Diabetes and Complications of Diabetes Lectures 15 19

Diabetes and Complications of Diabetes Lectures 15 -19 SELIN EYICI

Type 1 Diabetes Mellitus LO 1: Define type 1 diabetes mellitus, recall the epidemiology, explain the aetiology, pathophysiology, clinical presentations and explain the physiological basis of treatment
Definition & Epidemiology of T 1 DM • An autoimmune condition where there is destruction of insulin producing beta cells of the islets of Langerhans. • 10% of people with diabetes have T 1 • Slightly more M than F diagnosed • 15 x greater risk if first degree relative has diabetes • Ambiguity!
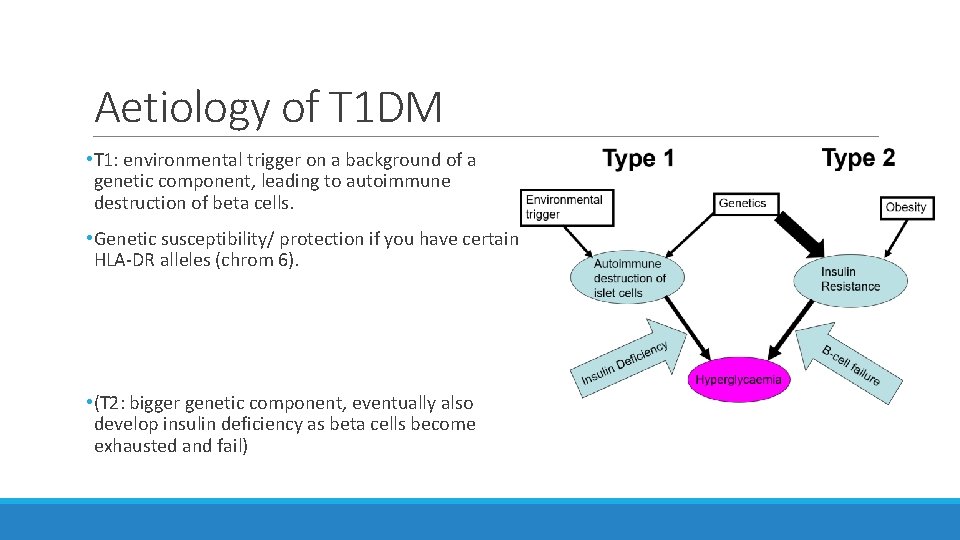
Aetiology of T 1 DM • T 1: environmental trigger on a background of a genetic component, leading to autoimmune destruction of beta cells. • Genetic susceptibility/ protection if you have certain HLA-DR alleles (chrom 6). • (T 2: bigger genetic component, eventually also develop insulin deficiency as beta cells become exhausted and fail)

Pathophysiology of T 1 DM 1) Environmental triggers and regulators along with immune dysregulation* leads to destruction of beta cells. 2) Eventually auto-antibodies are produced- further destruction 3) Patients lose first phase insulin (an indicator that a patient will go on to develop diabetes but can take years). 4) All beta cells are destroyed over time. • Auto-antibodies – marker for diagnosis, disease progress, immune modulation treatment: ØIslet Cell Antibodies (ICA)- group O human pancreas ØGlutamic Acid Decarboxylase Antibodies • A relapsing and remitting disease theory: levels of effector T cells (that cause destruction of beta cells) and regulatory T cells (that control this destruction) fluctuate but over time effector T cells increase in number*.

Pathophysiology cont. Action of insulin • Insulin has negative effects on: ØHepatic glucose output ØProtein breakdown in muscle ØGlycerol being taken out of fatty tissue into the periphery • Insulin has positive effects on: ØGlucose being taken up by muscle Insulin deficiency= a lot of glucose going out into the circulation but isn’t taken up by the tissues= HYPERGLYCAEMIA
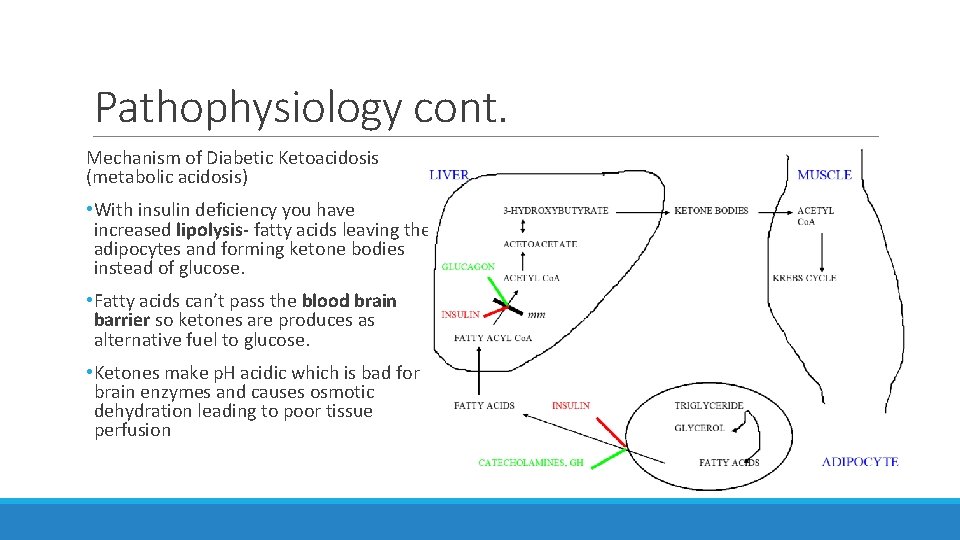
Pathophysiology cont. Mechanism of Diabetic Ketoacidosis (metabolic acidosis) • With insulin deficiency you have increased lipolysis- fatty acids leaving the adipocytes and forming ketone bodies instead of glucose. • Fatty acids can’t pass the blood brain barrier so ketones are produces as alternative fuel to glucose. • Ketones make p. H acidic which is bad for brain enzymes and causes osmotic dehydration leading to poor tissue perfusion

