Development and Application of Theoreticallybased EOS on Phase

Development and Application of Theoretically-based EOS on Phase Equilibrium Calculations Principle Investigator : Yan-Ping Chen Research Group : Min-Long Yu Chyau-Song Wu Jung-Chin Tsai Department of Chemical Engineering National Taiwan University

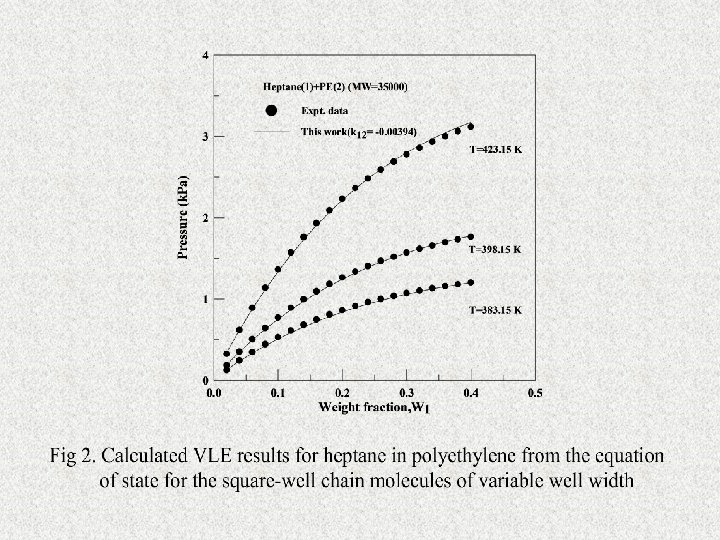
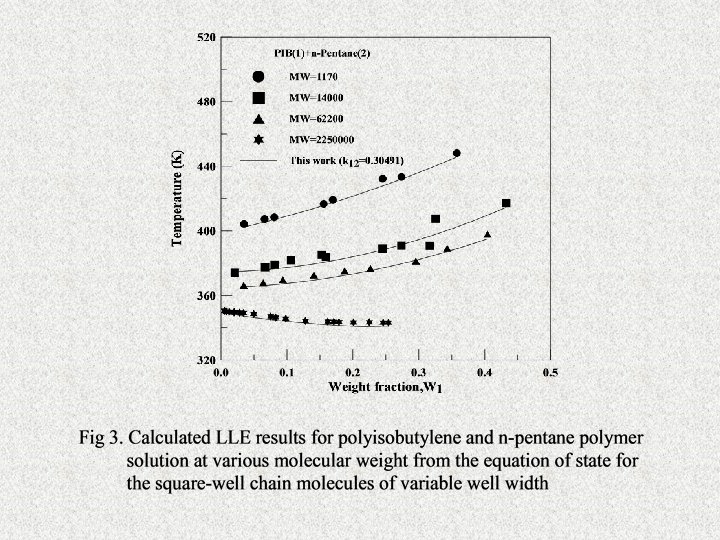
Research Topics FCorrelation of Liquid-liquid Phase Equilibria Using the SAFT Equation of State FCalculations of Vapor-Liquid Equation of State of Polymer Solutions Using SAFT Equation of State FCalculation and Prediction of VLE of Polymer Solutions by the GFD Equation of State FDevelopment and Application of an Equation of State for the Square-Well Chain Molecules of Variable Well Width Based on a Modified Coordination Number Model


Correlation of Solid Solubility for Heavy Components in Supercritical Fluids Principle Investigator: Yan-Ping Chen Research Group: Ping-Chin Chen Chung-Chia Huang Jaw-Shin Cheng Department of Chemical Engineering National Taiwan University

