Di Peptidyl Peptidase4 DPP4 Inhibitors Hengameh Abdi Endocrine

Di. Peptidyl Peptidase-4 (DPP-4) Inhibitors Hengameh Abdi Endocrine Research Center Research Institute for Endocrine sciences Shahid Beheshti University of Medical Sciences 16 Nov 2017 25 Aban 1396 Tehran

Outlines n n n Case presentation Mechanism of action Efficacy Safety ¨ Weight gain ¨ Hypoglycemia ¨ Gastrointestinal adverse effects ¨ Focus on cardiovascular outcomes ¨ Pancreatic disease DPP-4 inhibitors and bone Conclusions 2

Case presentation § A 68 -year-old woman with 12 -year history of type 2 diabetes is referred to you. She takes metformin 2000 mg and gliclazide MR 30 mg daily. She respects the diet recommended by the nutritionist. § She has background retinopathy and peripheral neuropathy. § Cardiovascular history is unremarkable. § She claims that in the past, with increasing gliclazide dose, several hypoglycemic episodes had occurred. She fears hypoglycemia because she lives alone. § BMI: 29. 5 kg/m² 3

Case presentation § Laboratory tests: § FBS: 146 mg/dl § Hb. A 1 c: 8. 2% § Cr: 1. 2 mg/dl (e. GFR ~ 50 ml/min) § Which therapeutic approach would you recommend to her? A) Add basal insulin B) Add sitagliptin C) Add pioglitazone D) Add premixed insulin 4

DPP-4 inhibitors (Gliptins) § § § Sitagliptin: The first approved gliptin (2006) Saxagliptin Vildagliptin Linagliptin Alogliptin § Once-weekly DPP-4 inhibitors: Omarigliptin, … 5

Outlines n Case presentation n Mechanism of action n Efficacy Safety ¨ Weight gain ¨ Hypoglycemia ¨ Gastrointestinal adverse effects ¨ Focus on cardiovascular outcomes ¨ Pancreatic disease DPP-4 inhibitors and bone Conclusions n n n 6

Incretins and DPP-4: § Incretins: GLP 1 (Glucagon-Like Peptide-1) GIP (Glucose-dependent Insulinotropic Polypeptide) § GLP 1 is produced from the proglucagon gene in intestinal L cells and is secreted in response to nutrients. Glucose-dependent mode of action of incretin hormones § GLP 1 has a very short half-life in plasma (1 to 2 minutes) due to aminoterminal degradation by the enzyme dipeptidyl peptidase IV (DPP 4). As a result of DPP-4 activity, intact, biologically active GLP 1 represents only 10– 20% of total plasma GLP-1. 7
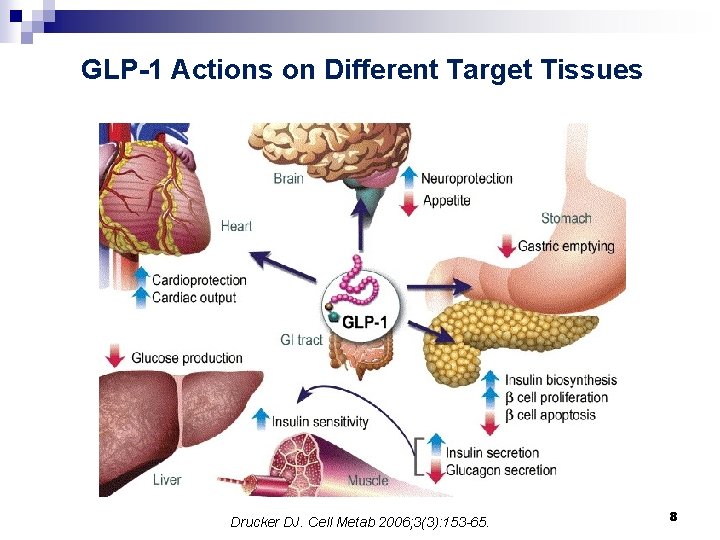
GLP-1 Actions on Different Target Tissues Drucker DJ. Cell Metab 2006; 3(3): 153 -65. 8

Outlines n n Case presentation Mechanism of action Efficacy Safety ¨ Weight gain ¨ Hypoglycemia ¨ Gastrointestinal adverse effects Focus on cardiovascular outcomes ¨ Pancreatic disease DPP-4 inhibitors and bone Conclusions ¨ n n 9