Clinical presentations of T 1 DM SYMPTOMS SIGNS • polyuria • dehydration • nocturia • cachexia • polydipsia • hyperventilation • blurring of vision • smell of ketones in breath • ‘thrush’ • glycosuria • weight loss • ketonuria • fatigue

Treatment of T 1 DM Aims of treatment: • Reduce early mortality • Avoid acute metabolic decompensation (diabetic ketoacidosis) • Prevent long term complications (retinopathy, nephropathy, neuropathy, macrovascular disease).

Treatment of T 1 DM cont. 1) Insulin after meals and background insulin to mimic basal insulin level. ØShort acting human insulin with meals and long acting non-C peptide insulin bound to zinc or protamine (genetic engineering to alter ADME). 2) Newer analogues- basal insulin that lasts longer. ØGiven to patients that suffer from severe hypoglycaemia. Human insulin is cheaper and just as effective. 3) Insulin pump Ø Pre-programmed basal rates and bolus for meals but doesn’t measure blood glucose so cant form a feedback loop. Could stop working. Very expensive. 4) Islet cell transplant Ø After donor dies, islet cells are harvested and injected into the liver. For patients with severe hypoglycaemia that can’t be controlled by the pump. Have to take immunosuppressants.

Treatment of T 1 DM cont. Diet: • Reduce calories as fat and refined carbohydrates • Increase calories as complex carbohydrates • Increase soluble fibre • Have regular meals and snacks Monitoring: • Capillary monitoring • Hb. A 1 c

Question 1 Which autoantibody is used as a marker for T 1 DM a) Anti-cyclic citrullinated peptide antibody (anti- CCP) b) Glutamic Acid Decarboxylase Antibodies (GADA) c) Anti-thyroid peroxidase antibody (anti-TPO) d) perinuclear anti-neutrophil cytoplasmic antibodies (p-ANCA)
Question 2 a) Explain two methods of hepatic glucose output. (2 marks) b) What happens to lipolysis in insulin deficiency and explain how it affects ketoacidosis. (2 marks)

Acute Complications of T 1 DM LO 2: Define hypoglycaemia, explain its clinical presentation, risk factors for development, and management including prevention • Hyperglycaemia • Diabetic ketoacidosis • Hypoglycaemia

Definition of Hypoglycaemia- Plasma glucose < 3. 6 mmol/ L

Clinical Presentation of Hypoglycaemia Signs and symptoms: Due to increased autonomic activation (mechanism unknown): • • • Palpitations/ tachycardia Tremor Sweating Pallor/ cold extremities Anxiety Due to impaired CNS function (glucose required for brain to function): • • • Drowsiness Confusion Altered behaviour Focal neurology (impairment in CNS that affects certain part of body) Coma

Risk factors for Developing Hypoglycaemia • Poor diabetes control- low Hb. A 1 • Why it happens: Unaccustomed exercise, missed meals/ snacks, alcohol (unaware of hypo), inappropriate insulin regime • When it happens: pre- lunch or during sleep (common and not recognised!)

Management of Hypoglycaemia (inc. Prevention) Treating hypoglycaemia: • Oral: • Glucose (solution/ tablet) • Complex carbohydrates Parenteral (if consciousness impaired): • IV dextrose • 1 mg glucagon IM

Question 3 a) Name a clinical feature of hypoglycaemia with regards to: CNS: (1 mark), ANS: (1 mark) b) What oral treatment can be used for hypoglycaemia? (1 mark) c) In what circumstances would use parenteral treatment? (1 mark) d) What parental treatment is available for hypoglycaemia and how does it work? (1 mark)

