Development of a Novel Intranasal Physiologically Based Absorption

Development of a Novel Intranasal Physiologically Based Absorption Model and Its Application in ARS-1 Epinephrine Nasal Spray Development to Enhance Understanding of Absorption in Neffy. TM Single and Repeated Dose (L/R) in Healthy Adults Chaejin Kim 1, Jeffrey S. Barrett 2, Richard Lowenthal 3, Sarina Tanimoto 3, Stephan Schmidt 1, Valvanera Vozmediano 1 1. Center for Pharmacometrics and Systems Pharmacology, Department of Pharmaceutics, College of Pharmacy, University of Florida, Orlando, FL, 2. Critical Path Institute, Tucson, AZ 3. ARS Pharmaceuticals, San Diego, CA Center for Pharmacometrics and Systems Pharmacology Department of Pharmaceutics University of Florida Dr. Vozmediano’s Lab Presentation by Chaejin Kim, Funded by ARS Pharmaceuticals. Pharm. D. MPH
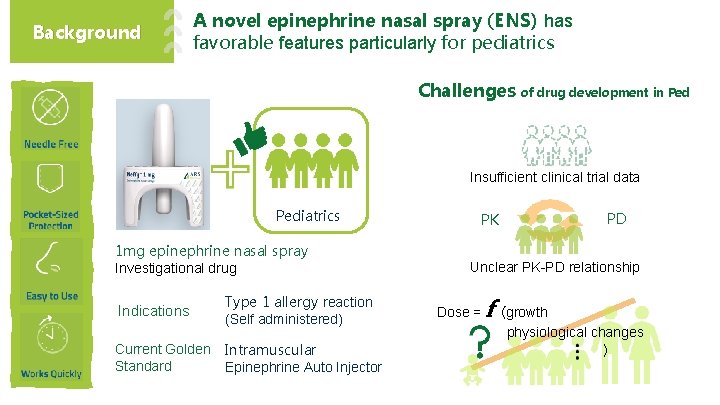
Background A novel epinephrine nasal spray (ENS) has favorable features particularly for pediatrics Challenges of drug development in Ped Insufficient clinical trial data Pediatrics 1 mg epinephrine nasal spray Investigational drug Indications Type 1 allergy reaction (Self administered) Current Golden Standard Intramuscular Epinephrine Auto Injector PK PD Unclear PK-PD relationship Dose = f (growth physiological changes )
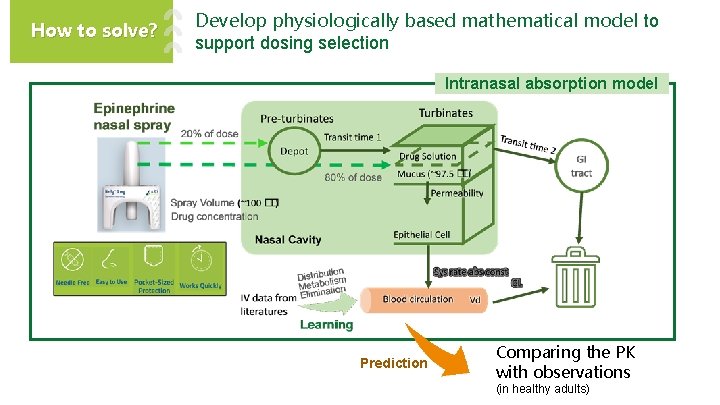
How to solve? Develop physiologically based mathematical model to support dosing selection Intranasal absorption model Prediction Comparing the PK with observations (in healthy adults)
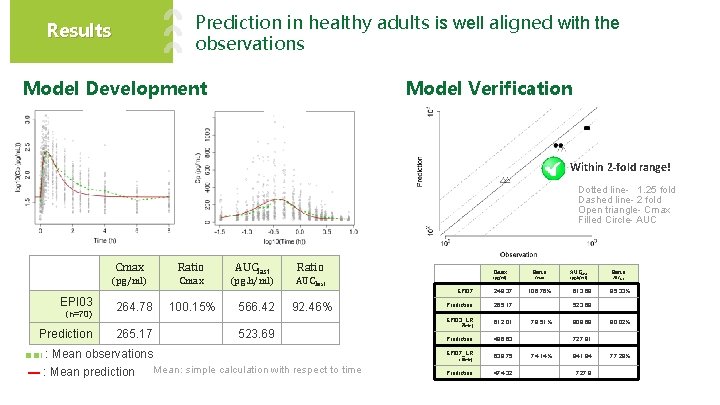
Prediction in healthy adults is well aligned with the observations Results Model Verification Model Development Within 2 -fold range! Dotted line- 1. 25 fold Dashed line- 2 fold Open triangle- Cmax Filled Circle- AUC EPI 03 (n=70) Prediction Cmax Ratio AUClast Ratio (pg/ml) Cmax (pg. h/ml) AUClast 264. 78 265. 17 100. 15% 566. 42 92. 46% 523. 69 : Mean observations ▬ : Mean prediction Ratio (pg/ml) Cmax EPI 07 248. 37 Prediction 265. 17 EPI 03_LR 612. 01 (5 min) Prediction 486. 63 EPI 07_LR 639. 75 (10 min) Mean: simple calculation with respect to time Cmax Prediction 474. 32 106. 76% AUClast (pg. h/ml) 613. 69 Ratio AUClast 85. 33% 523. 69 79. 51% 909. 68 80. 02% 727. 91 74. 14% 941. 84 727. 9 77. 28%
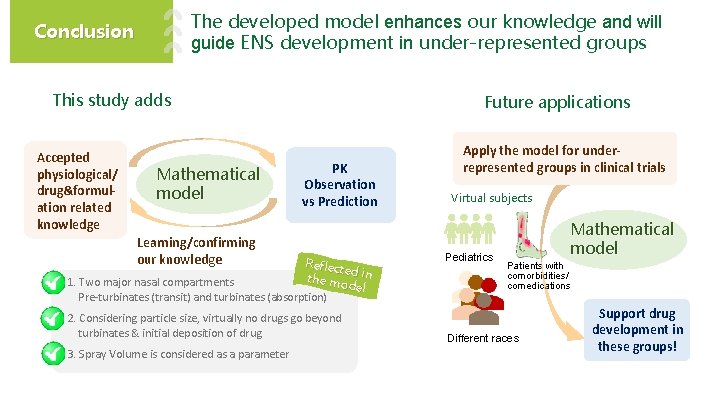
The developed model enhances our knowledge and will guide ENS development in under-represented groups Conclusion This study adds Accepted physiological/ drug&formulation related knowledge Mathematical model Future applications PK Observation vs Prediction Learning/confirming our knowledge Reflect ed the mo in 1. Two major nasal compartments del Pre-turbinates (transit) and turbinates (absorption) 2. Considering particle size, virtually no drugs go beyond turbinates & initial deposition of drug 3. Spray Volume is considered as a parameter Apply the model for underrepresented groups in clinical trials Virtual subjects Pediatrics Mathematical model Patients with comorbidities/ comedications Different races Support drug development in these groups!

Right treatment Right time For right person Thank you!
- Slides: 6