Developing and Sustaining Community and SchoolBased Immunization Programs

Developing and Sustaining Community and School-Based Immunization Programs for School Age Children Pedro A. Piedra, M. D. Baylor College of Medicine Houston, Texas

Texas Experience With Mass Vaccination of Children Against Influenza-Associated Morbidity -1 1998 -2001 study years 1: 15% coverage of children 1. 5 -18 years with a single dose of an influenza vaccine averted 8 -18% or 400 -1000 medicallyattended influenza related illnesses in adults >35 years of age. 2003 -04 study year 2: 30% coverage of children 5 -18 years with a single dose of an influenza vaccine provided immediate, direct and indirect protection in children even when administered during an influenza outbreak and when the dominant circulating virus was antigenically distinct from the vaccine strain. 1: Piedra et al. Vaccine 2005; 23: 1540. 2: Piedra et al. Pediatrics 2007; 120: e 553134: 71.

Texas Experience With Mass Vaccination of Children Against Influenza-Associated Morbidity -2 2004 -06 study years 1: Approximately 30% coverage of school-aged children with a single dose of an influenza vaccine resulted in broadening herd protection to adults and children averting 4 -18% of medically-attended influenza related illnesses. 2005 -06 study year 2: Approximately 30% coverage of school-aged children may have prevented the second influenza wave (influenza B) from developing in the intervention cities. 1: Piedra et al. 9 th Annual Conference on Vaccine Research, May 8, 2006 in Baltimore, MD 2: Piedra et al. 2007 Pediatric Academic Societies, on May 8, 2007 in Toronto, Canada (publication no. 8755. 1). .
Site of Field Trial, Central Texas

Study Goals To control seasonal influenza through active immunization of school-aged children with live attenuated (LAIV-T) and inactivated influenza (IIV-T) vaccines. To determine the proportion of children who should be vaccinated to reduce the incidence of medically attended acute respiratory illnesses (MAARI) among all age groups during the influenza epidemic.

Study Design Community-based, non-randomized, openlabel trial in children Intervention cities (Temple, Belton and surrounding towns): LAIV-T and IIV-T were offered to children Comparison cities (Waco and surrounding towns, Bryan & College Station): LAIV-T and IIVT were not offered through the study

Enrollment Base line year (1997 -98) Clinic-based: four year trial (1998 -02): 1. 5 to <19 years Base line year (2002 -03) Community-based: three year trial (200306): 5 to <19 years Base line year (2006 -07) School-based: year 1 (2007 -08): 4 to <19 years and school staff

Vaccination Sites in Intervention Communities: 1998 -2007 Sites 98 99 00 01 03 04 05 07 S&W clinic ++++ +++ ++ ++ + Mall ND ND ++ ++ ++ ND Belton clinic + + + ND ND ND Outreach ND ND ND + + ++ ++ + School clinic ND ND ++++ ND: not done +: <25%; ++: 25<50%; +++: 50<75%; ++++: 75 -100%


