Development of a Physiologically Based Pharmacokinetic and Pharmacodynamic
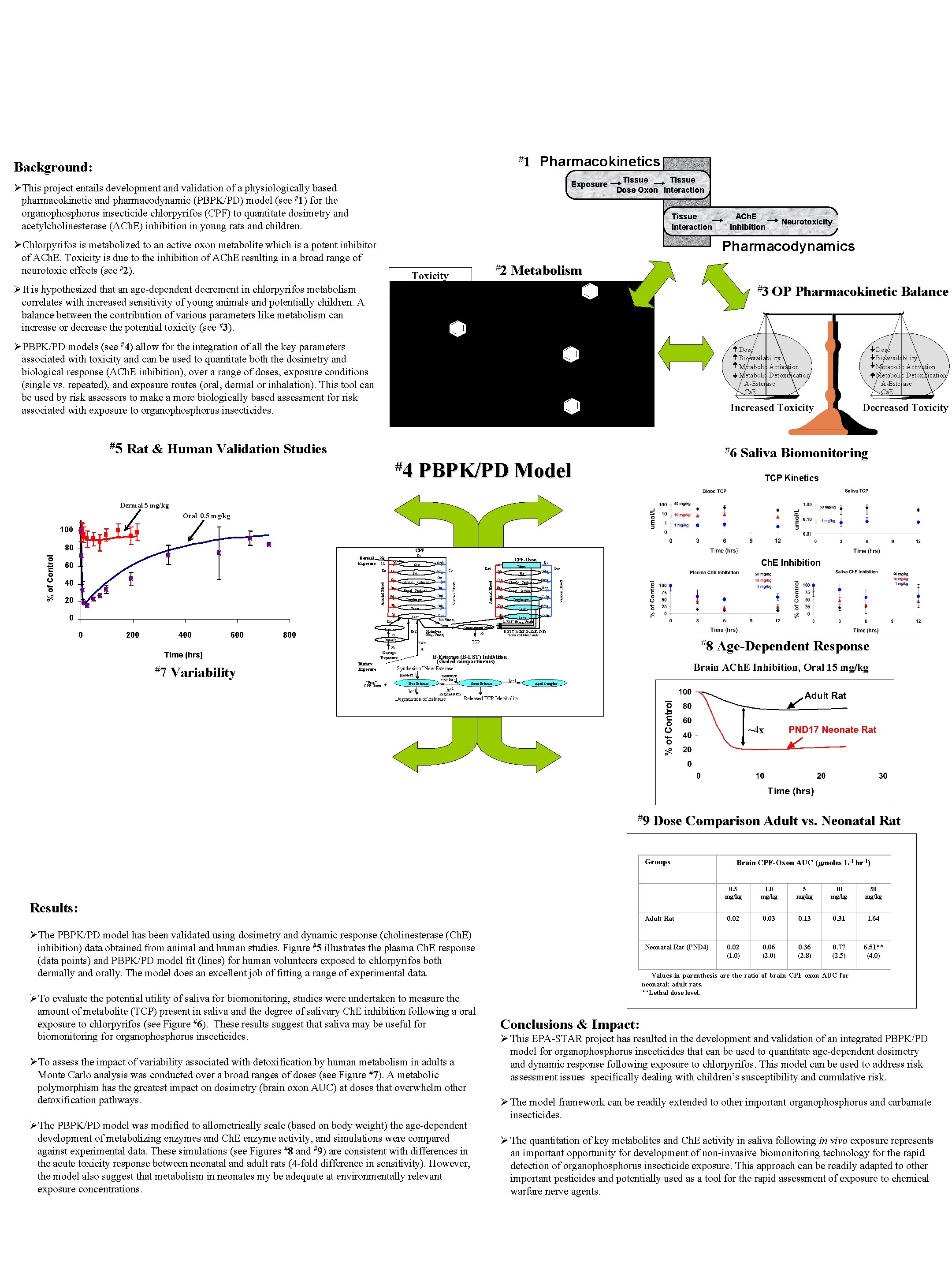
Development of a Physiologically Based Pharmacokinetic and Pharmacodynamic Model to Quantitate Biomarkers of Exposure to Organophosphorus Insecticides Charles Timchalk, Ahmed Kousba and Torka S. Poet Battelle, Pacific Northwest Division, Richland WA Pharmacokinetics #1 Background: Exposure ØThis project entails development and validation of a physiologically based pharmacokinetic and pharmacodynamic (PBPK/PD) model (see #1) for the organophosphorus insecticide chlorpyrifos (CPF) to quantitate dosimetry and acetylcholinesterase (ACh. E) inhibition in young rats and children. Tissue Dose Oxon Interaction Tissue Interaction Neurotoxicity Pharmacodynamics ØChlorpyrifos is metabolized to an active oxon metabolite which is a potent inhibitor of ACh. E. Toxicity is due to the inhibition of ACh. E resulting in a broad range of neurotoxic effects (see #2). #2 Toxicity ACh. E Inhibition ØIt is hypothesized that an age-dependent decrement in chlorpyrifos metabolism correlates with increased sensitivity of young animals and potentially children. A balance between the contribution of various parameters like metabolism can increase or decrease the potential toxicity (see #3). Metabolism #3 OP ØPBPK/PD models (see #4) allow for the integration of all the key parameters associated with toxicity and can be used to quantitate both the dosimetry and biological response (ACh. E inhibition), over a range of doses, exposure conditions (single vs. repeated), and exposure routes (oral, dermal or inhalation). This tool can be used by risk assessors to make a more biologically based assessment for risk associated with exposure to organophosphorus insecticides. #5 ACh. E Inhibition Pharmacokinetic Balance Dose Bioavailability Metabolic Activation Metabolic Detoxification A-Esterase Ca. E Increased Toxicity Rat & Human Validation Studies #4 #6 PBPK/PD Model Decreased Toxicity Saliva Biomonitoring TCP Kinetics Dermal 5 mg/kg Oral 0. 5 mg/kg 100 CPF Ca Qsk Qf 20 Cvs Slowly Perfused Qr Rapid Perfused Diaphragm Qb Cvb Brain Ql 200 400 600 Oxon Intestine 800 Ka. S Ks. I Stomach #7 Variability Dietary Exposure Cvso Slowly Perfused Qro Cvro Rapid Perfused Qdo Cvdo Diaphragm Qbo Cvbo Brain Liver Km 3, 4, Vmax 3, 4 Cvlo Compartment Model Hydrolysis Km 2, Vmax 2 Ke B-EST (ACh. E, Bu. Ch. E, Ca. E) * Liver and blood only. #8 TCP B-Esterase (B-EST) Inhibition (shaded compartments) Free Esterase Inhibition u. M -1 hr -1 Oxon-Esterase Age-Dependent Response Brain ACh. E Inhibition, Oral 15 mg/kg Synthesis of New Esterase mole hr -1 “Free” CPF-Oxon + Qso Cvo Fa Gavage Exposure Time (hrs) Cvfo Fat A-EST* Kzero Fa Qfo Ch. E Inhibition Qc Blood Qlo Cvl Km 1 Vmax , 1 Liver Ka. I 0 Cvr Cvd Qd 0 Cao Cv Cvf Fat Qs 40 CPF-Oxon Cvsk Skin Venous Blood SA Arterial Blood 60 Qc Venous Blood Dermal Exposure Kp Arterial Blood % of Control 80 hr-1 Aged Complex