Developing fuels DF 3 Petrol is popular Petrol






- Slides: 11

Developing fuels DF 3 Petrol is popular

Petrol and Crude Oil n n Petrol is a mixture of many different compounds blended to give the right properties 30% - 40% of each barrel of crude oil goes to make petrol Thick black liquid with gases and solids dissolved inside Crude oil is a mixture of hydrocarbons (molecules made of a chemical combination of carbon and hydrogen atoms)

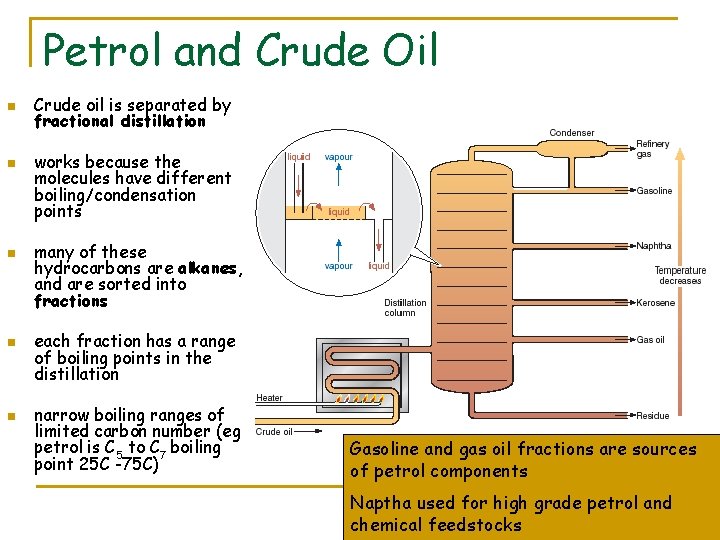
Petrol and Crude Oil n n n Crude oil is separated by fractional distillation works because the molecules have different boiling/condensation points many of these hydrocarbons are alkanes, and are sorted into fractions each fraction has a range of boiling points in the distillation narrow boiling ranges of limited carbon number (eg petrol is C 5 to C 7 boiling point 25 C -75 C) Gasoline and gas oil fractions are sources of petrol components Naptha used for high grade petrol and chemical feedstocks

Petrol and Crude Oil

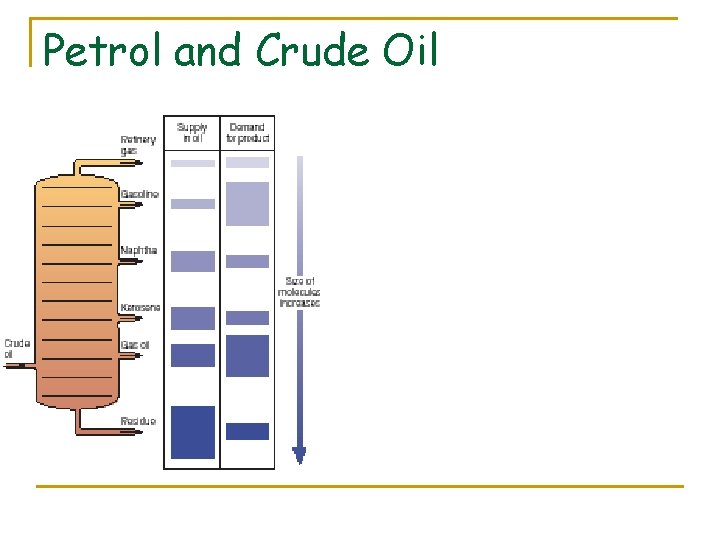
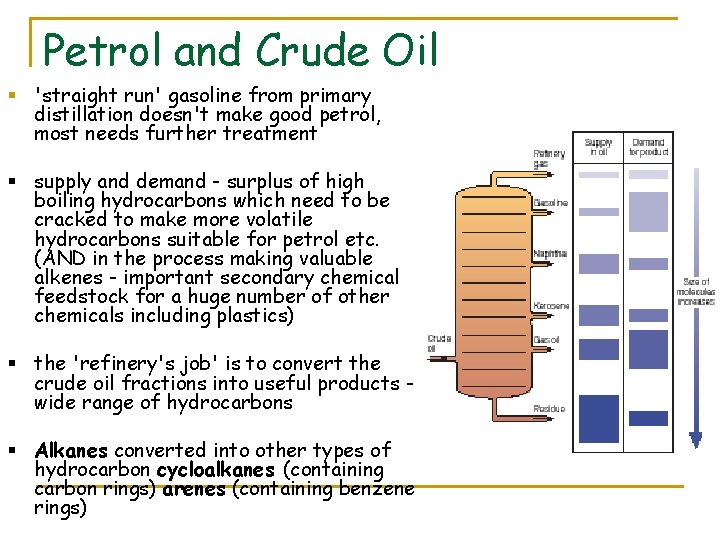
Petrol and Crude Oil § 'straight run' gasoline from primary distillation doesn't make good petrol, most needs further treatment § supply and demand - surplus of high boiling hydrocarbons which need to be cracked to make more volatile hydrocarbons suitable for petrol etc. (AND in the process making valuable alkenes - important secondary chemical feedstock for a huge number of other chemicals including plastics) § the 'refinery's job' is to convert the crude oil fractions into useful products wide range of hydrocarbons § Alkanes converted into other types of hydrocarbon cycloalkanes (containing carbon rings) arenes (containing benzene rings)

What’s left over? n After distillation there is a residue left over n Can be used to make useful products n First is distilled again under reduced pressure n Vacuum distillation avoids high temps that would be needed at atmospheric pressure (which would crack the hydrocarbons) n More volatile oils distils n Oils are used as fuel oils in power stations or ships n Others are used as base for lubricating oils

Winter and summer petrol n n n Not as simple as sending straight run gasoline to the pump. Petrol has to be blended to get the right properties – Volatility In a car engine mixture of petrol vapour and air is ignited When weather is cold petrol is difficult to vaporise – car difficult to start How do petrol blenders solve the problem?