Aim: To evaluate the comparative effectiveness and safety of monotherapy and selected metformin-based combinations in adults with type 2 diabetes. Ann Intern Med. 2016; 164: 740 -751. 10
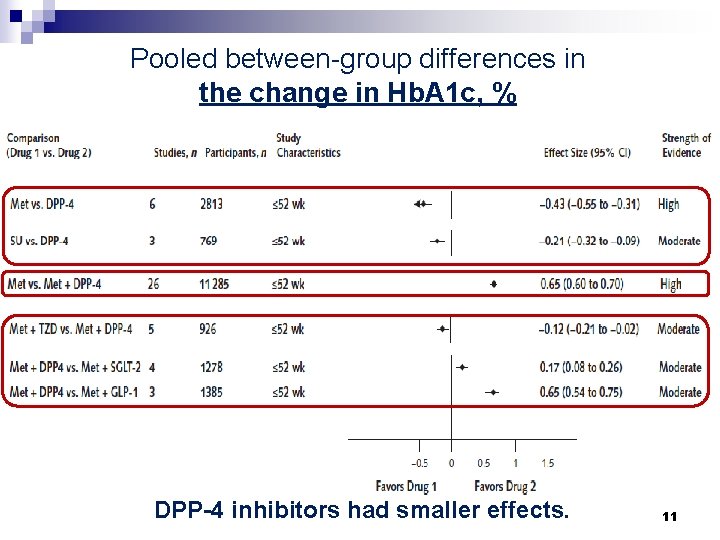
Pooled between-group differences in the change in Hb. A 1 c, % DPP-4 inhibitors had smaller effects. 11
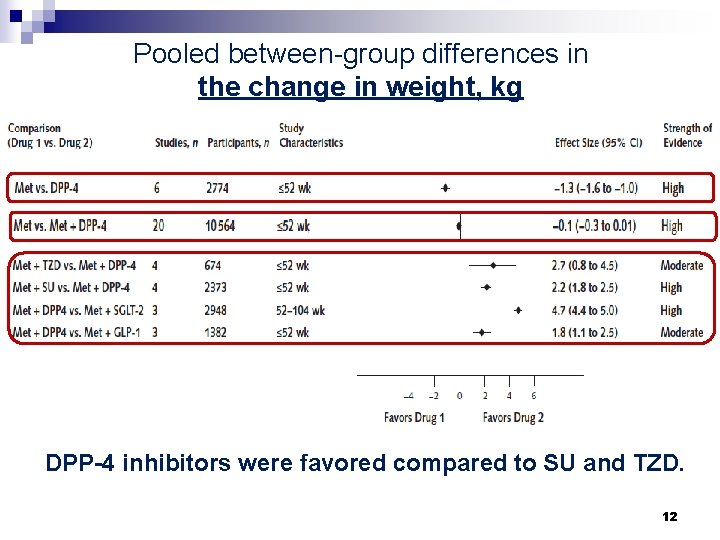
Pooled between-group differences in the change in weight, kg DPP-4 inhibitors were favored compared to SU and TZD. 12
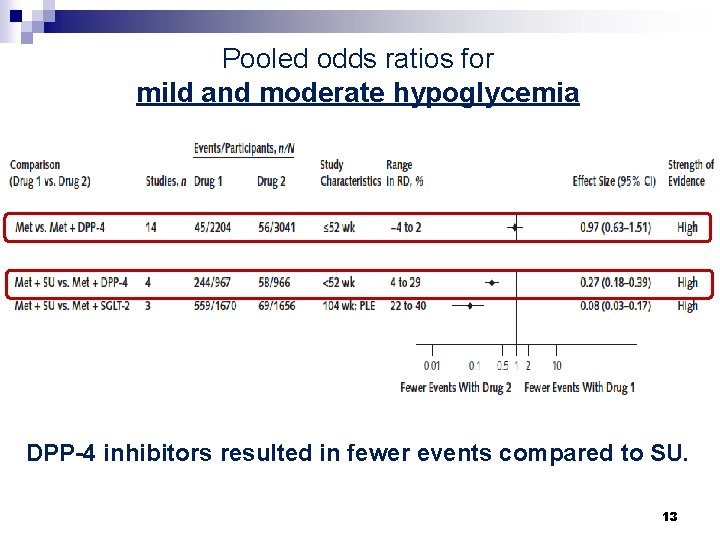
Pooled odds ratios for mild and moderate hypoglycemia DPP-4 inhibitors resulted in fewer events compared to SU. 13