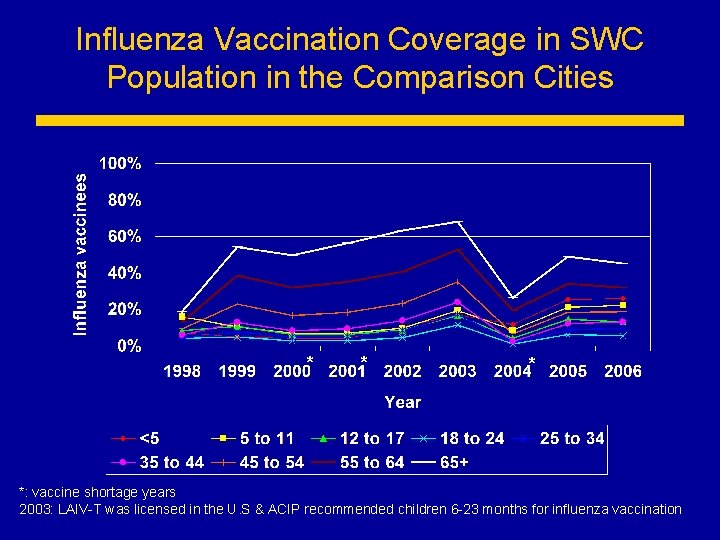
Influenza Vaccination Coverage in SWC Population in the Comparison Cities * *: vaccine shortage years 2003: LAIV-T was licensed in the U. S & ACIP recommended children 6 -23 months for influenza vaccination
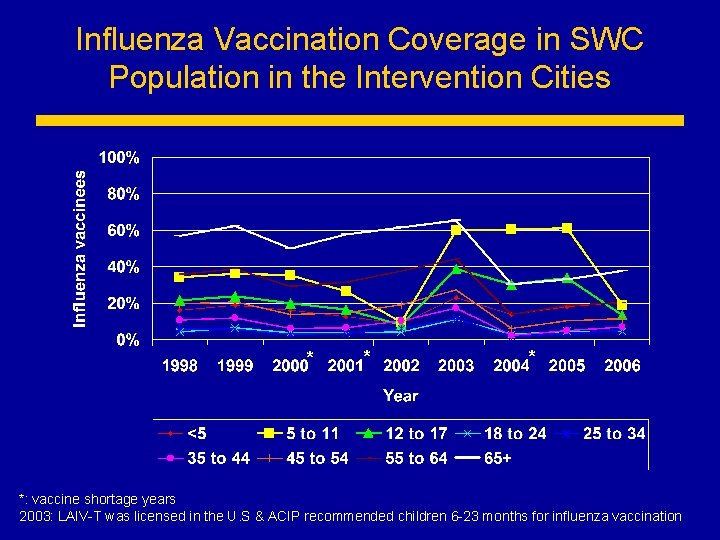
Influenza Vaccination Coverage in SWC Population in the Intervention Cities * *: vaccine shortage years 2003: LAIV-T was licensed in the U. S & ACIP recommended children 6 -23 months for influenza vaccination
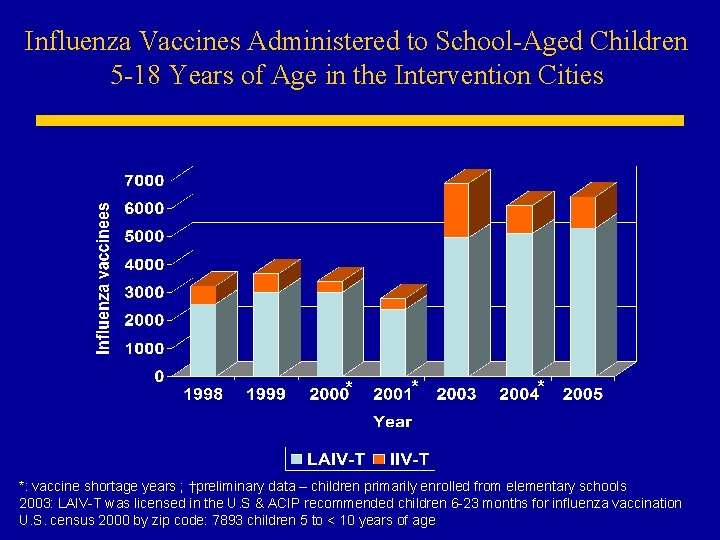
Influenza Vaccines Administered to School-Aged Children 5 -18 Years of Age in the Intervention Cities * *: vaccine shortage years ; †preliminary data – children primarily enrolled from elementary schools 2003: LAIV-T was licensed in the U. S & ACIP recommended children 6 -23 months for influenza vaccination U. S. census 2000 by zip code: 7893 children 5 to < 10 years of age

Models for Universal Influenza Vaccination of Children Private provider-based model Medical home Major health care providers in the community make available influenza clinics Public health-based model Medical home County or city health department make available influenza clinics Private and public health provider model Coalition between private and public health care providers for operating influenza clinics School-based model

Coalition of Interested Parties Conducted the Schoolbased Influenza Vaccination Program in Central Texas Independent School Districts Bell County Health Department Scott & White Clinic University of Mary-Hardin-Baylor School of Nursing Baylor College of Medicine

Ingredients of a School-based Influenza Vaccination Program Community awareness on the burden of influenza Trust and acceptance by schools and parents Support by public and private health care providers Offer influenza vaccines to all persons in schools Children Staff Stream line the consent/assent process Monitor school outcomes Student absenteeism Influenza like illness

Mission Statement Annual school-based influenza vaccination of schoolchildren will improve the health and wellness in schools, maximize school attendance, and reduce the burden of influenza in our communities. Curtailing influenza will also improve student learning, increase the allocation of funds from State and Federal sources, and decrease household employment absenteeism.




