DEVELOPMENT OF LASER ABLATION FOR H 2 CONTENT

DEVELOPMENT OF LASER ABLATION FOR H 2 CONTENT MEASUREMENT IN METALS Irene Martini TE-VSC 1

Outline Motivation ü CLIC accelerating structures: static and dynamic pressure requirements ü Future plans: measurement of H 2 in thin films Study of Hydrogen Content measurement by laser ablation ü Experimental setup: vacuum and optical system ü Experimental sequence ü Limits: outgassing and dynamic hydrogen background ü Detection of electrons ü Trials ü Test on Copper ü Test on Titanium Conclusion TE-VSC 2
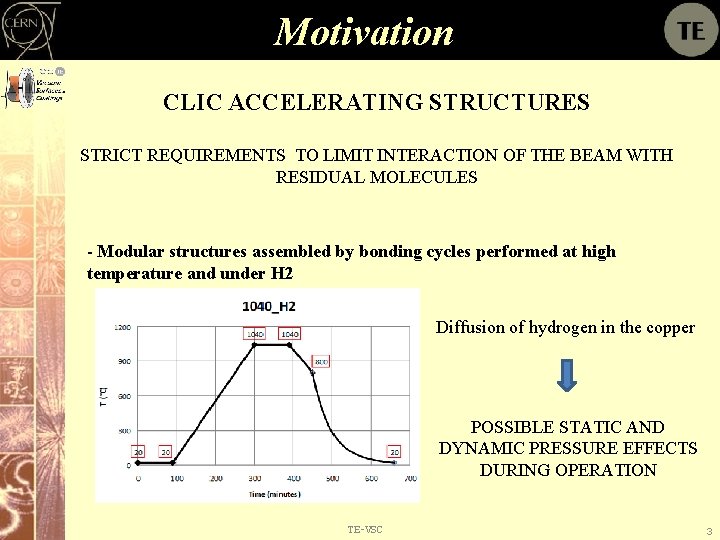
Motivation CLIC ACCELERATING STRUCTURES STRICT REQUIREMENTS TO LIMIT INTERACTION OF THE BEAM WITH RESIDUAL MOLECULES - Modular structures assembled by bonding cycles performed at high temperature and under H 2 Diffusion of hydrogen in the copper POSSIBLE STATIC AND DYNAMIC PRESSURE EFFECTS DURING OPERATION TE-VSC 3
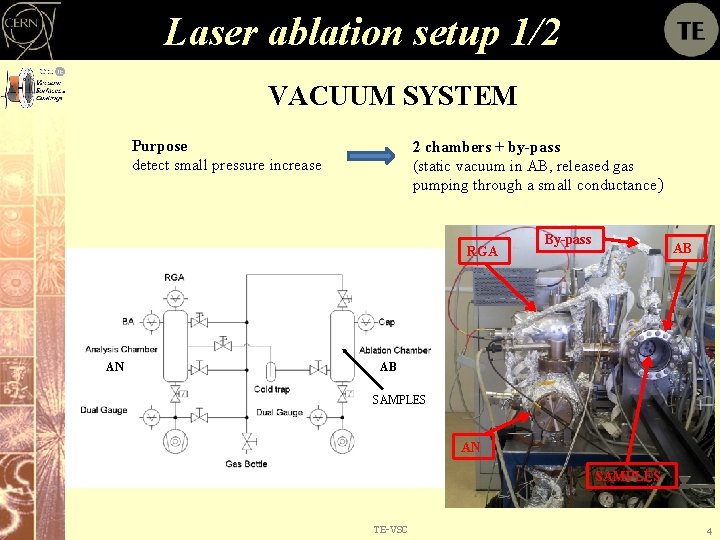
Laser ablation setup 1/2 VACUUM SYSTEM Purpose detect small pressure increase 2 chambers + by-pass (static vacuum in AB, released gas pumping through a small conductance) RGA AN By-pass AB AB SAMPLES AN SAMPLES TE-VSC 4
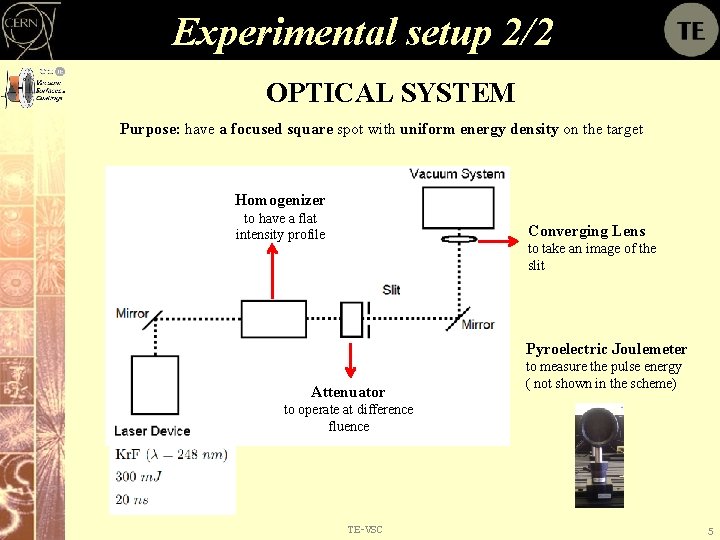
Experimental setup 2/2 OPTICAL SYSTEM Purpose: have a focused square spot with uniform energy density on the target Homogenizer to have a flat intensity profile Converging Lens to take an image of the slit Pyroelectric Joulemeter Attenuator to measure the pulse energy ( not shown in the scheme) to operate at difference fluence TE-VSC 5
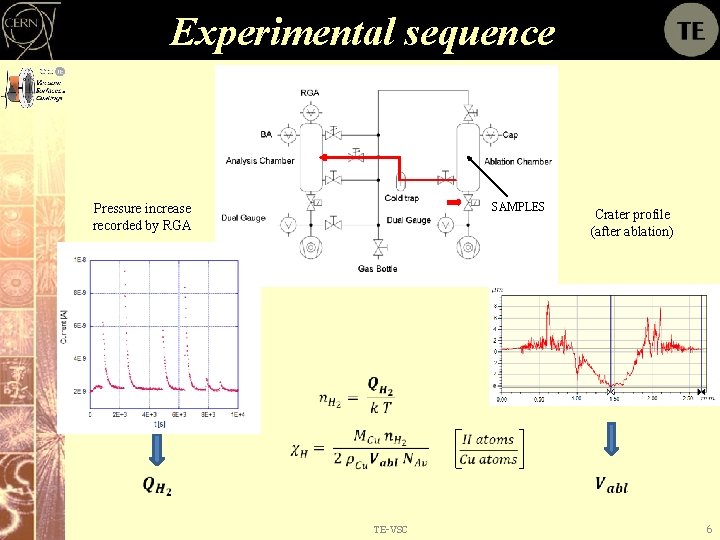
Experimental sequence SAMPLES Pressure increase recorded by RGA Crater profile (after ablation) TE-VSC 6
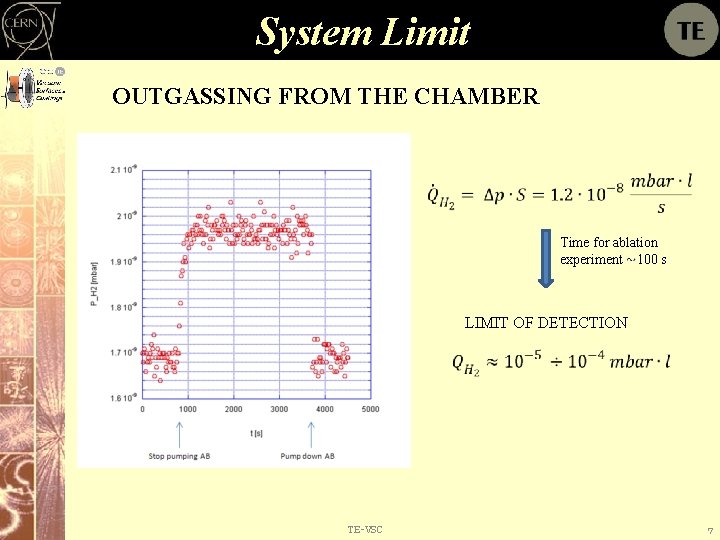
System Limit OUTGASSING FROM THE CHAMBER Time for ablation experiment 100 s LIMIT OF DETECTION TE-VSC 7