Severe hypoglycemia n Based on 7 RCTs including 8345 individuals (high quality evidence), Met + DPP-4 resulted in significantly less episodes of severe hypoglycemia compared to the Met + SU: ¨ ¨ Pooled OR for studies < 52 wk: 0. 2 (95% CI 0. 1 -0. 6) Pooled OR for studies ≥ 52 wk: 0. 1 (95% CI 0. 03 -0. 3) 14
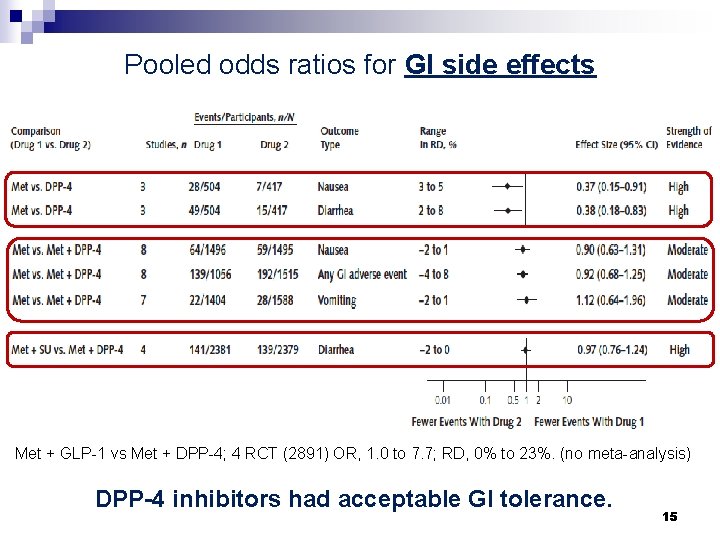
Pooled odds ratios for GI side effects Met + GLP-1 vs Met + DPP-4; 4 RCT (2891) OR, 1. 0 to 7. 7; RD, 0% to 23%. (no meta-analysis) DPP-4 inhibitors had acceptable GI tolerance. 15

Concluding remarks: § DPP-4 inhibitors have smaller effect on Hb. A 1 c. § DPP-4 inhibitors reduced or maintained the body weight. § DPP-4 inhibitors have less hypoglycemia risk compared to sulphonylureas. Ann Intern Med. 2016; 164: 740 -751. 16

Outlines n n Case presentation Mechanism of action Efficacy Safety ¨ Weight gain ¨ Hypoglycemia ¨ Gastrointestinal adverse effects ¨ Focus on cardiovascular outcomes Pancreatic disease DPP-4 inhibitors and bone Conclusions ¨ n n 17

Cardiovascular Effects of DPP-4 inhibitors § US Food and Drug Administration (FDA) issued an updated guidance for industry in 2008 requiring that preapproval and postapproval studies for all new antidiabetic drugs rule out excess cardiovascular risk. FDA/CDER Dec 2008. 18
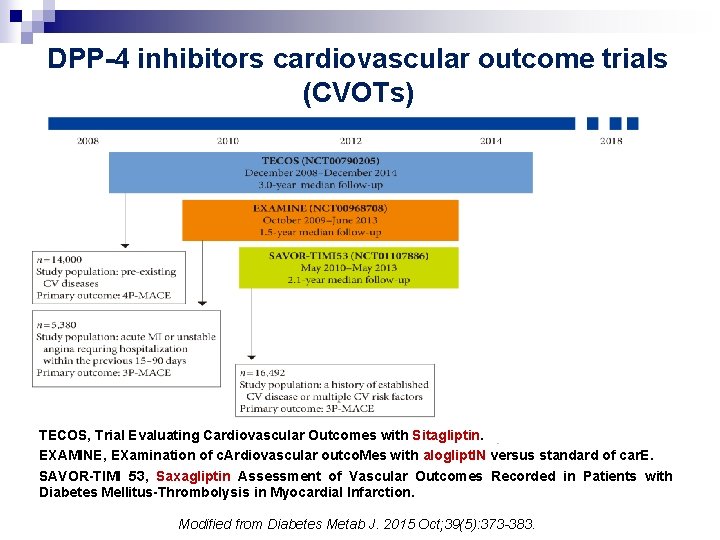
DPP-4 inhibitors cardiovascular outcome trials (CVOTs) TECOS, Trial Evaluating Cardiovascular Outcomes with Sitagliptin. EXAMINE, EXamination of c. Ardiovascular outco. Mes with aloglipt. IN versus standard of car. E. SAVOR-TIMI 53, Saxagliptin Assessment of Vascular Outcomes Recorded in Patients with Diabetes Mellitus-Thrombolysis in Myocardial Infarction. Modified from Diabetes Metab J. 2015 Oct; 39(5): 373 -383.

