Developing a Research Study Protocol From an Idea
























































- Slides: 61

Developing a Research Study Protocol “From an Idea to Implementation” Sadaf Aslam, MD Clinical Research Instructor USF Health, Clinical and Translational Sciences Institute July 18 th, 2012

What do we need to know? n Research terminology n Basic study designs/research question n Required elements of a research study protocol n Identify and avoid common mistakes n Understand PI, IRB and Institutional responsibility

How is research defined? n n Research “Systematic investigation including research development, testing and evaluation, designed to develop or contribute to a generalizable knowledge’ Human Subject “A living individual about whom an investigator obtains data through intervention or interaction or through identifiable private information” Federal policy [45 CFR 46. 102(f)]

What is Clinical Research? Breaking the Scientific Bottleneck Basic Sciences Research: Finding the cause of disease Translational Research: Making the results applicable to the population Association of American Medical Colleges American Medical Association Clinical Research : Patient oriented research-studies on mechanisms of disease, therapies and intervention, new technology and includes clinical trials Epidemiological and behavioral studies Outcomes and Health services research (NIH )

Clinical Trials n n Any prospective studies designed to evaluate the safety and efficacy of new drug, device or any behavioral intervention mostly against a control gold standard of clinical research Any biomedical and health related study that follows a predefined protocol either by observation or intervention involving human subjects

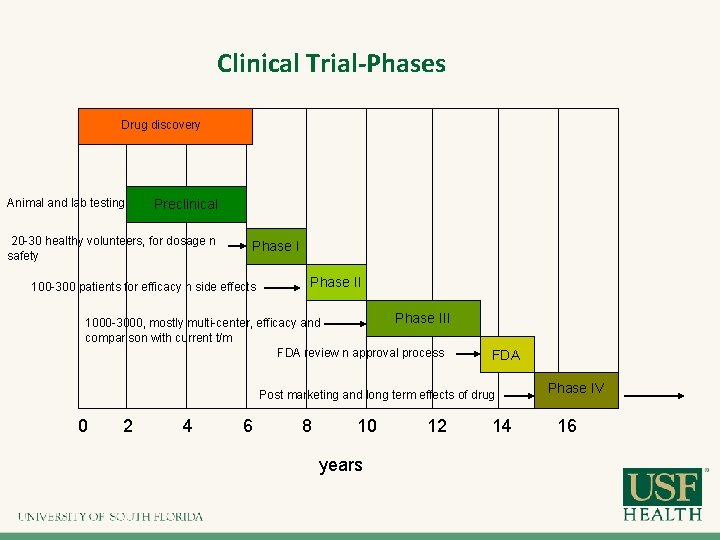
Clinical Trial-Phases Drug discovery Animal and lab testing Preclinical 20 -30 healthy volunteers, for dosage n safety Phase I 100 -300 patients for efficacy n side effects Phase III 1000 -3000, mostly multi-center, efficacy and comparison with current t/m FDA review n approval process FDA Post marketing and long term effects of drug 0 2 4 6 8 10 years 12 14 Phase IV 16

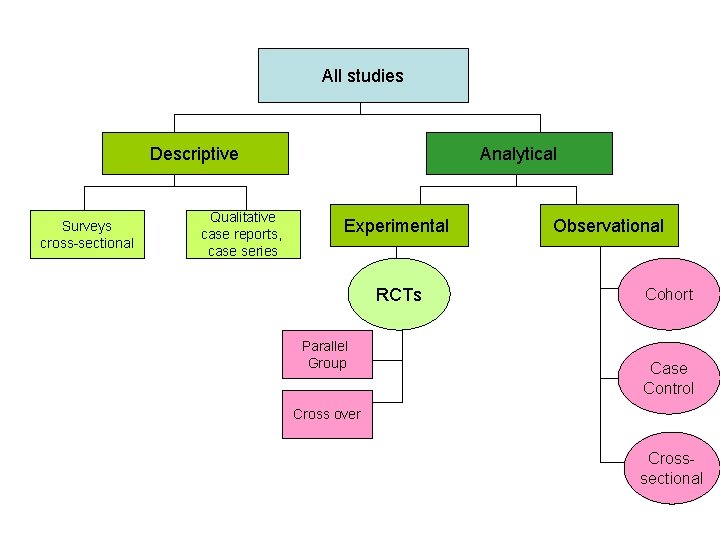
All studies Descriptive Surveys cross-sectional Qualitative case reports, case series Analytical Experimental RCTs Parallel Group Observational Cohort Case Control Cross over Crosssectional

