Deviations Writing Quality Assurance Reports QARs Global Quality

Deviations: Writing Quality Assurance Reports (QARs) Global Quality Operations - Training and Program Development

Why is it difficult to write QARs? Time and / or stress l Don’t like writing / find it difficult l Too many different groups / people involved l We understand our own operations and thus don’t fully explain everything l We don’t have all the information / facts l Politics / blame And l Expectations are not clear l Not sure the right level of detail l Don’t have training l 2

Learning Objectives l l Identify the “ 5 Cs” to keep in mind when writing QARs Identify at least 3 tips for staying objective in our writing List the components of the WICS Model Identify SMART requirements for writing corrective and preventative actions 3

Agenda l l l Introduction Prepare for Writing • Purpose, Audience • Organise Ideas Technical Writing • Writing Styles • Five C’s Language Use WICS Model • • WICS / DMAIC WICS by section Closing Points 4

Preparing for Writing l You need to: • Establish your purpose • Know your audience • Gather the facts • Organize your Ideas 5

Establish your Purpose l What is the purpose of QARs? • • To accurately describe an event To identify why it happened (the cause) To facilitate actions to avoid it happening again So we can fully understand what happened (when we read it in a few years time, maybe with an Auditor!) 6

Know your Audience l Ask yourself these questions: • How knowledgeable is my audience? • What do they already know? • What do they need to know? • What is my audience’s purpose for reading? • To approve the content? • To make a decision? • To know what to do next? • Does my audience have special concerns about the subject? What are they? 7
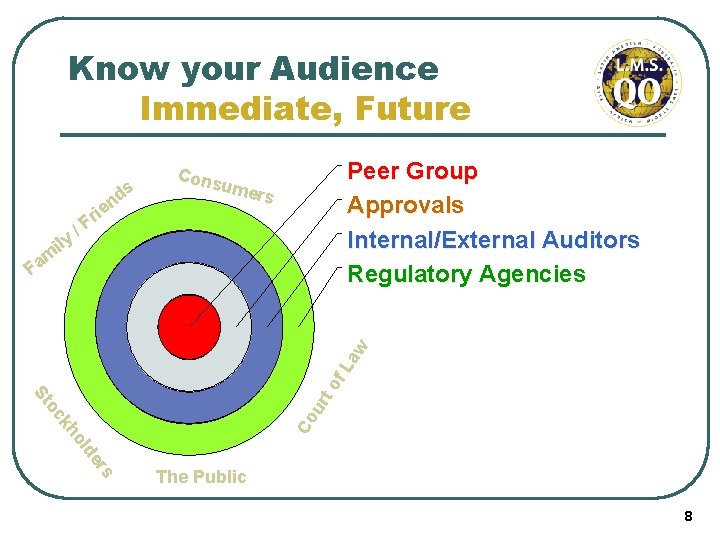
Know your Audience Immediate, Future s d n ily am ie r /F Peer Group Approvals Internal/External Auditors Regulatory Agencies Cons umer s rs de ol kh Co oc ur St to f. L aw F The Public 8

Know your Audience l Always define acronyms the first time you use them. l Reduce your use of jargon l Write for a knowledge level that would minimize questions if you presented the information to your “peer group”. Lets define “peer group” 9

Organize your Ideas l What do you know? l What else do you have? • Process and/or Structure Description • Facts on the event • Supporting information • Applicable scientific principles / concepts • Unsubstantiated ideas / testimonials and remaining questions 10

Agenda l l l Introduction Prepare for Writing • Purpose, Audience • Organise Ideas Technical Writing • Writing Styles • Five C’s • Language Use WICS Model • • WICS / DMAIC WICS by section Closing Points 11

Writing Styles l Literary writing conveys a mood, a story, and to paint a picture for the audience. The purpose is to entertain. l Technical writing conveys facts in an objective way. Feelings and picturesque speech are out of place - objectivity is the key. The purpose is to transmit information. 12

The 5 Cs l Comply with the 5 C’s Rule • • • CLEAR CONCISE COMPLETE COHERENT CONSISTENT 5 C’s 13

The 5 Cs - Clear l l The sentence should have one and only one meaning. Precise and organized words Gets to the point Known language 14

The 5 Cs - Clear The operator was unsure of the speed of the mixer. The speed of the mixer is defined in the SOP but during this batch, the operator did not record the speed. or The speed of the mixer is not defined anywhere. 15

The 5 Cs - Concise Simple words, few syllables l Avoid duplication l Short sentences l Short paragraphs l It is NOT a Literary contest! 16

The 5 Cs - Complete l Contains all needed information • • • WHO – Position Title, Name (If appropriate/ needed) WHAT – Action, event description WHERE – Place, Facility, specific operation section WHEN – Time, day, stage, shift HOW – Methods, technique, conditions, etc. WITH WHAT – Materials, instruments, equipments, resources 17