20
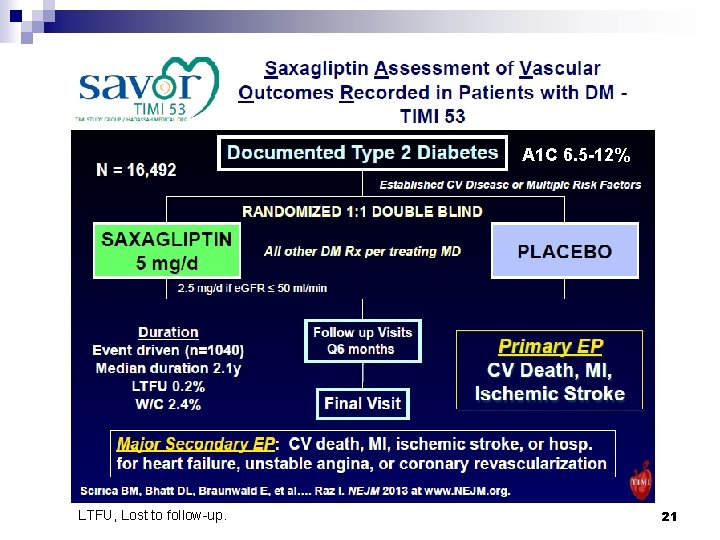
A 1 C 6. 5 -12% LTFU, Lost to follow-up. 21
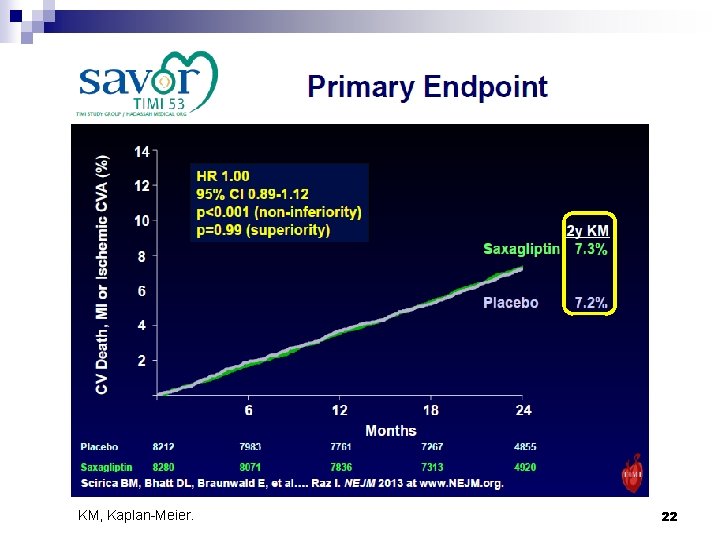
KM, Kaplan-Meier. 22
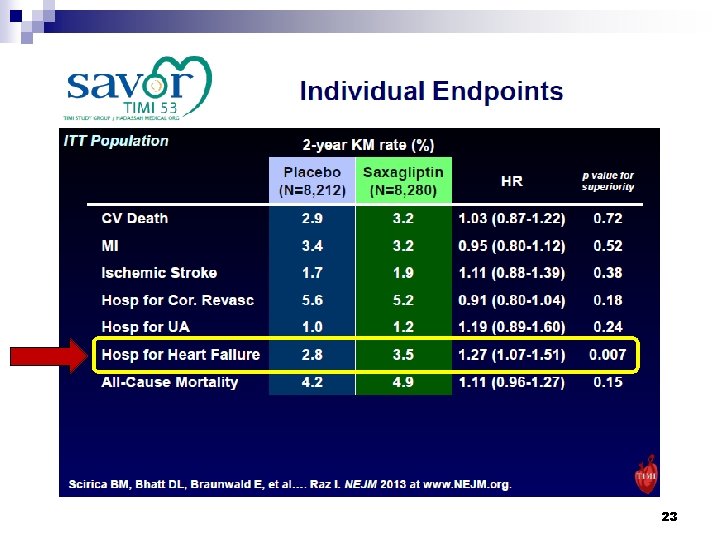
23

Conclusions § DPP-4 inhibition with saxagliptin did not increase or decrease the rate of ischemic events, though the rate of hospitalization for heart failure was increased. Scirica BM, et al. N Engl J Med 2013; 369: 1317 -26. 24

EXamination of c. Ardiovascular outco. Mes with aloglipt. IN versus standard of car. E. N Engl J Med 2013; 369: 1327 -35. 25

N=5, 380; Median follow-up: 1. 5 yr Takeda Development Center Americas, Inc. Presentation 26

