Development of Periodic Table Johann Dbereiner 1817 Law

Development of Periodic Table Johann Döbereiner (1817) Law of Triads Elements could be classified into groups of three, or triads. Trends in physical properties such as density, melting point, and atomic mass were observed. John Newlands (1863) Law of Octaves Arranged the 62 known elements into groups of seven according to increasing atomic mass. He proposed that an eighth element would then repeat the properties of the first element in the previous group.

Dimitri Mendeleev was a Russian chemist in the mid-1800 s. Mendeleev cataloged thousands of facts about the 63 elements known at the time. He became convinced that groups of elements had similar, "periodic" properties. Elements on Mendeleev's table were arranged according to their increasing atomic weight, leaving blank spaces where he was sure other, unknown elements would fit. Because of this, Mendeleev is considered to be the Father of the Periodic Table

Modern Periodic Table • Henry G. J. Moseley • Determined the atomic numbers of elements from their X-ray spectra (1914) • Arranged elements by increasing atomic number Periodic Law: The physical and chemical properties of the elements are periodic functions of their atomic numbers. 1887 - 1915

Glenn Seaborg: Present day periodic table • • After discovering U, Th, Pa, Seaborg was advised to revise the periodic table. • After examining the properties of the elements, Seaborg was convinced that the elements were part of the inner transition elements because they behaved similarly to these elements. • Instead of creating a new system, he integrated the elements into a new section called the actinide series, which is part of the inner transition elements.

Glenn T. Seaborg He is the only person to have an element named after him while still alive. "This is the greatest honor ever bestowed upon me - even better, I think, than winning the Nobel Prize. " 1912 - 1999
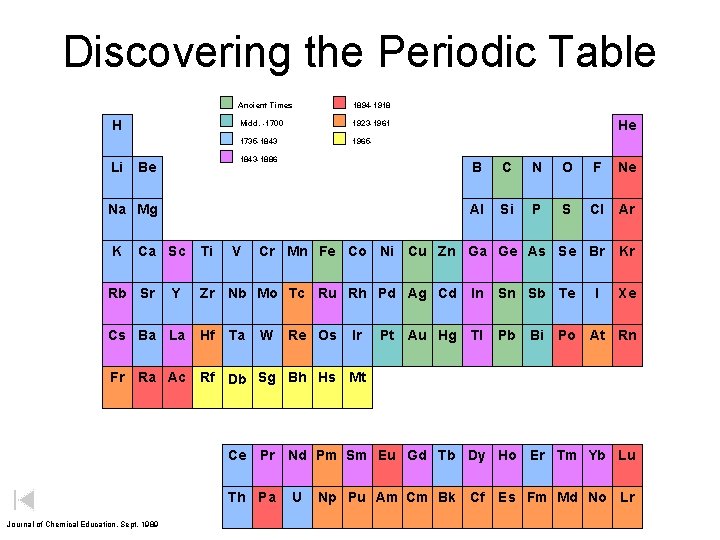
Discovering the Periodic Table H Li Ancient Times 1894 -1918 Midd. -1700 1923 -1961 1735 -1843 1965 - 1843 -1886 Be B C N O F Ne Al Si P S Cl Ar Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Na Mg K Ca Sc Rb Sr Y Cs Ba La Fr Ti V Zr Nb Mo Tc Ru Rh Pd Ag Cd Hf Ta He W Re Os Ir Pt Au Hg In Sn Sb Te Tl Pb Bi I Xe Po At Rn Ra Ac Rf Db Sg Bh Hs Mt Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa Journal of Chemical Education, Sept. 1989 U Np Pu Am Cm Bk Cf Es Fm Md No Lr

Introduction to the Periodic Table • Elements are arranged in seven horizontal rows, in order of increasing atomic number from left to right and from top to bottom. • Rows are called periods and are numbered from 1 to 7. • Elements with similar chemical properties form vertical columns, called groups, which are numbered from 1 to 18. • Groups 1, 2, and 13 through 18 are the main group elements. • Groups 3 through 12 are in the middle of the periodic table and are the transition elements. • The two rows of 14 elements at the bottom of the periodic are the lanthanides and actinides.
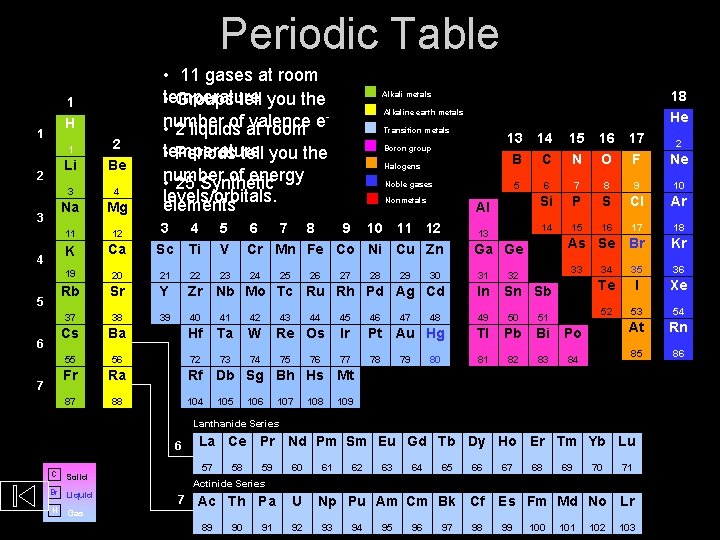
Periodic Table 1 H 1 Li 2 Be 3 4 Na Mg 11 12 3 K Ca 19 1 2 3 4 5 6 7 • 11 gases at room temperature • Groups tell you the number of at valence • 2 liquids room e 18 He Alkali metals Alkaline earth metals Transition metals • temperature Periods tell you the number of energy • 25 Synthetic levels/orbitals. elements Boron group Halogens 13 B 14 C 15 N 16 O 17 F Ne 5 6 7 8 9 10 Si P S Cl Ar 14 15 16 17 18 As Se Br Kr Noble gases Nonmetals Al Sc 4 Ti 5 V 6 7 8 9 10 11 12 Cr Mn Fe Co Ni Cu Zn 20 21 22 23 24 Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In 37 38 39 40 41 42 49 50 51 Cs Ba Hf Ta W 55 56 72 73 74 Fr Ra 87 88 25 26 43 27 44 Re Os 75 76 28 29 30 47 13 Ga Ge 31 48 33 32 Sn Sb 45 46 Ir Pt Au Hg Tl Pb Bi Po 77 78 81 82 83 84 79 80 34 35 36 Te I Xe 52 53 54 At Rn 85 86 Rf Db Sg Bh Hs Mt 104 105 106 107 108 109 Lanthanide Series 6 C Br Liquid H Gas La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu 57 Solid 58 59 60 61 62 63 64 65 66 67 68 69 70 71 Actinide Series 7 Ac Th Pa 89 90 91 U 92 Np Pu Am Cm Bk Cf 93 94 95 96 97 98 Es Fm Md No Lr 99 100 101 2 103
- Slides: 10