB

Limitations of SGLT 2 -i
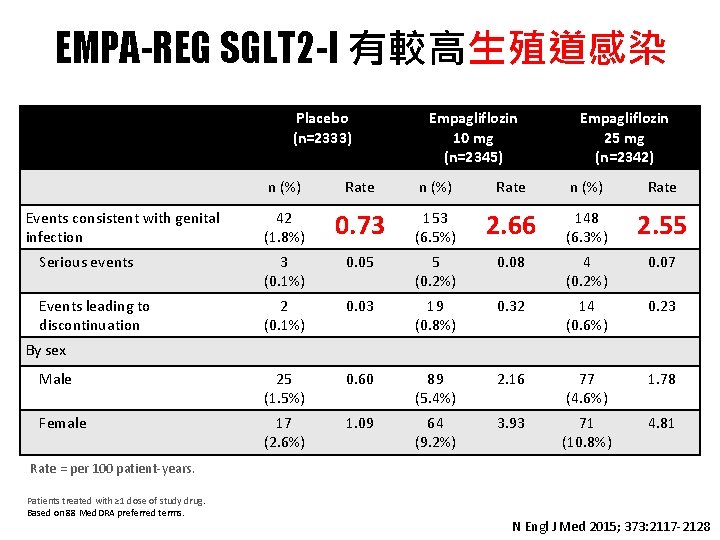
EMPA-REG SGLT 2 -I 有較高生殖道感染 Placebo (n=2333) Empagliflozin 10 mg (n=2345) Empagliflozin 25 mg (n=2342) n (%) Rate 42 (1. 8%) 0. 73 153 (6. 5%) 2. 66 148 (6. 3%) 2. 55 Serious events 3 (0. 1%) 0. 05 5 (0. 2%) 0. 08 4 (0. 2%) 0. 07 Events leading to discontinuation 2 (0. 1%) 0. 03 19 (0. 8%) 0. 32 14 (0. 6%) 0. 23 Male 25 (1. 5%) 0. 60 89 (5. 4%) 2. 16 77 (4. 6%) 1. 78 Female 17 (2. 6%) 1. 09 64 (9. 2%) 3. 93 71 (10. 8%) 4. 81 Events consistent with genital infection By sex Rate = per 100 patient-years. Patients treated with ≥ 1 dose of study drug. Based on 88 Med. DRA preferred terms. N Engl J Med 2015; 373: 2117 -2128

Canagliflozin 組有較高骨折及截肢風險 Amputation P<0. 001 All fracture P=0. 02 N Engl J Med 2017; 377: 644 -657
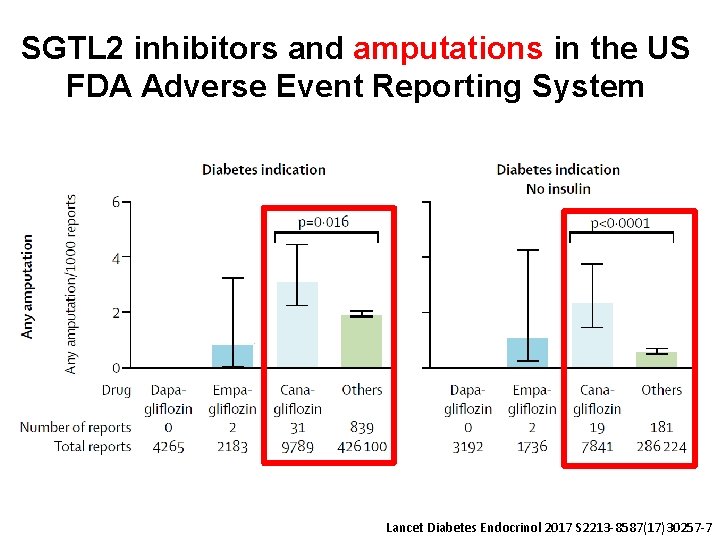
SGTL 2 inhibitors and amputations in the US FDA Adverse Event Reporting System Lancet Diabetes Endocrinol 2017 S 2213 -8587(17)30257 -7
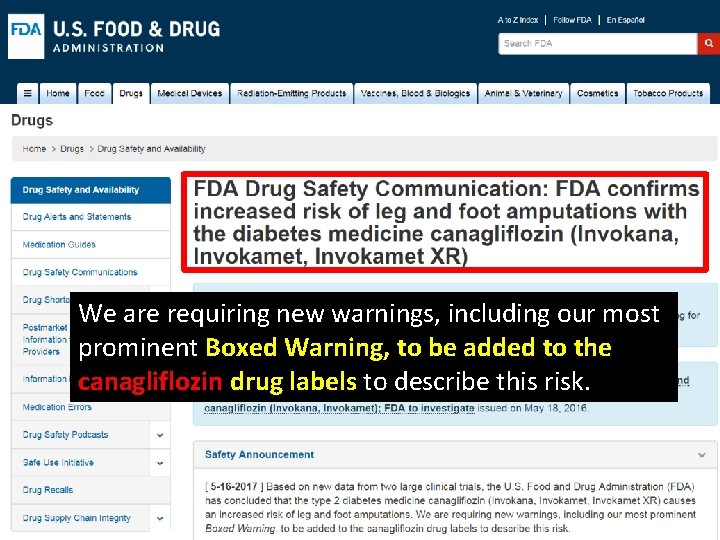
We are requiring new warnings, including our most prominent Boxed Warning, to be added to the canagliflozin drug labels to describe this risk.

ADA 2016/2017 guideline A 1 C goal for non-pregnant adults is 7% (53 mmol/mol). Lo. E A A 1 C 6. 5% for selected individual patients if this can be achieved without significant hypoglycemia or other adverse effects of treatment. Lo. E C A 1 C 8% may be appropriate for patients with a history of severe hypoglycemia, limited life expectancy, advanced microvascular or macrovascular complications, extensive comorbid conditions, or long-standing diabetes and general goal is difficult to attain. Lo. E B

Case Scenario • • 82 y/o female, type 2 DM and hypertension, AV block s/p DDDR pacemaker Heart failure, LVEF 34% to 47% Tl-201: Mixed myocardial infarction with ischemia in septal segment of LV DPP 4 inhibitors Metformin SGLT 2 inhibitors Sulfonylurea Pioglitazone (TZD)

You have to learn the rules of the game. And then you have to play better than anyone else.
- Slides: 83