Research Topics Cubic Type Equation of State Plus Excess Volume Model Cubic EOS + GE Model Huron-Vidal type mixing rule coupled with the volume correction term (b. E) is applied Solution Model Approach Modified regular solution model coupled with the Flory. Huggins equation is also applied
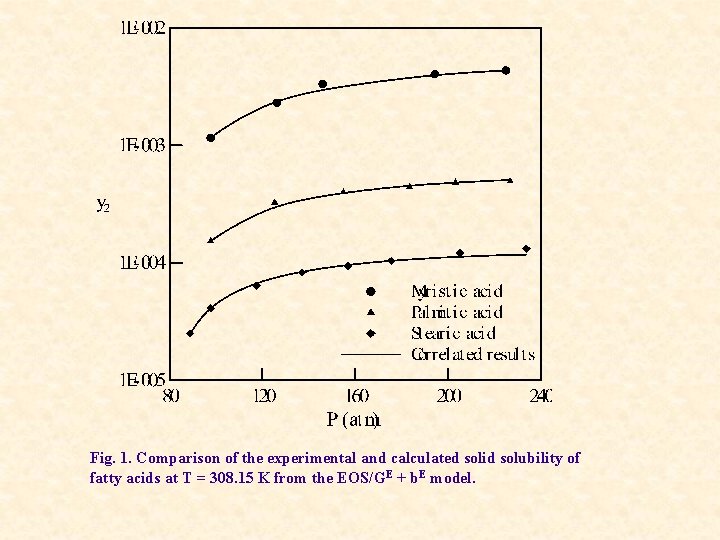
Fig. 1. Comparison of the experimental and calculated solid solubility of fatty acids at T = 308. 15 K from the EOS/GE + b. E model.
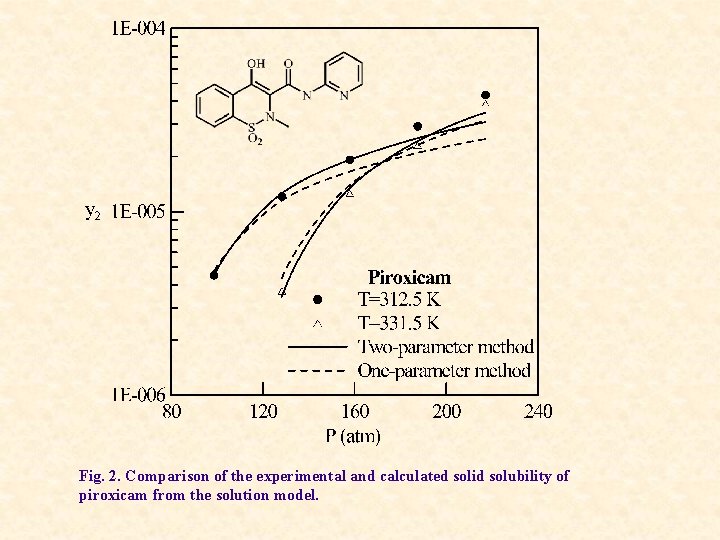
Fig. 2. Comparison of the experimental and calculated solid solubility of piroxicam from the solution model.

Experimental Measurements of Phase Equilibrium at High Pressures Principle Investigator: Yan-Ping Chen Research Worker: Kong - Wei Cheng-Shi Cheng Tz-Bang Du Department of Chemical Engineering National Taiwan University