Type 2 Diabetes Mellitus LO 3: Define type 2 diabetes mellitus, recall the epidemiology, explain the aetiology, pathophysiology, clinical presentations and explain the physiological basis of treatment
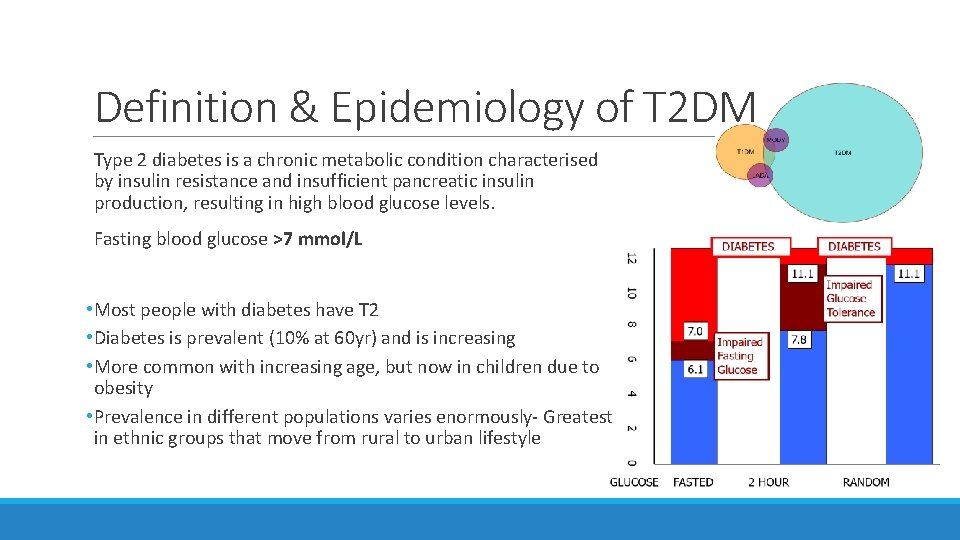
Definition & Epidemiology of T 2 DM Type 2 diabetes is a chronic metabolic condition characterised by insulin resistance and insufficient pancreatic insulin production, resulting in high blood glucose levels. Fasting blood glucose >7 mmol/L • Most people with diabetes have T 2 • Diabetes is prevalent (10% at 60 yr) and is increasing • More common with increasing age, but now in children due to obesity • Prevalence in different populations varies enormously- Greatest in ethnic groups that move from rural to urban lifestyle
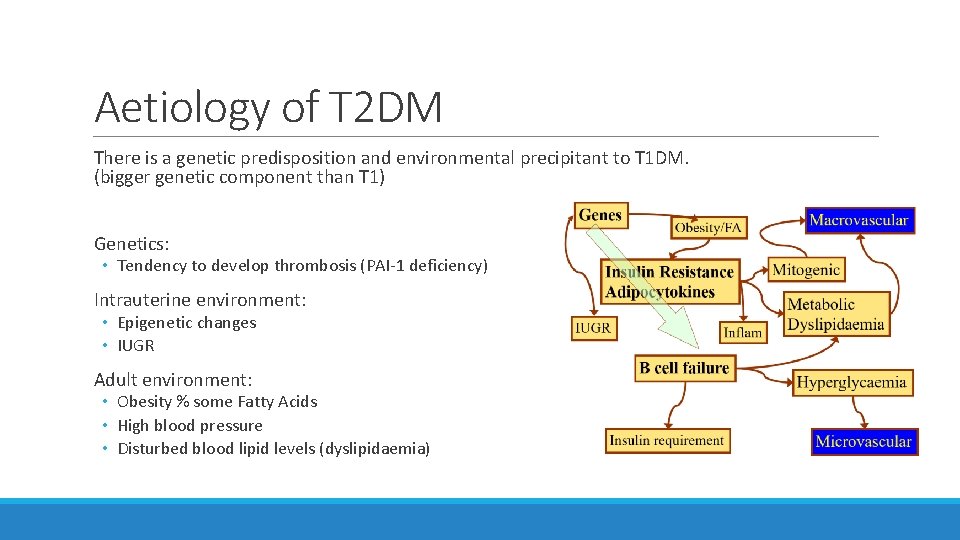
Aetiology of T 2 DM There is a genetic predisposition and environmental precipitant to T 1 DM. (bigger genetic component than T 1) Genetics: • Tendency to develop thrombosis (PAI-1 deficiency) Intrauterine environment: • Epigenetic changes • IUGR Adult environment: • Obesity % some Fatty Acids • High blood pressure • Disturbed blood lipid levels (dyslipidaemia)