ü MACE, cardiovascular death, nonfatal myocardial infarction, or nonfatal stroke. 27
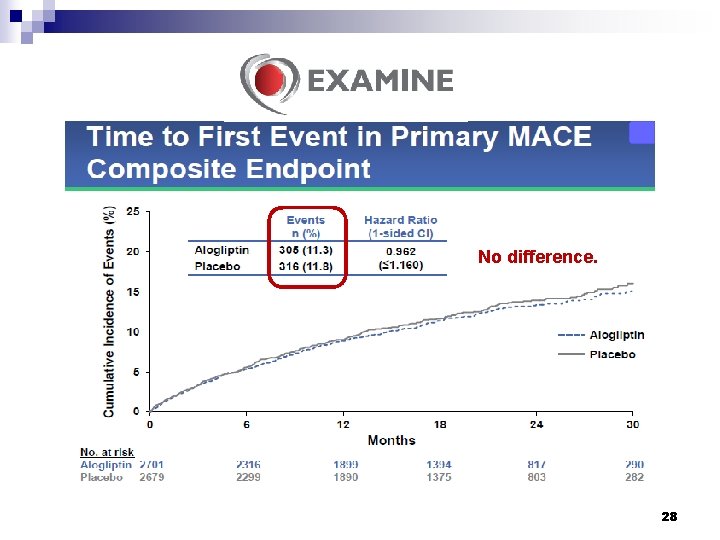
No difference. 28

EXamination of c. Ardiovascular outco. Mes with aloglipt. IN versus standard of car. E. Lancet 2015; 385: 2067– 76. 29
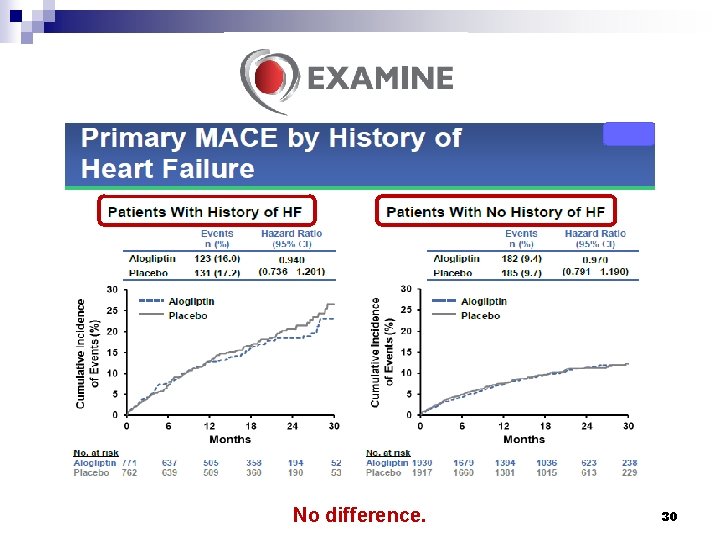
No difference. 30
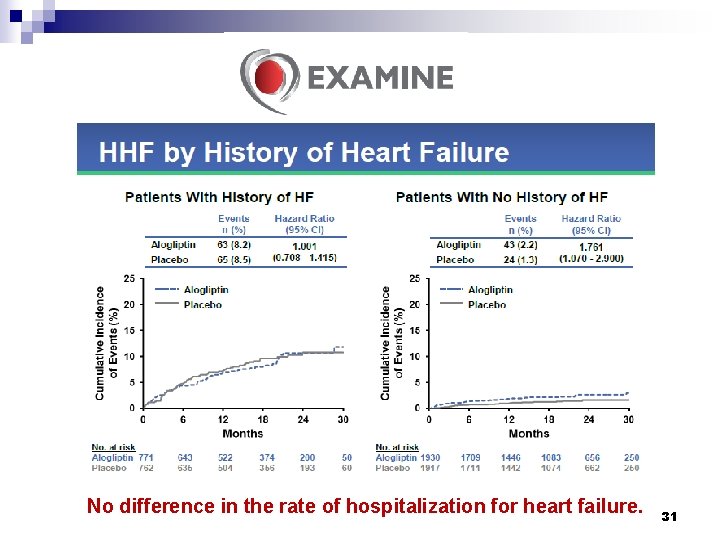
No difference in the rate of hospitalization for heart failure. 31