hr-1 Regeneration Released TCP Metabolite Degradation of Esterase ~4 x #9 Dose Comparison Adult vs. Neonatal Rat Groups Results: ØThe PBPK/PD model has been validated using dosimetry and dynamic response (cholinesterase (Ch. E) inhibition) data obtained from animal and human studies. Figure #5 illustrates the plasma Ch. E response (data points) and PBPK/PD model fit (lines) for human volunteers exposed to chlorpyrifos both dermally and orally. The model does an excellent job of fitting a range of experimental data. ØTo evaluate the potential utility of saliva for biomonitoring, studies were undertaken to measure the amount of metabolite (TCP) present in saliva and the degree of salivary Ch. E inhibition following a oral exposure to chlorpyrifos (see Figure #6). These results suggest that saliva may be useful for biomonitoring for organophosphorus insecticides. ØTo assess the impact of variability associated with detoxification by human metabolism in adults a Monte Carlo analysis was conducted over a broad ranges of doses (see Figure #7). A metabolic polymorphism has the greatest impact on dosimetry (brain oxon AUC) at doses that overwhelm other detoxification pathways. ØThe PBPK/PD model was modified to allometrically scale (based on body weight) the age-dependent development of metabolizing enzymes and Ch. E enzyme activity, and simulations were compared against experimental data. These simulations (see Figures #8 and #9) are consistent with differences in the acute toxicity response between neonatal and adult rats (4 -fold difference in sensitivity). However, the model also suggest that metabolism in neonates my be adequate at environmentally relevant exposure concentrations. Brain CPF-Oxon AUC ( moles L-1 hr-1) 0. 5 mg/kg 1. 0 mg/kg 5 mg/kg 10 mg/kg 50 mg/kg Adult Rat 0. 02 0. 03 0. 13 0. 31 1. 64 Neonatal Rat (PND 4) 0. 02 (1. 0) 0. 06 (2. 0) 0. 36 (2. 8) 0. 77 (2. 5) 6. 51** (4. 0) Values in parenthesis are the ratio of brain CPF-oxon AUC for neonatal: adult rats. **Lethal dose level. Conclusions & Impact: Ø This EPA-STAR project has resulted in the development and validation of an integrated PBPK/PD model for organophosphorus insecticides that can be used to quantitate age-dependent dosimetry and dynamic response following exposure to chlorpyrifos. This model can be used to address risk assessment issues specifically dealing with children’s susceptibility and cumulative risk. Ø The model framework can be readily extended to other important organophosphorus and carbamate insecticides. Ø The quantitation of key metabolites and Ch. E activity in saliva following in vivo exposure represents an important opportunity for development of non-invasive biomonitoring technology for the rapid detection of organophosphorus insecticide exposure. This approach can be readily adapted to other important pesticides and potentially used as a tool for the rapid assessment of exposure to chemical warfare nerve agents.
- Slides: 1