Year 1 of the School-based Influenza Vaccination Program Elementary schools 7 Independent school districts: 25 elementary schools 3 parochial schools School clinic: ½ day/school Vaccinated 5144 students (~45%): 4386 LAIV, 758 TIV 887 school staff: 563 LAIV, 322 TIV 996 other children through outreach & clinic
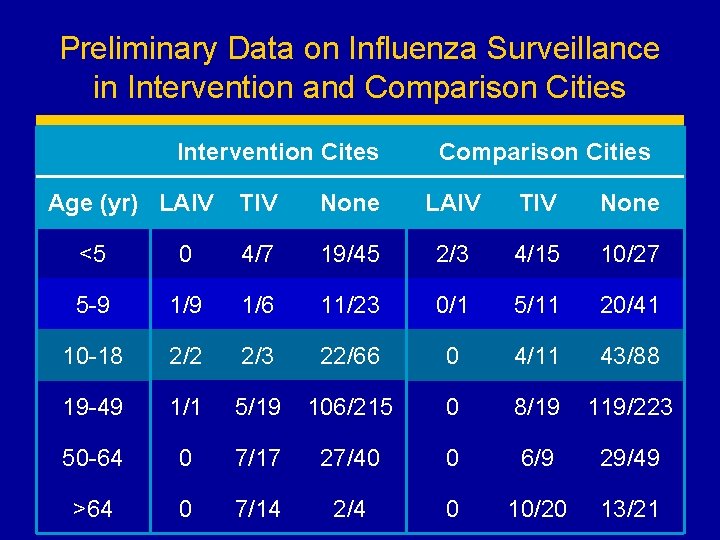
Preliminary Data on Influenza Surveillance in Intervention and Comparison Cities Intervention Cites Age (yr) LAIV Comparison Cities TIV None LAIV TIV None <5 0 4/7 19/45 2/3 4/15 10/27 5 -9 1/6 11/23 0/1 5/11 20/41 10 -18 2/2 2/3 22/66 0 4/11 43/88 19 -49 1/1 5/19 106/215 0 8/19 119/223 50 -64 0 7/17 27/40 0 6/9 29/49 >64 0 7/14 2/4 0 10/20 13/21

Conclusion The school-based influenza vaccination program was well accepted by the community and offers an option for rapid and efficient vaccination of school children and staff.

CONTROL OF EPIDEMIC INFLUENZA NIH Grant 2 U 01 AI 41050 P. A. Piedra, Principal Investigator, Baylor College of Medicine W. P. Glezen, Study Chair, Baylor College of Medicine M. J. Gaglani, PI, Scott & White Clinics G. Herschler, Study Coordinator, Scott & White Clinics Charles Fewlass, Data Management, Scott & White Clinics Dianne Harvey, Record Management, Scott & White Clinics Claudia Kozinetz, Epidemiologist, Baylor College of Medicine Betz Halloran, Statistical Consultant, Fred Hutchinson Ira M. Longini, Statistical Consultant, Fred Hutchinson Sonnie Kim, Influenza Program Officer, NIAID