Conclusions § Among patients with type 2 diabetes who had a recent acute coronary syndrome, the rates of major adverse cardiovascular events were not increased with the DPP-4 inhibitor alogliptin as compared with placebo. § In patients with type 2 diabetes and recent acute coronary syndromes, alogliptin did not increase the risk of heart failure outcomes. White WB, et al. N Engl J Med 2013; 369: 1327 -35. Zannad F, et al. Lancet 2015; 385: 2067 -76. 32

§ Design: Randomized, double-blind, placebo-controlled, event-driven trial at 673 sites in 38 countries. § Population: Eligible patients had type 2 diabetes with established cardiovascular disease and were at least 50 years of age, with A 1 C of 6. 5 to 8. 0% when treated with stable doses of one or two oral antihyperglycemic agents or insulin (with or without metformin). Green JB, et al. TECOS Study Group NEJM 2015; 373: 232 -42 33

§ Intervention: either sitagliptin at a dose of 100 mg daily (or 50 mg daily if the baseline e. GFR was ≥ 30 and < 50 ml/minute/1. 73 m²) or matching placebo. § Primary composite outcome: cardiovascular death, nonfatal myocardial infarction, nonfatal stroke, or hospitalization for unstable angina (4 -point MACE). § Duration (median): 3 years ü MACE, Major Adverse Cardiovascular Event. Green JB, et al. TECOS Study Group NEJM 2015; 373: 232 -42 34
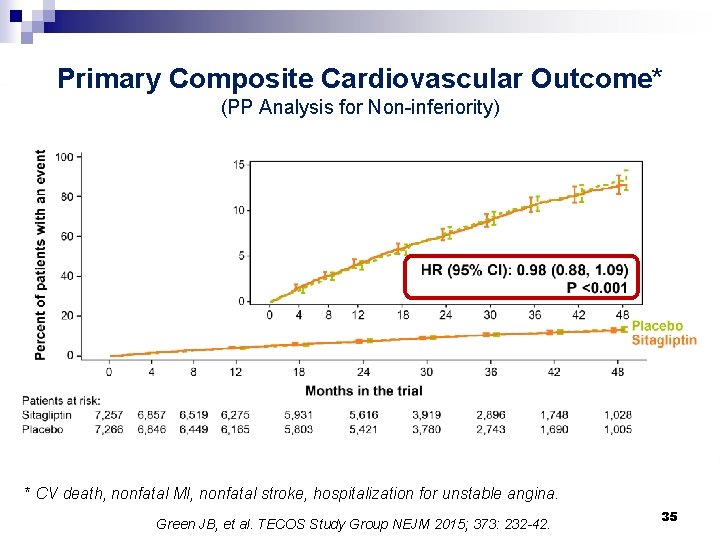
Primary Composite Cardiovascular Outcome* (PP Analysis for Non-inferiority) * CV death, nonfatal MI, nonfatal stroke, hospitalization for unstable angina. Green JB, et al. TECOS Study Group NEJM 2015; 373: 232 -42. 35
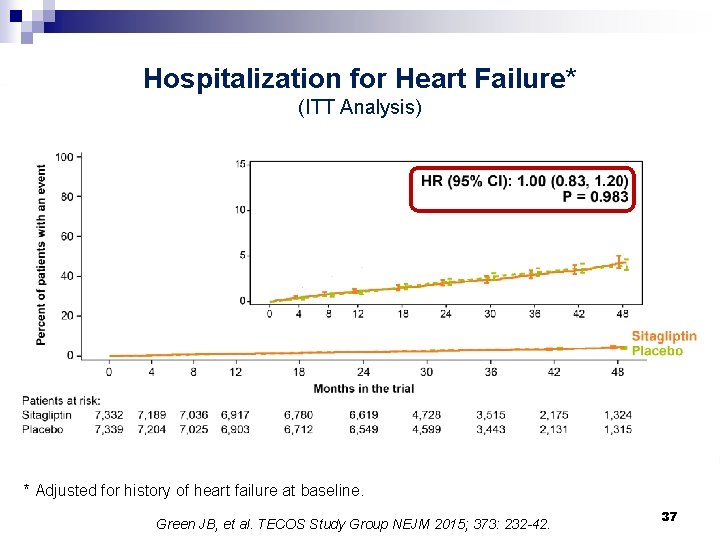
Hospitalization for Heart Failure* (ITT Analysis) * Adjusted for history of heart failure at baseline. Green JB, et al. TECOS Study Group NEJM 2015; 373: 232 -42. 37

Conclusions n Among patients with type 2 diabetes and established cardiovascular disease, adding sitagliptin to usual care did not appear to increase the risk of major adverse cardiovascular events, hospitalization for heart failure, or other adverse events. 38