Pathophysiology of T 2 DM 1) Genes causes insulin resistance, modulated by adipocytokines. 2) Other risk factors discussed (obesity etc) 3) Insulin resistance is present for a long time before glucose gets elevated and it causes: • Dyslipidaemia * • Stimulates mitogenic pathway= ↑smooth muscle hypertrophy ↑BP ^ both ↑ risk of macrovascular disease and speed up development of diabetes 4) Insulin resistance gets worse and damages beta cells gradual beta cell failure so person can’t make enough insulin to cover for their decreased insulin sensitivity hyperglycaemia 5) There is a loss of 1 st phase insulin (preformed insulin stored in granules in the beta cells). They can still make 2 nd phase insulin but it takes them longer and it is immature insulin which doesn’t work properly hyperglycaemia 6) Eventually, absolute insulin deficiency
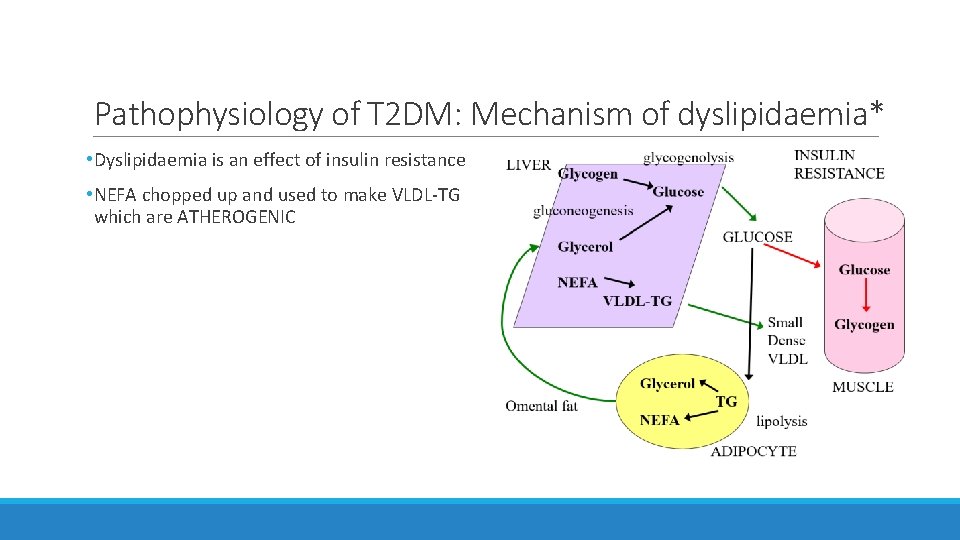
Pathophysiology of T 2 DM: Mechanism of dyslipidaemia* • Dyslipidaemia is an effect of insulin resistance • NEFA chopped up and used to make VLDL-TG which are ATHEROGENIC
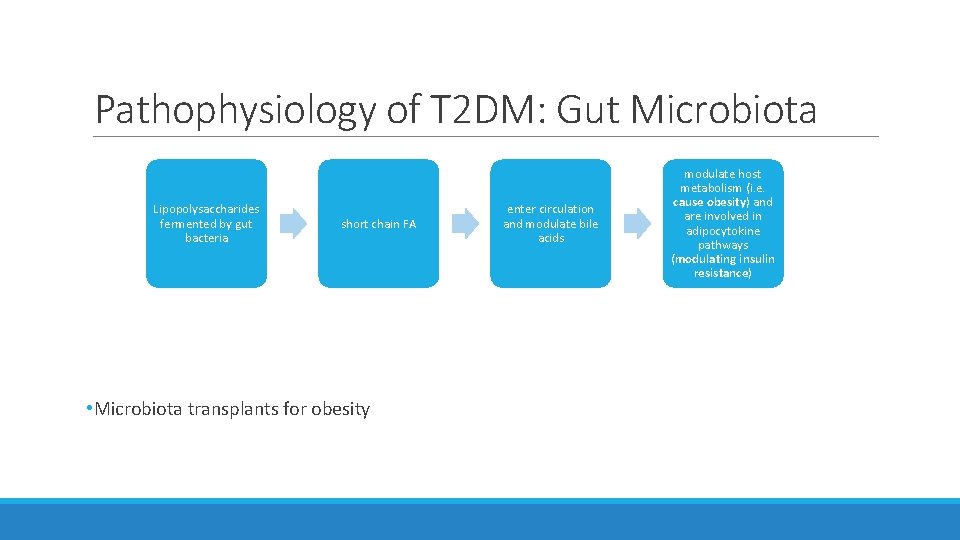
Pathophysiology of T 2 DM: Gut Microbiota Lipopolysaccharides fermented by gut bacteria short chain FA • Microbiota transplants for obesity enter circulation and modulate bile acids modulate host metabolism (i. e. cause obesity) and are involved in adipocytokine pathways (modulating insulin resistance)

Clinical Presentations of T 2 DM • Osmotic symptoms • Infections • With a complication of diabetes: • Acute: hyperosmolar coma • Chronic: ischaemic heart disease (macrovascular), retinopathy (microvascular)