The 5 Cs - Complete Graphical and other visual tools may be extremely useful, eg. Charts, process flows, diagrams, photographs, etc. “A picture speaks a thousand words” Defect B Defect A With QARs, it’s important to attach enough information to support the decisions that were made. 18
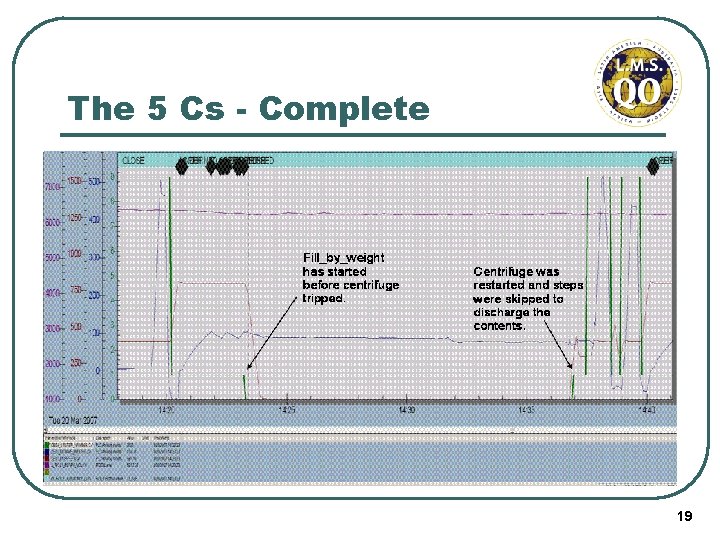
The 5 Cs - Complete 19

The 5 Cs - Coherent l l Information, facts, data, descriptions & conclusions should follow an orderly sequence which is easily understood by a reader not familiar with the site, products or processes. To do this: • Limit the number of ideas in a sentence. • Use “white space” to separate ideas • Use logical patterns to present your information, eg. By time sequence, by topic/subject, cause and effect, etc. 20

The 5 Cs - Coherent l l l Chunking information is easier on the reader If using paragraphs, start the paragraph with a topic sentence (e. g. Impact on Water System) Remember that white space helps the reader • Create using margins, tables, spacing, indentations 21

The 5 Cs - Consistent l Be consistent or logical with your use of: • Tense (eg. past / present / future) • Sentence structure • Descriptors / identifiers (eg. Equipment name • • or equipment number) Terminology (eg. Operator, technician, etc) Format (eg. Bullets, numbers, paragraphs indented, etc) 22

Language Use - Subjective vs. Objective – Based on observable phenomena, presented factually. • • The contamination weighs 14. 9 g, is 5 cm long and 2 cm wide. It is black and rectangular in shape. Subjective – Information is altered by the observers' opinions / thoughts / experiences. • • The contamination is a small object that feels quite heavy. It has often appeared before, but I had thought we had fixed it. 23

Language Use – Tips for staying objective l l l Always consider unintended consequences: • • E. g. Due to a lack of knowledge of the technician… E. g. Due to the excessive heat in the laboratory… E. g. Due to the SOPs being out of date… E. g. Due to our inability to deliver the results on time…. Avoid unnecessary adverbs and adjectives: • Writing furiously, working strangely, drastic drift Avoid unsupported superlatives: • This is the biggest particle size we’ve ever seen… …more… 24

Language Use – Tips for staying objective l Avoid imprecise words that you can’t quantify/justify: …less than usual… l Use universal statements carefully: E. g. All, never, always, every, etc. l Base speculation on sound logic, or justifiable science. l Don’t use double negatives ‘Resources will not be unlimited’ ‘There is no reason why the piece of equipment should not be replaced’ 25

Language Use - Legal l Avoid statements that reflect on Compliance or Legal Status: • • l l A “Violation” of Law “Defective” product Legal judgments and interpretations should be avoided. Do not use inflammatory words such as: • Careless, terrible, dangerous, intentional, severe, reckless, incompetent, etc. 26

Language Use - Grammar & punctuation How much does punctuation really matter? Woman without her man is nothing. Woman, without her man, is nothing. Woman, without her, man is nothing. 27

Language Use - Purpose of punctuation l l l Separates thoughts Shows changes in ideas within a sentence Can indicate emotion or urgency! Handout available on English punctuation use (Request from Vicci) Be careful using Microsoft Word (using the spill check function) 28

Agenda l l l Introduction Prepare for Writing • Purpose, Audience • Organize Ideas Technical Writing • Writing Styles • Five C’s Language Use WICS Model • WICS / DMAIC • WICS by section Closing Points 29