Pooled estimate of hospitalization for heart failure in 3 large DPP-4 inhibitor CVOTs Mc. Guire DK, et al. JAMA Cardiol 2016; 1(2): 126 -135. 39

Outlines n n Case presentation Mechanism of action Efficacy Safety ¨ Weight gain ¨ Hypoglycemia ¨ Gastrointestinal adverse effects ¨ Focus on cardiovascular outcomes ¨ Pancreatic n n disease DPP-4 inhibitors and bone Conclusions 40

Pancreatic Safety of DPP-4 Inhibitors § A meta-analysis considering pancreatitis risk of incretinbased treatments: § 28 RCTs (19, 241 patients) compared DPP-4 inhibitors vs control: § 23 pancreatitis events (0. 12%); (OR 1. 06 [0. 46 -2. 45]). Li L, et al. BMJ 2014; 348 41
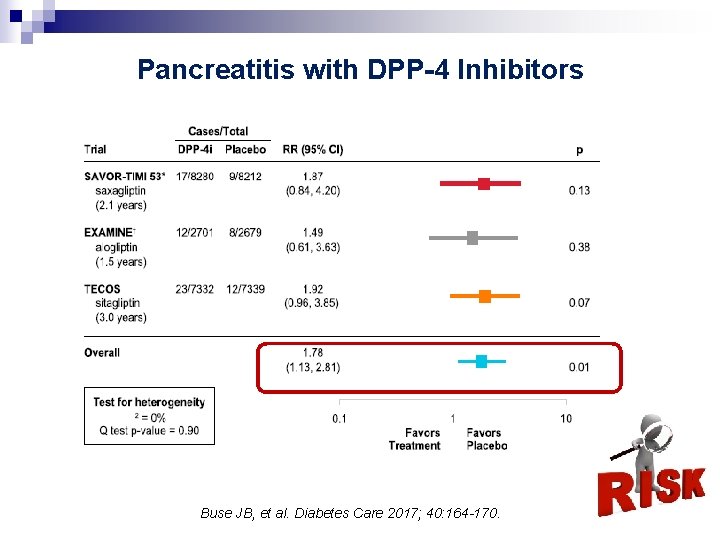
Pancreatitis with DPP-4 Inhibitors Buse JB, et al. Diabetes Care 2017; 40: 164 -170. 42
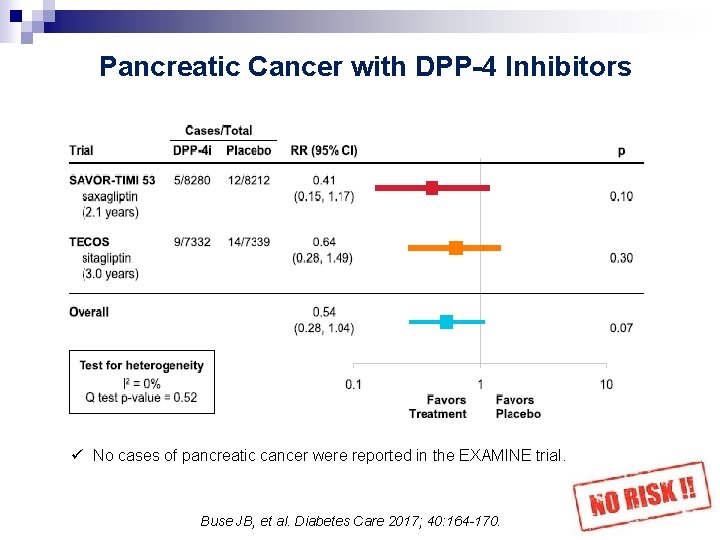
Pancreatic Cancer with DPP-4 Inhibitors ü No cases of pancreatic cancer were reported in the EXAMINE trial. Buse JB, et al. Diabetes Care 2017; 40: 164 -170. 43