Winter and summer petrol n n Petrol companies make different blends for different times of year Winter – more volatile compounds vaporise more easily, more small molecules such as butane and pentane Summer – in hot weather you don’t want too many volatile compounds – petrol would vaporise to easily and you would lose petrol from the tank – costly and polluting Different blend for different countries How would petrol blends differ in Russia and Egypt ?

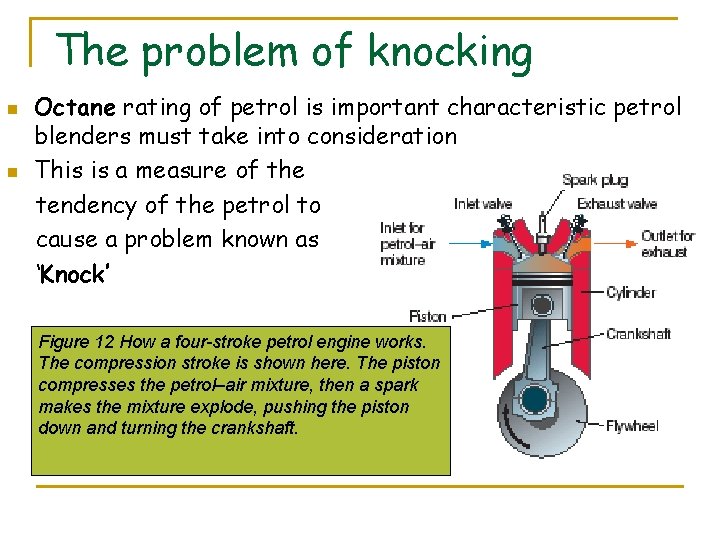
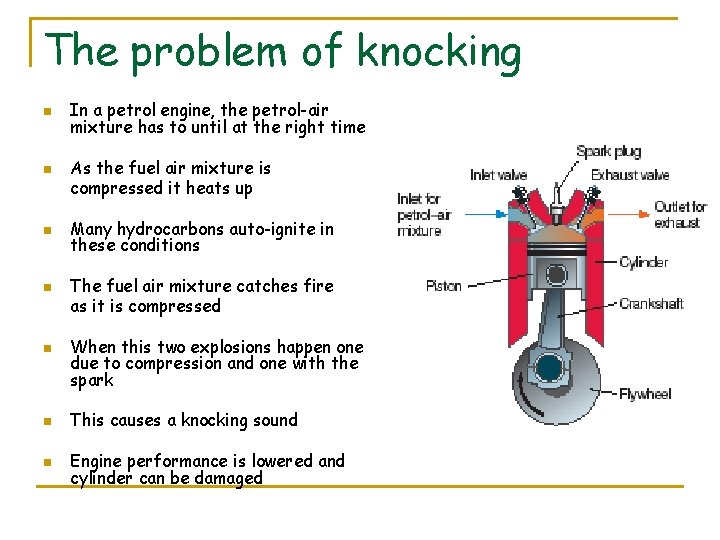
The problem of knocking n n Octane rating of petrol is important characteristic petrol blenders must take into consideration This is a measure of the tendency of the petrol to cause a problem known as ‘Knock’ Figure 12 How a four-stroke petrol engine works. The compression stroke is shown here. The piston compresses the petrol–air mixture, then a spark makes the mixture explode, pushing the piston down and turning the crankshaft.

Octane number n n n The tendency of a fuel to auto ignite is measured by its octane number. 2, 2, 4 – trimethylpentane is a branched alkane with a low tendency to auto-ignite – given an octane number of 100 Heptane- straight chain alkane - auto-ignites easily and is given an octane number of 0 The octane number of any fuel is the percentage of 2, 2, 4 – trimethylpentane in a mixture of 2, 2, 4 – trimethylpentane and heptane which knocks at the same compression ratio as a given fuel For example 4 -star petrol has an octane number of 97 and knocks at the same compression ratio as a mixture of 97% 2, 2, 4 – trimethylpentane and 3% heptane CH 3 l l CH 3 -C-CH 2 -CH-CH 3 l CH 3 2, 2, 4 – trimethylpentane Low tendency to auto ignite scores 100 CH 3 -CH 2 -CH 2 -CH 3 Heptane High tendency to auto ignite scores 0

The problem of knocking n n n In a petrol engine, the petrol-air mixture has to until at the right time As the fuel air mixture is compressed it heats up Many hydrocarbons auto-ignite in these conditions The fuel air mixture catches fire as it is compressed When this two explosions happen one due to compression and one with the spark n This causes a knocking sound n Engine performance is lowered and cylinder can be damaged