What is the WICS Model? l l Model to help you structure writing/reviewing QARs Uses a review/assessment sheet Gives you guidance and a way to cross check Broken into: • What Happened • Impact/Investigation • Cause • Stop and Support (W) (I) (C) (S) 30

31
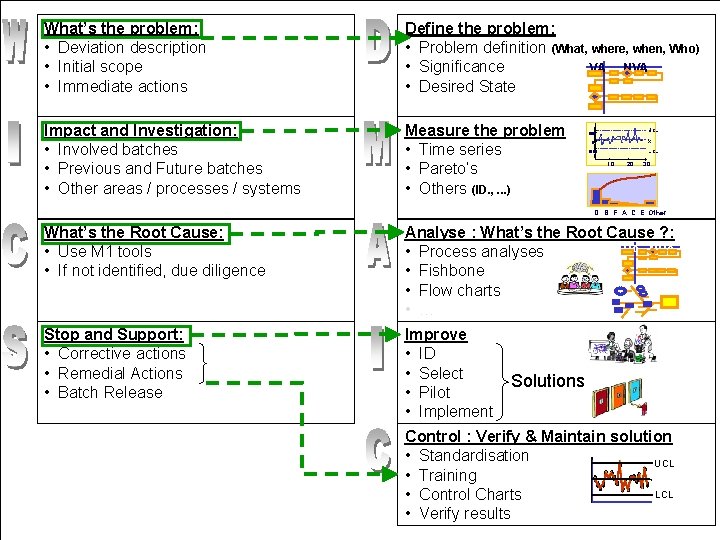
What’s the problem: • Deviation description • Initial scope • Immediate actions Define the problem: • Problem definition (What, where, when, Who) VA NVA • Significance • Desired State Impact and Investigation: • Involved batches • Previous and Future batches • Other areas / processes / systems Measure the problem • Time series • Pareto’s • Others (ID. , . . . ) UCL 1000 X 0 LCL -1000 10 20 30 D B F A C E Other What’s the Root Cause: • Use M 1 tools • If not identified, due diligence Analyse : What’s the Root Cause ? : VA NVA • Process analyses • Fishbone • Flow charts • … Stop and Support: • Corrective actions • Remedial Actions • Batch Release Improve • ID • Select • Pilot • Implement Solutions Control : Verify & Maintain solution • Standardisation UCL • Training LCL • Control Charts • Verify results

W – What Happened? l Includes these sections: • Deviation description • Immediate actions (reaction) • Scope of the incident 33

W – What Happened? l Deviation description • • Focus on being objective and factual Do not include irrelevant items (information that belongs in another section) Speculation is not appropriate in this section Provide detail: such as dates, times, Provide context; such as where in the process the observations were made. Provide comparison; such as what is normal or expected or required Use your process knowledge and experience Check for clarity with a “peer” 34

W – What Happened? l Immediate actions taken • • l Depending upon the type of incident, actions may be necessary to prevent further product/production loss and/or prevent further expansion of the problem. DO NOT include remedial or corrective actions (unless they really were immediate and did not need approval) Examples: • • Stop further production Adjust processing parameters (e. g. adding reactant to drive a process to completion, increase mix times, etc. ) Turn off tap, remove obstruction, etc. Notify Supervisor / Process Engineer 35

W – What Happened? l Scope of incident - initially • Define the batches that were in process at the • time. If no batches involved, define the area / process / equipment that are involved. 36

I – Impact / Investigation l Includes these sections: • What is the full scope of this incident? • How does the incident impact on the batch(s) • we produce? What other concerns does the incident raise? Note: This information can be included in an Impact Assessment Document (MSWord) 37

I – Impact / Investigation l Scope of incident • Verify the initial scope by: • Identify the last time/date the process and equipment were working properly • Direct observation or records (logbooks, schedules, etc) • If scope changes during the investigation, document the rationale 38

I – Impact / Investigation l Impact on the batch during manufacture must be understood • l l Batch was subsequently processed, put on hold, etc Impact on the compliance of the batch must be clear. E. g. what is the affect to: • Validation status • Regulatory filings / dossiers • c. GMP standards Don’t assume – check and document. 39
I – Impact / Investigation l l Remember to consider impact on: • • Previous batches Future batches Impact on areas / processes / systems may also need to be reviewed • Define what resolved each concern and the proof (records) maintained to support this. 40

C - Cause l l Root Cause: These are the conditions or actions which caused the deviation, that if corrected, would prevent recurrence. Methods of root cause analysis are many and varied. 41

C - Cause l l Simple or minor incidents may have a quick investigation with an easily identifiable root cause. Complex incidents may need a complete root cause investigation to establish: • The most probable cause(s) and the basis on which the final root cause(s) was reached. • What factors were specifically eliminated as a root cause, and the rationale for that judgment. Use your RFT Tools! 42