Outlines n n Case presentation Mechanism of action Efficacy Safety ¨ Weight gain ¨ Hypoglycemia ¨ Gastrointestinal adverse effects ¨ Focus on cardiovascular outcomes ¨ Pancreatic disease n DPP-4 inhibitors and bone n Conclusions 44
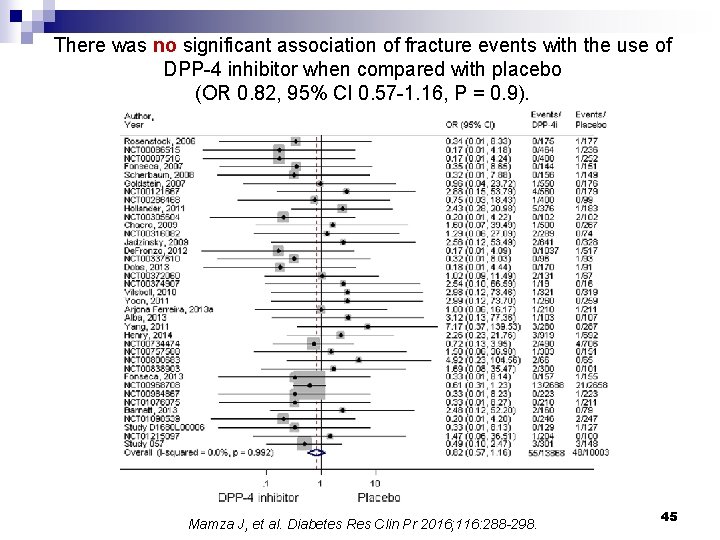
There was no significant association of fracture events with the use of DPP-4 inhibitor when compared with placebo (OR 0. 82, 95% CI 0. 57 -1. 16, P = 0. 9). Mamza J, et al. Diabetes Res Clin Pr 2016; 116: 288 -298. 45
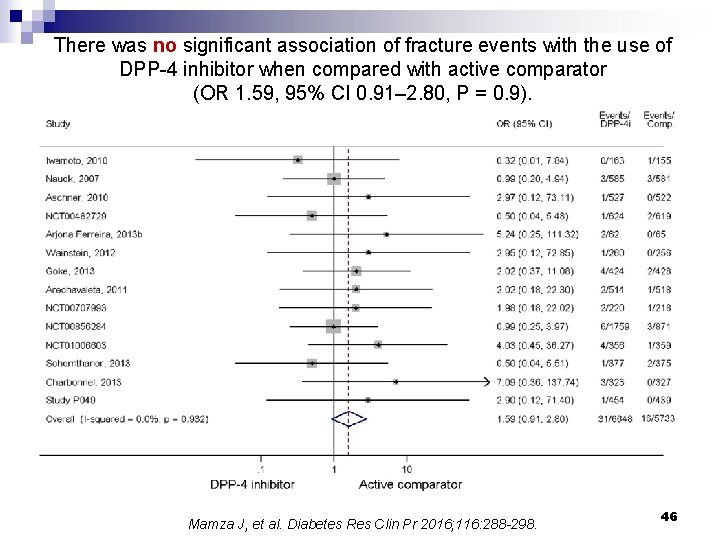
There was no significant association of fracture events with the use of DPP-4 inhibitor when compared with active comparator (OR 1. 59, 95% CI 0. 91– 2. 80, P = 0. 9). Mamza J, et al. Diabetes Res Clin Pr 2016; 116: 288 -298. 46

Outlines n Case presentation Mechanism of action Efficacy Safety ¨ Weight gain ¨ Hypoglycemia ¨ Gastrointestinal adverse effects ¨ Focus on cardiovascular outcomes ¨ Pancreatic disease DPP-4 inhibitors and bone n Conclusions n n 47

DPP-4 inhibitors as one available incretin-based therapy Modified from Inzucchi SE, et al. Diabetes Care 2015; 38: 140– 149. 48
49

Dosage of sitagliptin (available in Iran) n 100 mg daily n Renal impairment: Cr. Cl > 50 m. L/min: Dose adjustment not necessary. ¨ Cr. Cl 30 -50 m. L/min: 50 mg daily ¨ Cr. Cl < 30 m. L/min: 25 mg daily ¨ n Hepatic impairment: ¨ ¨ Mild to moderate impairment: Dose adjustment not necessary. Severe impairment: Not studied. 50

Case presentation § A 68 -year-old woman with 12 -year history of type 2 diabetes is referred to you. She takes metformin 2000 mg and gliclazide MR 30 mg daily. She respects the diet recommended by the nutritionist. § She has background retinopathy and peripheral neuropathy. § Cardiovascular history is unremarkable. § She claims that in the past, with increasing gliclazide dose, several hypoglycemic episodes had occurred. She fears hypoglycemia because she lives alone. § BMI: 29. 5 kg/m² 51

Case presentation § Laboratory tests: § FBS: 146 mg/dl § Hb. A 1 c: 8. 2% § Cr: 1. 2 mg/dl (e. GFR ~ 50 ml/min) § Which therapeutic approach would you recommend to her? A) Add basal insulin B) Add sitagliptin C) Add pioglitazone D) Add premixed insulin 52

Thanks for your patience Bisotun, Kermanshah, Iran.
- Slides: 52