Treatment of T 2 DM Aims of treatment: • • • ↓long- term complications (micro and macrovascular diseases) ↓ acute metabolic complication (unlikely in T 2 DM) ↓ symptoms Basis of management: need to manage different aspects of diabetes: BP, cholesterol, blood glucose and weight. • Education • Diet • Pharmacological treatment • ↓ kcal intake/ exercise more • ↓ refined carbs (sugar), ↑ complex carbs • ↓ fat as proportion of kcal, ↑ unsaturated fats as proportion of fat • ↑ soluble fibre (takes longer to absorb carbohydrates) • ↓ salt – for BP
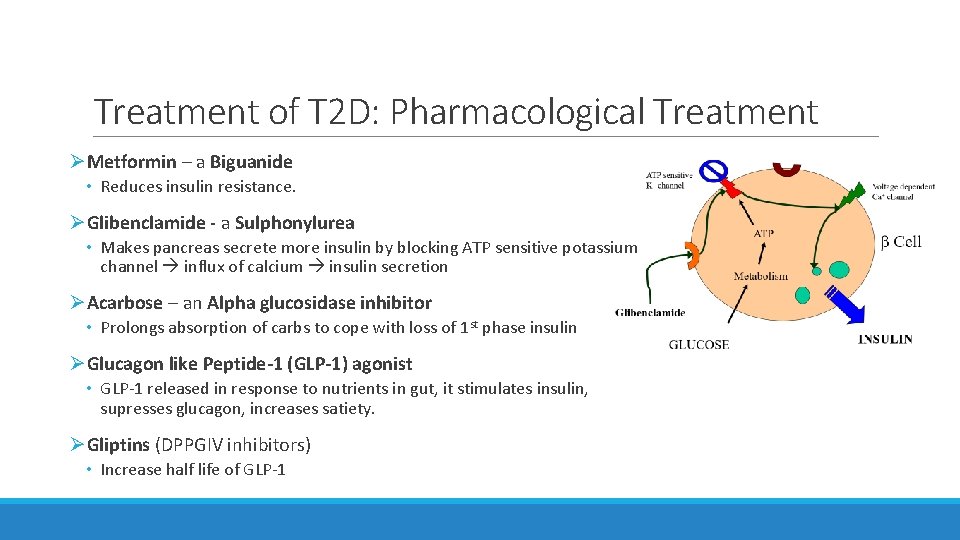
Treatment of T 2 D: Pharmacological Treatment ØMetformin – a Biguanide • Reduces insulin resistance. ØGlibenclamide - a Sulphonylurea • Makes pancreas secrete more insulin by blocking ATP sensitive potassium channel influx of calcium insulin secretion ØAcarbose – an Alpha glucosidase inhibitor • Prolongs absorption of carbs to cope with loss of 1 st phase insulin ØGlucagon like Peptide-1 (GLP-1) agonist • GLP-1 released in response to nutrients in gut, it stimulates insulin, supresses glucagon, increases satiety. ØGliptins (DPPGIV inhibitors) • Increase half life of GLP-1

Treatment of T 2 DM cont. Monitoring: • • Weight Hb. A 1 c Blood pressure Dyslipidaemia (cholesterol levels)
Question 4 A fat man with BMI 32 and fasting plasma glucose 12. 0 mmol/l comes to see you. a) What’s he probably got? (1 mark) b) What two non-drug treatments can you tell him to do? (1 mark) c) Name three classes of drugs that can be used for this condition, and name an example for each (3 marks)
Question 5 What is the basic pathogenesis of T 2 diabetes mellitus? (3 marks)

Complications of Diabetes LO 4: Explain the development and presentations of microvascular and macrovascular disease, recall risk factors for developing these complications and explain their management including prevention.

Microvascular and Macrovascular Disease Microvascular (disease of small blood vessels) • Retinal arteries Diabetic Retinopathy • Glomerular arteries Diabetic Nephropathy • Vasa Vasorum (tiny blood vessels that supply the nerves) Diabetic Neuropathy Macrovascular: (systemic disease commonly present in multiple arterial beds; disease of large blood vessels) • • Ischaemic Heart Disease Cerebrovascular Disease Peripheral Vascular Disease Renal Artery Stenosis Diabetic Foot disease- foot ulcer caused by diabetic neuropathy and peripheral vascular disease combined

Key Points about Micro and Macrovascular Disease • People without diabetes have nearly no risk of MICROvascular disease but they still get MACROvascular disease; diabetes just increases the risk of macrovascular disease • Having MICROvascular disease means you are more likely to have MACROvascular disease • MICROvascular disease morbidity MACROvascular disease morbidity AND mortality • Increased Hb. A 1 C= increased risk of both MICRO and MACRO • MACROvascular disease is more common in T 2 diabetics as they already have the traditional risk factors (central obesity, dyslipidaemia and hypertension) and with the independent risk factor of diabetes (insulin resistance and hyperglycaemia) the risk is heightened.