Dynamic H 2 Background Ablation on Silicon Expected: Measured: negligible Source of H 2 not related to actual content in the sample Hp: Desorption stimulated by photons and by charged particles (produce in laser-target interaction) ü Investigation of photodesorption (shot sample holder with unfocussed laser beam) : NO increase of H 2 partial pressure Desorption stimulated by charged particles Purpose: Investigate, remove or at least reduce the H 2 source ü Stop or slow down charged particle with ‘Buffer gas’ (Xe, CO 2) ü Trap charged particles with electric field TE-VSC 8
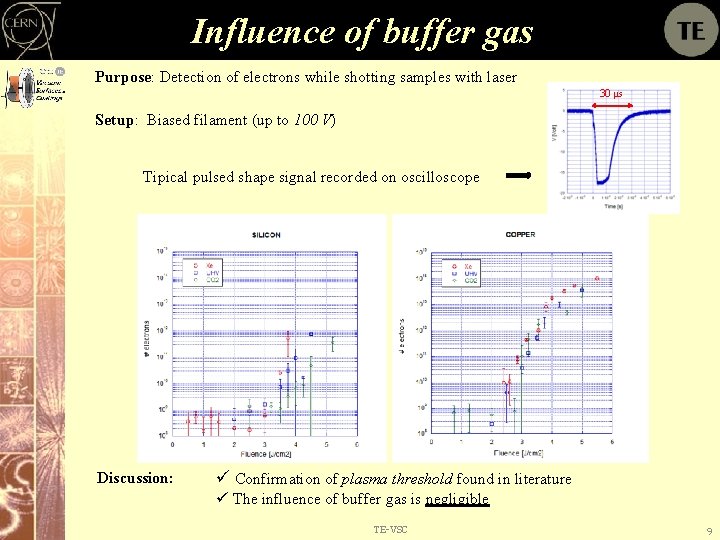
Influence of buffer gas Purpose: Detection of electrons while shotting samples with laser 30 μs Setup: Biased filament (up to 100 V) Tipical pulsed shape signal recorded on oscilloscope Discussion: ü Confirmation of plasma threshold found in literature ü The influence of buffer gas is negligible TE-VSC 9

