Development of the Periodic Table Early chemists used

Development of the Periodic Table

Early chemists used the properties of elements to sort them into groups, or triads. 1668: 14 elements

1864: Newlands and Meyer arrange 56 elements by atomic mass and elements notice similar properties. 1869: Dmitri Mendeleev got an idea from his 63 favorite card game… elements
1864: Newlands and Meyer arrange 56 elements by atomic mass and elements notice similar properties. 1869: Dmitri Mendeleev got an idea from his 63 favorite card game… elements

1864: Newlands and Meyer arrange 56 elements by atomic mass and elements notice similar properties. 1869: Dmitri Mendeleev got an idea from his 63 favorite card game… (*might be elements apocryphal) Mendeleev arranged the elements by increasing atomic mass and by similar properties.
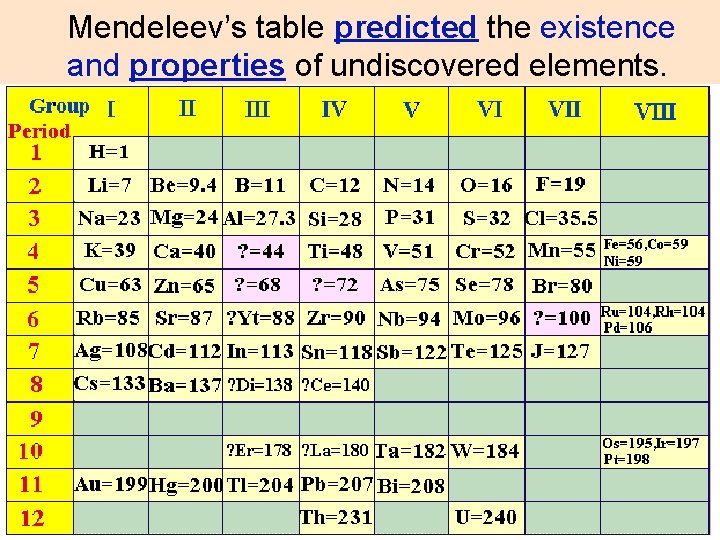
Mendeleev’s table predicted the existence and properties of undiscovered elements.

The four predicted elements lighter than the rare-earth elements, eka-boron, ekaaluminium, eka-manganese, and ekasilicon, proved to be good predictors of the properties of scandium, gallium, technetium and germanium respectively.

Elements later discovered (… in Mendeleev’s time): Gallium, Germanium, and Scandium Properties of Gallium Mendeleev’s predictions 68 Actual Properties 69. 7 6. 0 g/cm 3 5. 9 g/cm 3 Appearance soft gray metal Melting point Low melting point 29. 8°C Atomic mass Density

Gallium Elements later discovered (… in Mendeleev’s time): Gallium, Germanium, and Scandium Properties of Gallium Mendeleev’s predictions 68 Actual Properties 69. 7 6. 0 g/cm 3 5. 9 g/cm 3 Appearance soft gray metal Melting point Low melting point 29. 8°C Atomic mass Density