Acknowledgements Baylor Col Med D Wilson R Fader Fred Hutchison PA Piedra R Cameron F Bagwell I Longini PW Glezen C Connally M Riggs B Halloran CA Kozinetz A Mow N Zimmerman U of Texas K Patel M Watts P Kirkpatrick S Lilibridge T Bosshard S Slater S Clark Med. Immune S Thaker C Morgan W White F Malinoski S Kaplan J Wilson R Flores NIH Scott & White A Linder S Holleman L Lambert MJ Gaglani O Ametane TX Dept Health S Kim GB Herschler P Smith P Wright C Fewlass D Miller N Pascoe
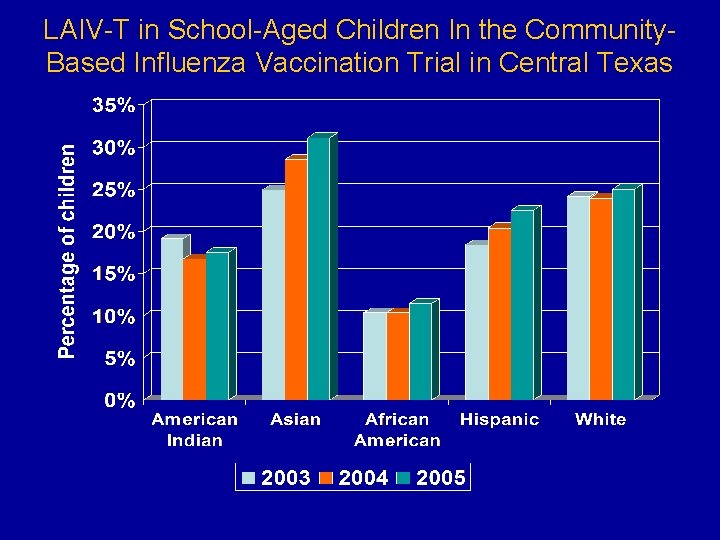
LAIV-T in School-Aged Children In the Community. Based Influenza Vaccination Trial in Central Texas
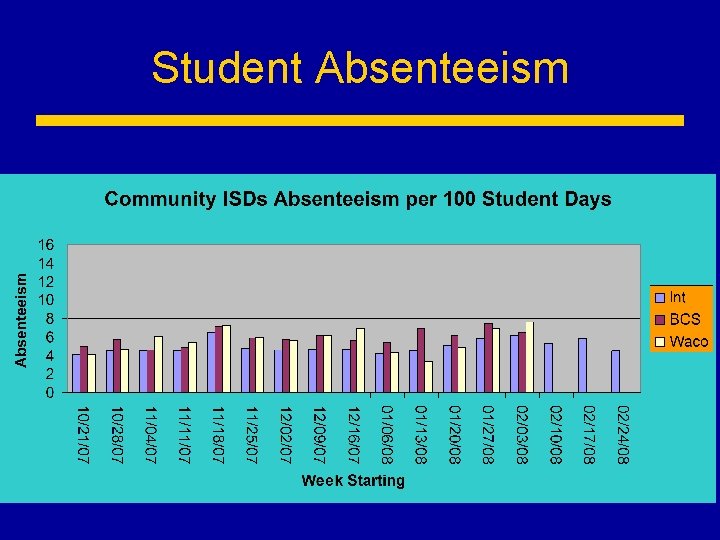
Student Absenteeism