C - Cause l Root Cause – None found? • It is equally important to document what factors were evaluated and eliminated, and on what basis they were eliminated as a root cause. • This is the key to due diligence • Demonstrates to compliance officials a good faith effort to seek out and find the root cause. l l Need to document the investigation and outcome – may be useful next time (to you!) Need to set detection/monitoring points to spot recurrence early 43

S – Stop and Support l Includes these sections: • Remedial and Corrective Actions • Lot Disposition 44

S – Stop and Support What is the difference… …between Immediate Reactions Investigative Actions Remedial Actions Corrective Actions 45

S – Stop and Support Immediate Reaction l l l Actions taken in response to the problem to immediately minimize or rectify the scope or impact of the problem. Must be recorded. Examples: • Repairing the pipe, turning off the tap, etc. 46

S – Stop and Support Investigative Actions l l l Actions taken to determine or clarify the scope, impact or root cause of the deviation on the product or process. Must be recorded. Additional processing / testing needs to be defined and agreed in conjunction with QA. 47

S – Stop and Support Remedial Actions l l l Actions taken until you can figure out how to really fix the problem. If it’s a simple and isolated incident, remedial action may be all that’s appropriate. No need to look further into the root cause or develop an action plan Must be implemented, communicated and recorded. Examples: • • • Substituting a component temporarily Retraining an individual Implementing an interim process or procedure 48

S – Stop and Support Corrective Actions l l l Actions taken to deal with an existing non-conformity • • To fix the process To fix the batch(s) • • Reprocessing / rework Inspection/sorting • • • Adding a new supplier to the approved vendor list Retraining operator(s) Calibrating a micrometer Need to determine what is the real root cause and develop a corrective action plan. These actions must be implemented, communicated and recorded. Example, for the batch: Example, for the process: 49

S – Stop and Support Preventive Actions l l Corrective and Preventive actions (CA and PA) are closely related CAPA are often misinterpreted / used, as they are based on different inputs: • • l CA is a reactive process designed to address existing non-conformities (something has gone wrong) PA is a proactive process used to address potential problems (nothing has gone wrong, yet!) Thus, PA is not relevant for Deviations / QARs…. Reference: Robitaille, D. 2001. “The Corrective Action Handbook”. Paton Press. 50

S – Stop and Support SMART Corrective Actions l l l Actions/commitments must be SMART • • • Specific Measurable Accountable Realistic Timely • So that you don’t have to go back and read the QAR again to understand what is needed. Must stand alone Must be verified • • Training – reference copy of training records. Update SOP – reference when SOP version was approved 51

S – Stop and Support SMART Corrective Actions l l Specific • With enough detail so that when you read it, you know what needs to be done; includes the old and new (to pinpoint what needs to be changed and allow easier approval) Measurable • Includes some sort of acceptance criteria (pass/fail; update; increase by %; install; reduce by distance; review of 5 Batch Records. ) 52

S – Stop and Support SMART Corrective Actions l l l Accountable • Allocated to someone by personal name; agreed commitments are tracked. Realistic • Linked to the conclusion and if implemented will reduce the likelihood of the problem recurring; is not just a “sticking plaster” or “stop-gap” to write something in the QAR Timely – Needs to be based on: • • • Criticality of the problem (effect on production process, product, people, etc. ) Frequency or likelihood of event occurring again Amount of resources needed (low hanging fruit) 53

S – Stop and Support Are these good Corrective Actions? l “Management will discuss this issue with all personnel at the next team meeting and all personnel will be reminded of the importance of the checkers role. ” l “Update recipe to change the temperatures. ” l “We will communicate this issue to all affected colleagues. ” l “Add to preventative maintenance schedule. ” 54

S – Stop and Support l Lot Disposition • Determined by QA based on the following criteria: • Have we fully addressed all of the concerns with this • • • product / lot? Have all corrective actions (for the batch/lot) been completed? Have we got to the real root cause? Does it all make sense (logical / scientific)? Can we prove all of this for the future (records)? Have we followed Quality Risk Management principles in making our batch disposition decision? 55

56

Closing Points l l l l QARs reflect on you and the company Use WICS to help you write a good QAR Poorly written QARs not only confuse issues but may detract from the substance of the report and lead to regulatory observations Take advantage of a chance to prepare / approve a document you can be proud of Grammar, punctuation and writing do make a difference QARs must stand on their own and not be subject to misinterpretation QARs should still be clearly understood if reading them a few years later 57

Finally… l l l Questions? Complete the training record Complete the learning evaluation 58

References and Reading l l Robitaille, Denise. 2001. “The Corrective Action Handbook”. Paton Press. Robitaille, Denise. 2001. “The Preventive Action Handbook”. Paton Press. 59
- Slides: 59