Getting started “The best time to contemplate the quality of evidence from a clinical trial is before it begins. Conceptualizing and designing good clinical trials is never an accident but results from careful planning” Clinical Trials: A Methodologic Perspective by Steven Piantadosi

Main Steps 1: Formulating a research question 2: Drafting a study Protocol 3: Regulatory submission and Approval 4: Implementation

Research Question - A research question is the starting point of all studies and addresses the uncertainty an investigator has and is trying to find an answer - Forms the foundation of your study - Must be specified before starting the analysis

Origin of a research question n n Published literature Scholarship Mentor New ideas and techniques Surroundings- clinical setting, grand rounds, journal clubs etc

Types of Questions n Background Generally ask what, when, how, where …about the disease, disorder, or treatment n Foreground Patient oriented questions cover specific aspects with interpretation of therapy, disease, prognosis, diagnostic, risk etc and give answers based on outcome

A 30 -year-old woman presents with progressive weakness of both legs. She has difficulty in walking, climbing stairs, and standing up from a sitting position. A diagnosis is made of "demyelinating disease of the central nervous system" and intravenous methylprednisolone is prescribed. The patient is informed that her symptoms and test results will be monitored over time. She wants to know what her chances are of developing multiple sclerosis (MS) in the next few years.

A 37 -year-old pregnant woman comes to the clinic for a routine checkup. This is her second pregnancy. She had her first child when she was 35 years old and had an amniocentesis at 18 weeks to test for Down syndrome. The amniocentesis was negative. She is currently 8 weeks pregnant and would like to know of any abnormalities. An ultrasound can be done in the first trimester for diagnosing Down syndrome, but it is unknown if the results are as reliable as the conventional test of amniocentesis.

In elderly patients with heart failure with an ejection fraction of 40%, is Quinapril effective in reducing the need for rehospitalization?

Key Concept n n P- Population I- Intervention C- Control O- Outcome

Examples of a research question n Is omega 3 fish oil beneficial for health? Changing it to hypothesis (prove or disprove) We hypothesize that taking omega 3 fish oil is beneficial for health Improved hypothesis: In post menopausal women, we hypothesize that women who take omega 3 fish oil will have a a lower risk of heart disease then those who don’t take it. Research Question: Does Omega 3 fish oil reduce the risk of CHD in post menopausal women?

Should postmenopausal women receive hormones? Population: Predictor: Outcome: postmenopausal women “hormones” ? Changing it to hypothesis (prove or disprove) We hypothesize that taking hormones is beneficial for postmenopausal women Ref: Designing Clinical Research by Dr. Stephen Hulley

Improved Research Question Does estrogen treatment prevent heart attacks in postmenopausal women? Population: postmenopausal women Predictor: estrogen treatment vs none Outcome: heart attacks Improved hypothesis: In post menopausal women, we hypothesize that women who take estrogen treatment will have a a lower risk of heart disease than those who don’t take it.

Cohort design Population: - 5000 women age 55 and above living in the Tampa Bay Area Predictor: - Post-menopausal estrogen at baseline Outcome: - Follow-up 5 -year incidence of heart attacks

Case-control design Population - Cases: 100 women with heart attacks at TGH ED - Controls: 100 women matched age and demographics TGH ED Predictor: - Post-menopausal estrogen intake Outcome: - Heart attack in Cases vs controls

Cross-sectional design Population: - 2000 women age 55+ seen at Women’s Health Center Predictor: - Taking post-menopausal estrogen? Outcome: - History of heart attack?