Trials ü Investigation of photodesorption NO INFLUENCE on desorption signal üStop or slow down charged particles with ‘Buffer gas’ (Xe, CO 2) NO EFFECTIVE ü Attempt to detect ions in the same way of electrons MEASUREMENT NOT REPRODUCIBLE ü Trap charged particles (positive and negative bias on the sample holder) same desorption signal as grounded sample holder configuration The H 2 source is STIMULATED DESORPTION from the wall of the chamber The attempts done to reduce it were unsuccessful TE-VSC 10
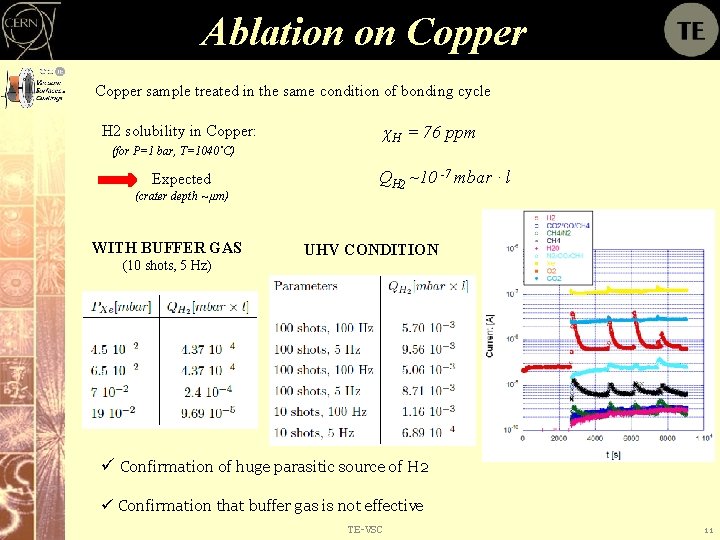
Ablation on Copper sample treated in the same condition of bonding cycle H 2 solubility in Copper: χH = 76 ppm (for P=1 bar, T=1040˚C) Expected (crater depth ~ μm) WITH BUFFER GAS (10 shots, 5 Hz) QH 2 ~10 -7 mbar · l UHV CONDITION ü Confirmation of huge parasitic source of H 2 ü Confirmation that buffer gas is not effective TE-VSC 11

Ablation on Titanium Test on treated and untreated titanium samples to compare the results. χH ~ 4 · 10 4 ppm H 2 solubility in Titanium: (for P=25 mbar, T=1020˚C) Expected QH 2 ~10 -4 mbar · l (crater depth ~ μm) TREATED TITANIUM UNTREATED TITANIUM ü No measured difference between H 2 release from treated sample and untreated sample TE-VSC 12

Conclusion Limits: ü Outgassing of the chamber ü Dynamic hydrogen pressure Attempts: üInvestigation of photodesorption ü Buffer gas (Xe, CO 2) ü Bias on the sample holder to trap charged particles üTest on Titanium (higher content) The method is not applicable due to the induced desorption from the wall TE-VSC 13

TE-VSC 14

TE-VSC 15
- Slides: 15