Microvascular Disease: Risk Factors • Severity of hyperglycaemia • Hyperglycaemia oxidative stress & hypoxia inflammatory signalling cascade local activation of pro-inflammatory cytokines inflammation retinopathy, nephropathy, neuropathy • Hypertension • Genetic • Hyperglycaemic memory • Even if you’ve had poor glycaemic control for a short period of time, it will cause an increased risk of microvascular complications compared to someone who has been well controlled throughout
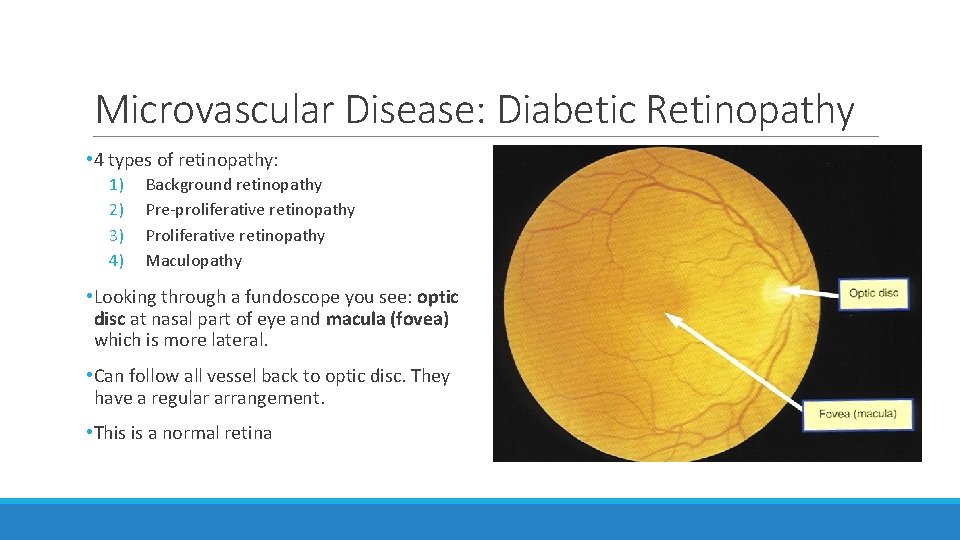
Microvascular Disease: Diabetic Retinopathy • 4 types of retinopathy: 1) 2) 3) 4) Background retinopathy Pre-proliferative retinopathy Proliferative retinopathy Maculopathy • Looking through a fundoscope you see: optic disc at nasal part of eye and macula (fovea) which is more lateral. • Can follow all vessel back to optic disc. They have a regular arrangement. • This is a normal retina
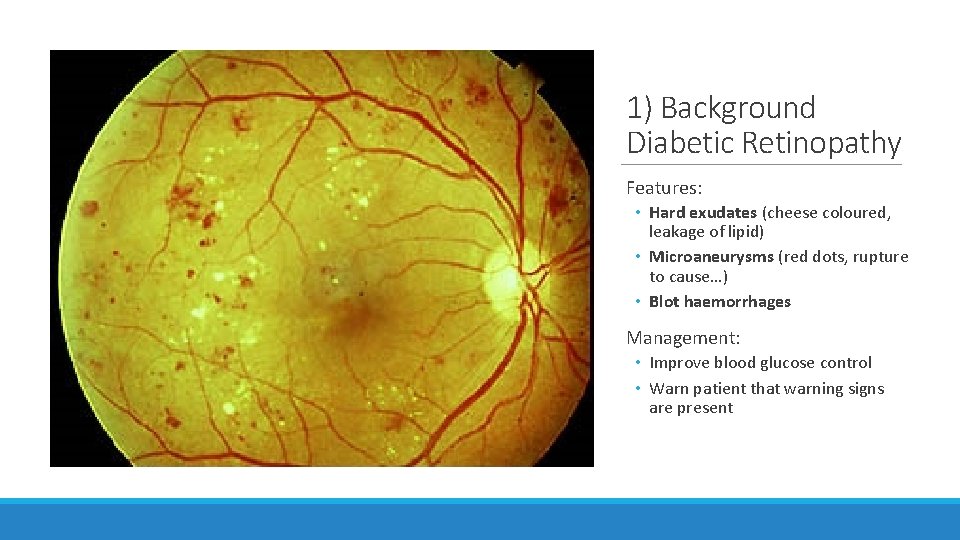