2004 -2005 Influenza Surveillance Total Intervention cities Comparison cities Influenza A Influenza B % positive 404 97 (24. 0%) 17 (4. 2%) 28. 2% 713 329 (46. 1%) 46 (6. 5%) 52. 6% 2004 -05 U. S. outbreak viruses Vaccine-like: A/Fujian/4112/2002 (H 3 N 2): 17%; and B/Shanghai/361/2002: 15. 2%. Drift variant: A/California/7/2004 (H 3 N 2): 59%; and B/Victoria/2/87 -like: 6. 3% Outbreak period: January 9, 2005 to March 12, 2005.
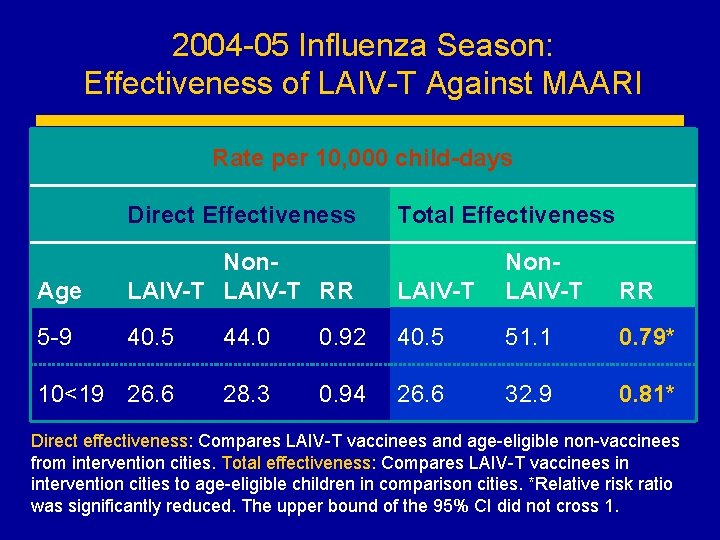
2004 -05 Influenza Season: Effectiveness of LAIV-T Against MAARI Rate per 10, 000 child-days Direct Effectiveness Age Non. LAIV-T RR 5 -9 40. 5 44. 0 10<19 26. 6 28. 3 Total Effectiveness LAIV-T Non. LAIV-T RR 0. 92 40. 5 51. 1 0. 79* 0. 94 26. 6 32. 9 0. 81* Direct effectiveness: Compares LAIV-T vaccinees and age-eligible non-vaccinees from intervention cities. Total effectiveness: Compares LAIV-T vaccinees in intervention cities to age-eligible children in comparison cities. *Relative risk ratio was significantly reduced. The upper bound of the 95% CI did not cross 1.
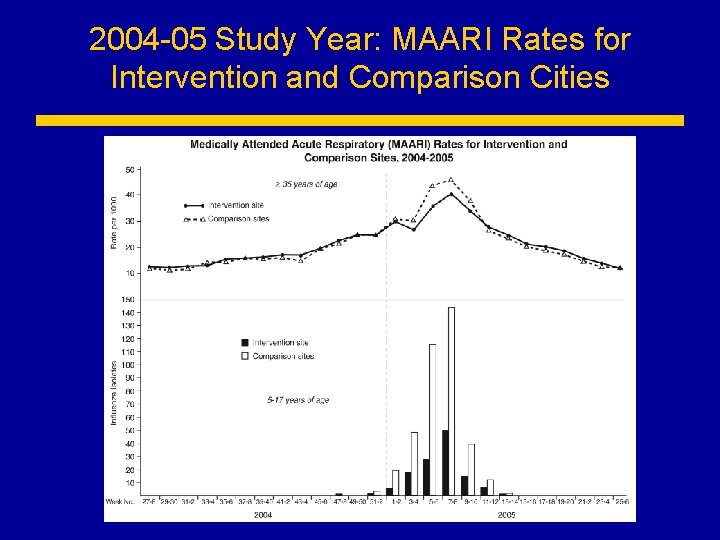
2004 -05 Study Year: MAARI Rates for Intervention and Comparison Cities
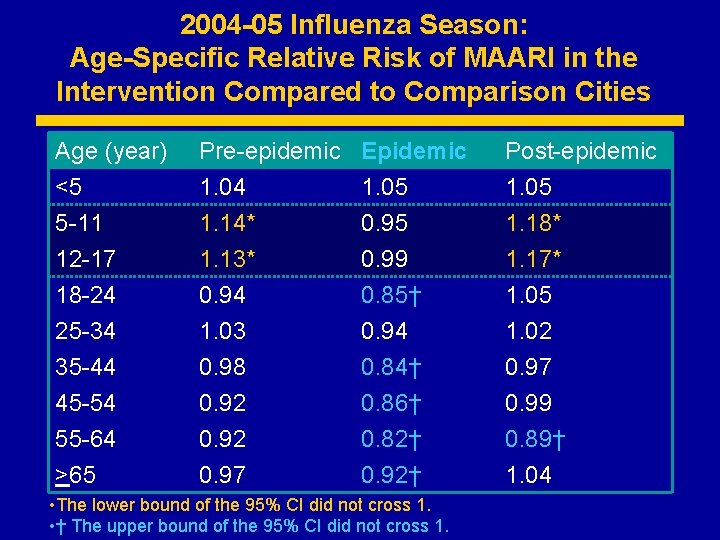
2004 -05 Influenza Season: Age-Specific Relative Risk of MAARI in the Intervention Compared to Comparison Cities Age (year) <5 5 -11 12 -17 Pre-epidemic 1. 04 1. 14* 1. 13* Epidemic 1. 05 0. 99 Post-epidemic 1. 05 1. 18* 1. 17* 18 -24 25 -34 35 -44 45 -54 55 -64 >65 0. 94 1. 03 0. 98 0. 92 0. 97 0. 85† 0. 94 0. 84† 0. 86† 0. 82† 0. 92† 1. 05 1. 02 0. 97 0. 99 0. 89† 1. 04 • The lower bound of the 95% CI did not cross 1. • † The upper bound of the 95% CI did not cross 1.