1864: Newlands and Meyer arrange 56 elements by atomic mass and elements notice similar properties. Dmitri Mendeleev got an idea from his 1869: favorite card game… 63 elements Mendeleev arranged the elements by increasing atomic mass and by similar properties. 1913: Moseley arranges elements by the atomic newly discovered “atomic number. ” numbers
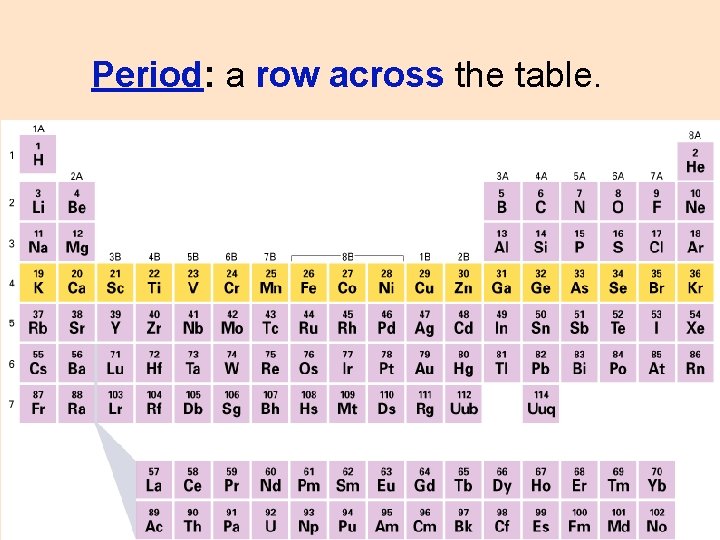
Period: a row across the table.
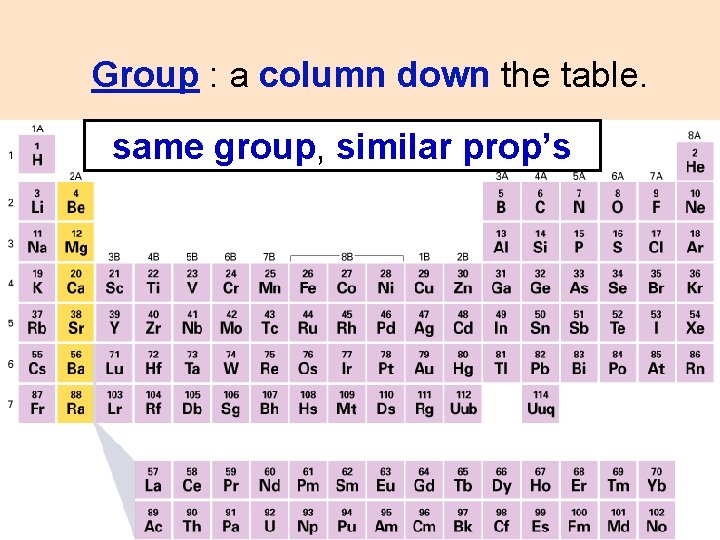
Group : a column down the table. same group, similar prop’s

The modern periodic table arranges elements by increasing atomic number. Periodic Law: periodic repetition of properties • properties change across a period. • properties repeat from period to period.
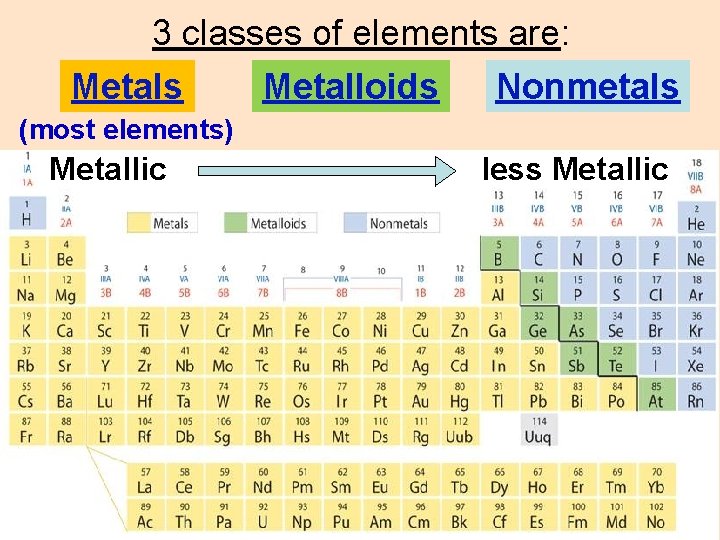
3 classes of elements are: Metals Metalloids Nonmetals (most elements) Metallic less Metallic

Metals luster (shiny) malleable (sheets) ductile (wires) good conductors (heat/electricity) Nonmetals most gases brittle solids Metalloids dull/shiny brittle poor conductors semiconductors

Quick Quiz! 1. Mendeleev arranged the elements in his periodic table in order of increasing _____ and by _____ properties. A. atomic number , similar B. number of protons , repeating C. number of electrons , similar D. atomic mass , repeating

Quick Quiz. 2. The modern periodic table is arranged by increasing. . A. mass number. B. density. C. atomic number. D. intelligence.

Quick Quiz. 3. Elements in the same group have… A. very little in common. B. the same atomic number. C. different chemical properties. D. similar chemical properties.

Quick Quiz. 4. Which one of the following is NOT a general property of metals? A. ductility B. malleability C. high luster D. poor conductor of heat and electricity

Quick Quiz. 5. Which list of elements contains only nonmetals? A. Sn , P , Cl B. S , Na , O C. Ge , Br , Ne D. O , I , H

Quick Quiz. 6. Identify each as metal, nonmetal, or metalloid: A. Copper: Metal B. Boron: Metalloid C. Sulfur: Nonmetal D. Sodium: Metal E. Tin: Metal F. Hydrogen: Nonmetal

Quick Quiz. 7. Identify the following sample of an unknown substance as a metal, nonmetal, or metalloid: solid at room temp. will break rather than bend conducts electricity only at high temp’s Metalloid
- Slides: 22