1) Background Diabetic Retinopathy Features: • Hard exudates (cheese coloured, leakage of lipid) • Microaneurysms (red dots, rupture to cause…) • Blot haemorrhages Management: • Improve blood glucose control • Warn patient that warning signs are present
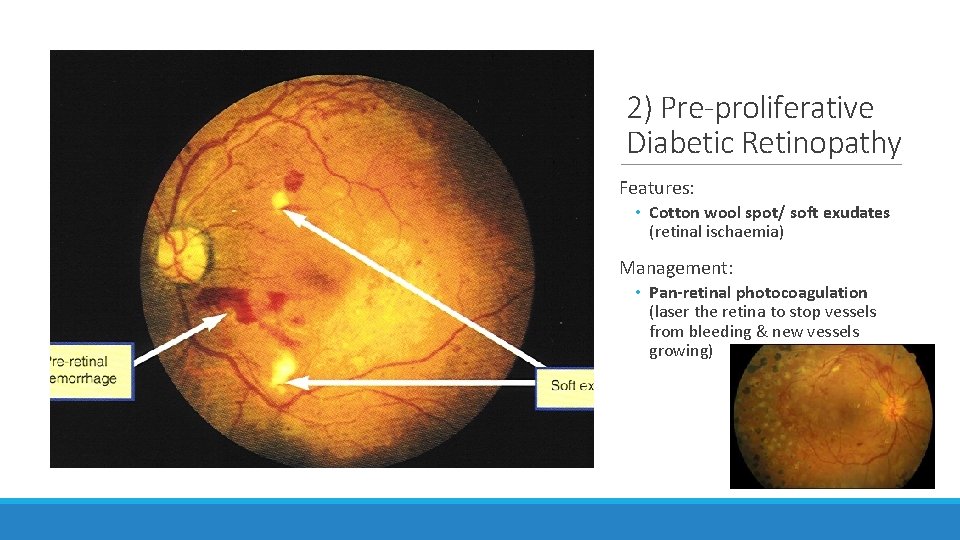
2) Pre-proliferative Diabetic Retinopathy Features: • Cotton wool spot/ soft exudates (retinal ischaemia) Management: • Pan-retinal photocoagulation (laser the retina to stop vessels from bleeding & new vessels growing)
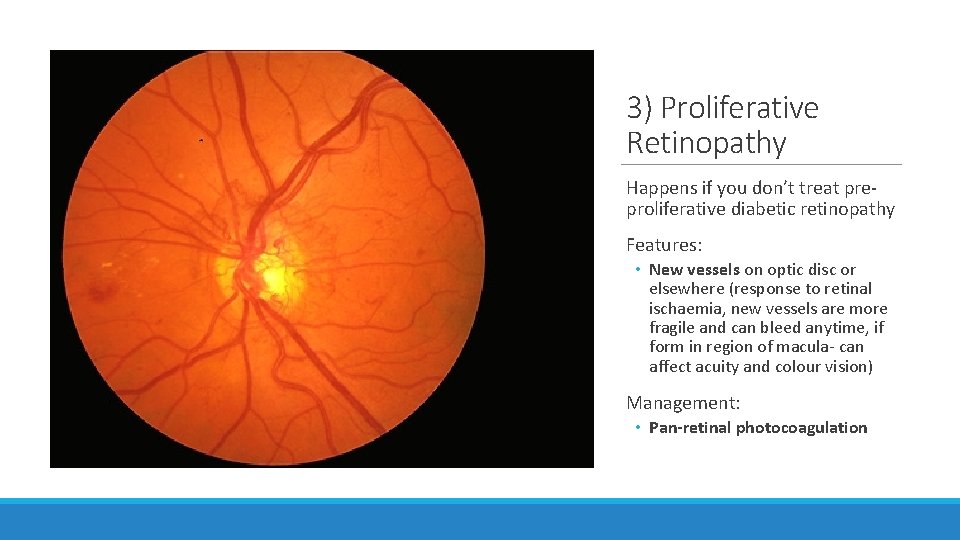
3) Proliferative Retinopathy Happens if you don’t treat preproliferative diabetic retinopathy Features: • New vessels on optic disc or elsewhere (response to retinal ischaemia, new vessels are more fragile and can bleed anytime, if form in region of macula- can affect acuity and colour vision) Management: • Pan-retinal photocoagulation
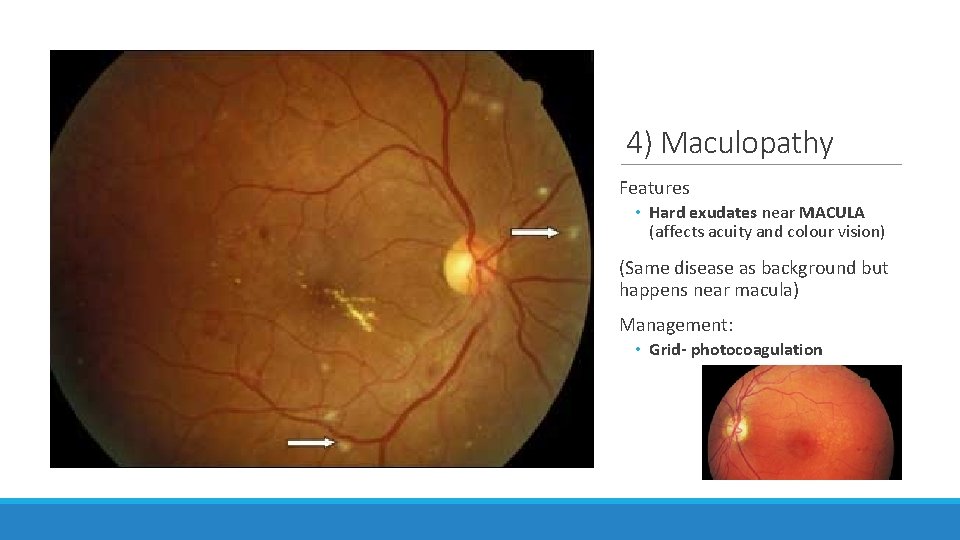
4) Maculopathy Features • Hard exudates near MACULA (affects acuity and colour vision) (Same disease as background but happens near macula) Management: • Grid- photocoagulation