2005 -2006 Influenza Surveillance Total Number Influenza A Influenza B Intervention cities 717 167 (23. 3%) 1 (0. 1%) Comparison cities 499 133 (26. 7%) 27 (5. 4%) Outbreak period: December 4, 2005 to January 28, 2006 = influenza A January 29, 2006 to April 8, 2006 = influenza B.
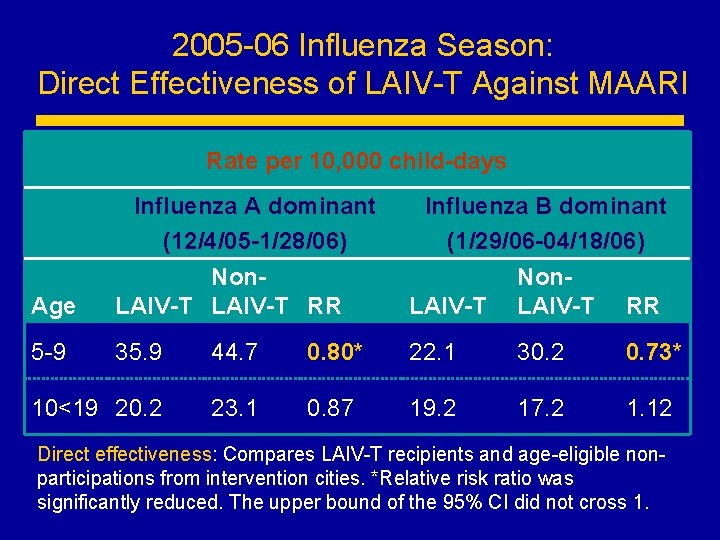
2005 -06 Influenza Season: Direct Effectiveness of LAIV-T Against MAARI Rate per 10, 000 child-days Influenza A dominant (12/4/05 -1/28/06) Age Non. LAIV-T RR 5 -9 35. 9 44. 7 10<19 20. 2 23. 1 Influenza B dominant (1/29/06 -04/18/06) LAIV-T Non. LAIV-T RR 0. 80* 22. 1 30. 2 0. 73* 0. 87 19. 2 17. 2 1. 12 Direct effectiveness: Compares LAIV-T recipients and age-eligible nonparticipations from intervention cities. *Relative risk ratio was significantly reduced. The upper bound of the 95% CI did not cross 1.
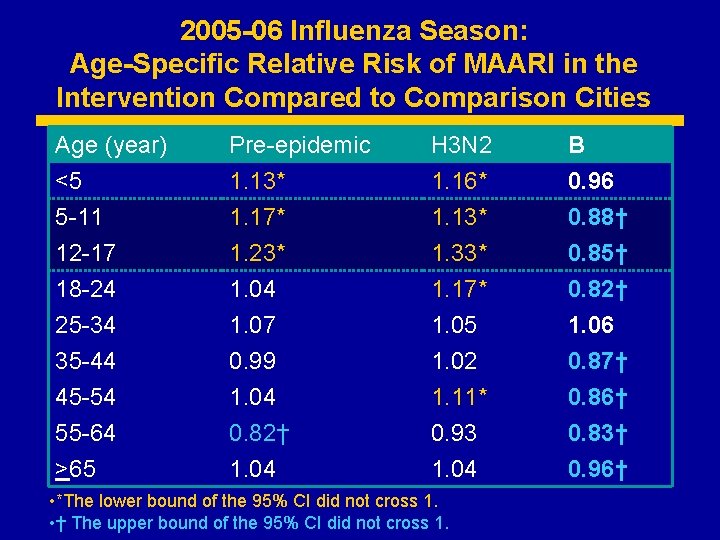
2005 -06 Influenza Season: Age-Specific Relative Risk of MAARI in the Intervention Compared to Comparison Cities Age (year) <5 5 -11 12 -17 Pre-epidemic 1. 13* 1. 17* 1. 23* H 3 N 2 1. 16* 1. 13* 1. 33* B 0. 96 0. 88† 0. 85† 18 -24 25 -34 35 -44 45 -54 55 -64 >65 1. 04 1. 07 0. 99 1. 04 0. 82† 1. 04 1. 17* 1. 05 1. 02 1. 11* 0. 93 1. 04 0. 82† 1. 06 0. 87† 0. 86† 0. 83† 0. 96† • *The lower bound of the 95% CI did not cross 1. • † The upper bound of the 95% CI did not cross 1.
- Slides: 36