Research Topics Vapor - Liquid Equilibria of of Binary and Ternary Mixtures in Carbon dioxide at Elevated Pressures Solubility of Solids in Supercritical Carbon dioxide Synthesis Conductive Polymer Composites in Supercritical Carbon dioxide
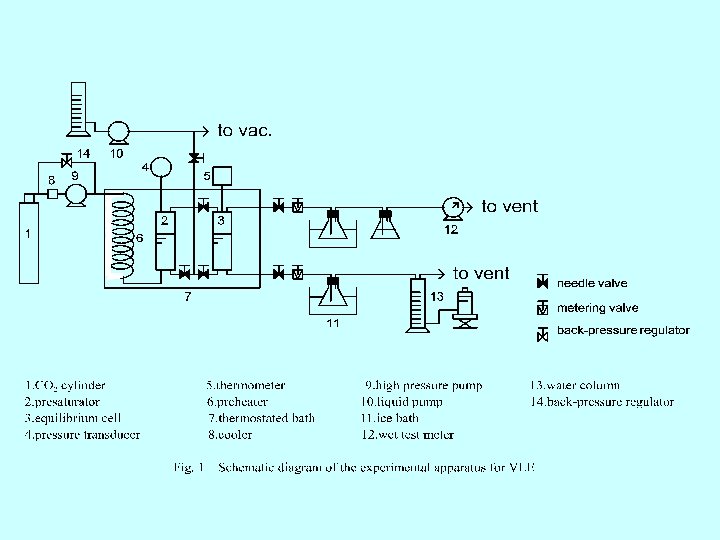
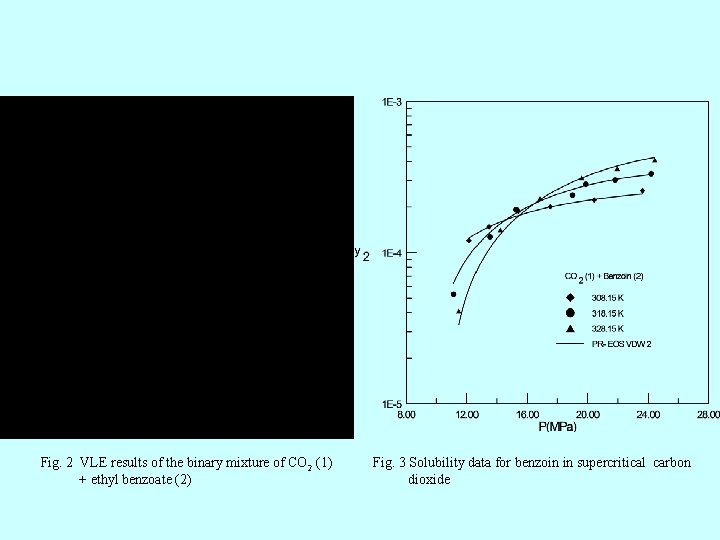
Fig. 2 VLE results of the binary mixture of CO 2 (1) + ethyl benzoate (2) Fig. 3 Solubility data for benzoin in supercritical carbon dioxide
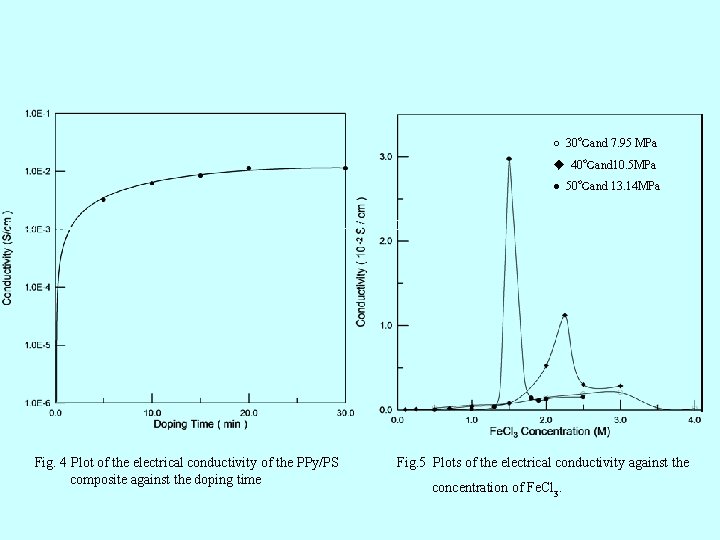
○ 30℃and 7. 95 MPa ◆ 40℃and 10. 5 MPa ● 50℃and 13. 14 MPa Fig. 6 Plots of the electrical conductivity against the concentration of Fe. Cl 3. Fig. 4 Plot of the electrical conductivity of the PPy/PS composite against the doping time Fig. 5 Plots of the electrical conductivity against the concentration of Fe. Cl 3.

Correlation of Mutual Diffusion Coefficients of Binary Liquid Mixtures Principle Investigator : Yan-Ping Chen Research Group : Yu-Du Hsu Department of Chemical Engineering National Taiwan University

Research Topics l. Correlation of liquid binary mutual diffusion coefficients by using a local composition model (UNIDIF) l. Binary interaction parameters of nonpolar and polar systems are optimally regressed l. The overall AAD in 1042 data points is 2. 3% l. Extension to group contribution correlation also indicates satisfactory results with AAD of 3. 6%
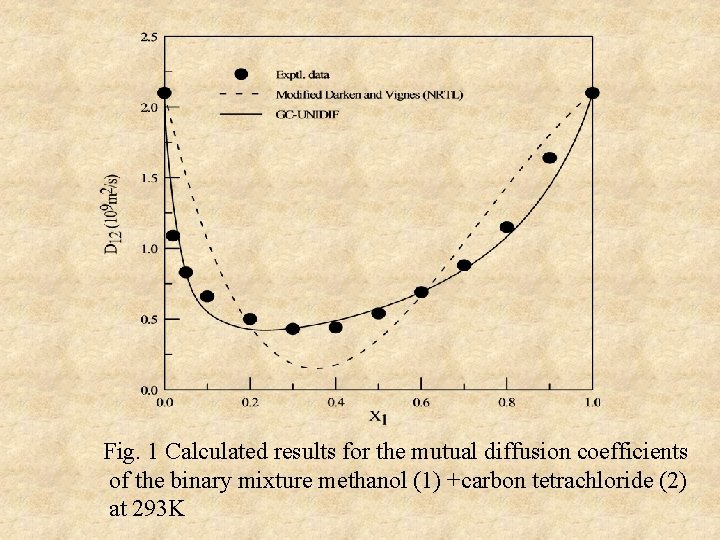
Fig. 1 Calculated results for the mutual diffusion coefficients of the binary mixture methanol (1) +carbon tetrachloride (2) at 293 K
- Slides: 17