Microvascular Disease: Diabetic Nephropathy Clinical features: • Progressive proteinuria – urine dipstick for albumin • Hypertension • Deteriorating kidney function Histological features: • Glomerular changes- less flexible and harder • Vascular- hardening • Tubointerstitial – secondary from^ Management: • • Blood glucose control (lower Hb. A 1 c) BP control- will slow down deterioration Inhibit renin-angiotensin system- ACE inhibitors reduce rate of creatinine increase (↑creatinine=↓kidney function Stop smoking

Microvascular Disease: Diabetic Neuropathy • Vasa vasorum get blocked Neuropathy • Most common cause of lower limb amputation (diabetic foot) • Types of Diabetic Neuropathy: • Peripheral polyneuropathy (sensory loss) • Mononeuropathy (motor loss) (e. g. pupil sparing III nerve palsy) • Autonomic neuropathy (loss of sympathetic and parasympathetic nerves)

Macrovascular Disease Ischaemic Heart Disease, Cerebrovascular Disease, Peripheral Vascular Disease, Renal Artery Stenosis Diabetes increased risk of atherosclerosis narrowing of arterial walls macrovascular disease Unlike with microvascular disease, managing blood glucose alone does not decrease the risk of macrovascular disease. You need to manage multiple risk factors of macrovascular disease: Modifiable: • • Dyslipidaemia (high cholesterol- treated with statin) High BP Smoking Diabetes Non- modifiable: age, sex, birth weight, genes
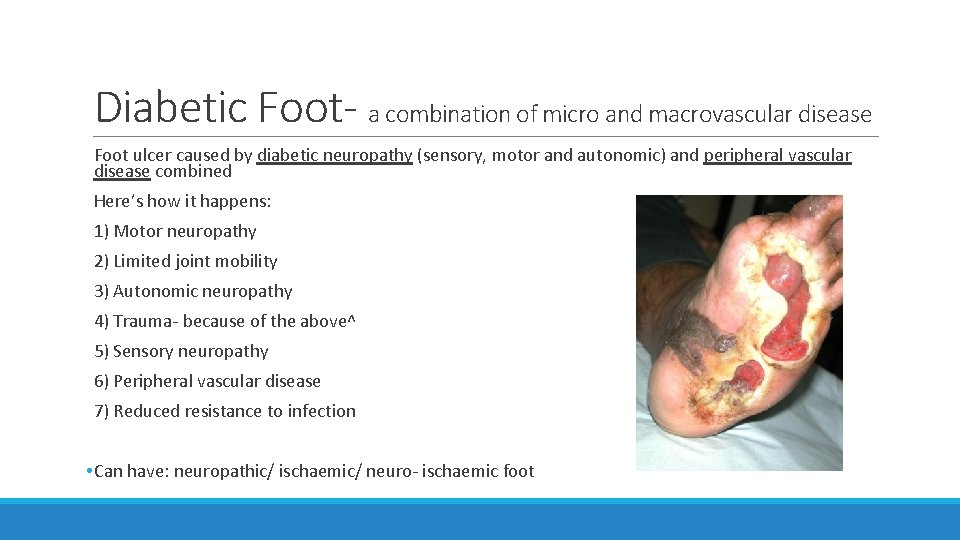
Diabetic Foot- a combination of micro and macrovascular disease Foot ulcer caused by diabetic neuropathy (sensory, motor and autonomic) and peripheral vascular disease combined Here’s how it happens: 1) Motor neuropathy 2) Limited joint mobility 3) Autonomic neuropathy 4) Trauma- because of the above^ 5) Sensory neuropathy 6) Peripheral vascular disease 7) Reduced resistance to infection • Can have: neuropathic/ ischaemic/ neuro- ischaemic foot

Diabetic Foot cont. To prevent: • Test pulse, temperature, numbness (microfilament), deformity • Foot care • Prevent hyperglycaemia, hypertension, high cholesterol, stop smoking To treat ulcer: • Relief of pressure • Antibiotics • Debridement • Revascularisation? • Amputation

Question 6 a) A man has a history of smoking, high BP and a diabetic foot, what would be your initial treatment? (1 mark) b) What other advice or additional things could you do to help? (2 marks) c) What causes ulcers in diabetics? (4 marks)
Question 7 Explain how you would distinguish between a patient with type 1 DM and type 2 DM? (4)
Thank you Please please fill in feedback before you leave: bit. ly/muslimmedics If you’re into flashcards: https: //www. brainscape. com/profiles/2683619
- Slides: 48