Characteristics of a Research Question in Context of a Study Design: FINER n n n Feasible Interesting Novel Ethical Relevant Designing Clinical Research by Stephen Hulley

Criteria for Feasibility n n Adequate number of subjects Knowledge and expertise Adequate resources regarding time and money If manageable in scope

Criteria for Interesting n Depends on your passion, motivation n Something to be an expert in n Finding the truth and making a difference

Criteria for Novelty n n Comes from thoroughly reviewing the literature Confirms or refutes previous findings Extends previous findings Provides new findings How to find Novelty? n n Mastering the published literature-resources- EBM, Medline Ovid, pubmed Perform systematic reviews and meta-analysis

Critically reviewing literature!! n Introduction: Why did they initiate the research? n Methods: What did they do? n Results: What did they find? n Discussion: What do the results mean?

Purpose of Thorough Literature Search n n n Places other’s work in the context of its contribution to the topic and provides critical review If the research question is significant and the work is original and important Identifies new ways to interpret and find any gaps in previous research Identify areas of prior research to prevent duplication of effort Helps formulate your own research question and study

Working with Literature n n Find -types, resources, skills, knowledge Manage – reading, keeping track, annotation Use – research topic, question, justification, design, method Review –writing background section, working on style n tone

Criteria for Ethical n n Respect for human subjects Respect for confidentiality Respect for informed consent Respect for beneficence and justice

Criteria for Relevance n n n To add on to scientific knowledge To benefit clinic, public health or health policy For future research directions

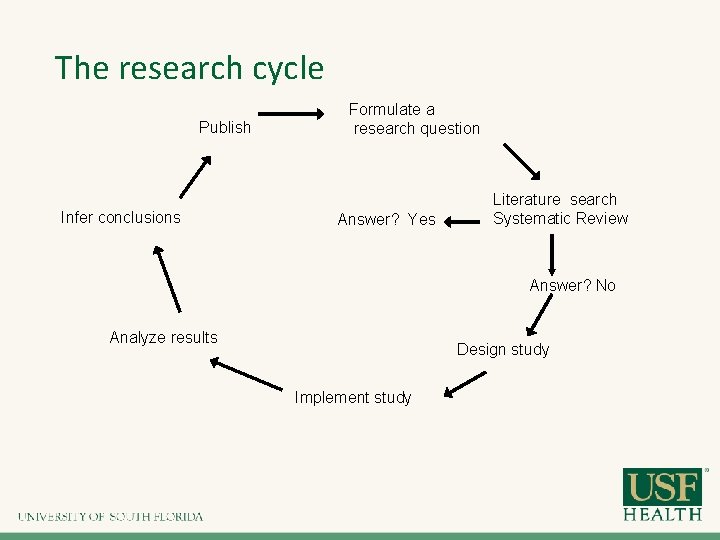
The research cycle Publish Infer conclusions Formulate a research question Answer? Yes Literature search Systematic Review Answer? No Analyze results Design study Implement study

Step 2: Drafting a study protocol

Protocol • A study protocol is a formal document which leads to a systematic approach about the modes to conduct a study based on ethical, scientific and organizational grounds • A document that describes the objective, design, methodology, statistical consideration and organization of a trial (ICH definition)

Outline of a study protocol 1. 2. 3. 4. 5. 6. 7. 8. Title Abstract/synopsis Background/significance/rationale Objectives/specific aims/research question Study design/Methodology/Procedures Informed consent process-confidentiality Overall Management Plan References

1: Title n Consistency across all documents 2: Abstract/Synopsis n Abstract- question, rationale, convenient reminder of the specifics of proposal for everyone specially some non scientific reviewers

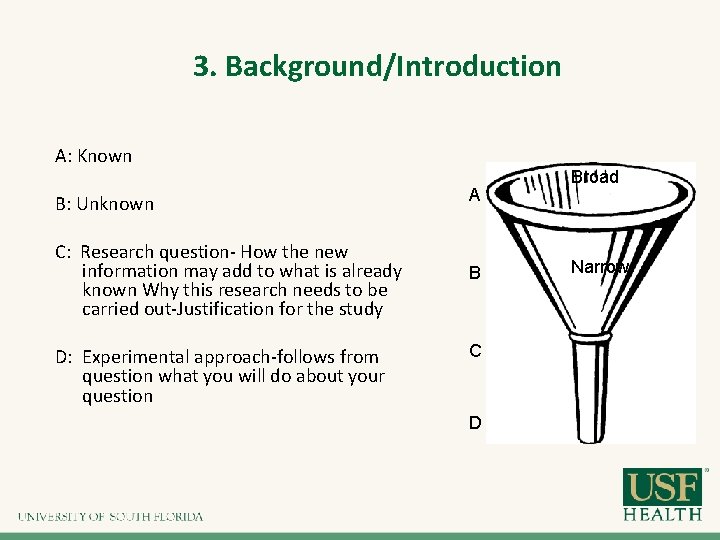
3. Background/Introduction A: Known B: Unknown A C: Research question- How the new information may add to what is already known Why this research needs to be carried out-Justification for the study B D: Experimental approach-follows from question what you will do about your question C D Broad Narrow

Example of Writing a Background Section: Title: The Effect of Dietary Omega-3 Fatty Acids on Coronary Atherosclerosis Ingestion of fish or other sources of Omega-3 fatty acids has been called a comprehensive strategy toward the prevention of atherosclerosis (1). Hundreds of epidemiologic studies, studies of mechanisms of action, and studies in experimental animals have shown that dietary intake of Omega-3 fatty acids has anti atherosclerotic potential (1– 9)……. . However, only a few clinical trials have shown that Omega-3 fatty acids have cardiovascular benefit in humans…… To test the hypothesis that consuming Omega-3 fatty acids for 2 years leads to less progression and more regression of coronary atherosclerosis, as assessed by coronary angiography, we will conduct a randomized, double-blind, placebocontrolled study in patients with coronary atherosclerosis. April 1999 · Annals of Internal Medicine · Volume 130 · Number 7 Clemens von Schacky, MD; Peter Angerer, MD; Wolfgang Kothny, MD; Karl Theisen, MD; and Harald Mudra, MD

4. Objectives • Objective is the research question that needs to be answered based on the rationale or hypothesis • Each objective should have a corresponding discussion in the statistical section Primary Objective: To evaluate the effect of omega 3 fish oil in reducing the risk for CHD in post menopausal women Secondary Objective: To assess the effects of the Omega 3 fish oil with respect to 6 months and 12 months development of variables such as glucose, lipids, hormones, and BMI

5. Design and Methods Section a) b) c) d) e) Study design/methods Sample size Study subject selection/population Statistical Analysis Plan Data collection methods and Data management

a) Study design/Methods n n n Descriptive: What is average number of servings of omega-3 fish oil per week in the diet of patients with a history of CHD ? Analytical: Is there an association between fish oil and risk of developing CHD? Clinical trial: Does treatment with omega 3 capsules reduce the incidence of CHD in diabetic patients? • • Randomization /Blinding Process of recruitment Flow chart Methods of measurement

b) Sample Size n n n Provide the rationale for the sample size and the clinical justification If sample size shows certain level of power and effect size (If phenomenon is rare, effect size will be small and more subjects are needed) Sample size/power analysis

c) Study Population /Selection of Subjects n Define Inclusion and exclusion criteria n If the selected population is appropriate to answer the RQ

d) Statistical Plan Details of the planstatistical test to test each hypothesis/expected data/ Coding Plan for any missing, spurious and unused data Procedures for reporting deviations from the original statistical plan Software you will be using for analysis

e) Data collection/management and access n How the data collected will be analyzed to determine the health outcomes n Validity and reliability of the data collecting instrument n Describe the sources of the data in case of chart reviews

6. Informed consent Requires all essential elements and it’s a process

Confidentiality n n Must comply with the data protection act Privacy and confidentiality (PHI- maintained by the covered entities) De-identification of the data In case of any communicable disease how to limit the reporting requirements

7. Overall Project management n Duration of your study and time line n Handling and storage of data n Reporting to participants, sponsors, community n Reporting of any conflict of interest n Identify any planned ways to present your results

8. References List all the references used in the background section or in the protocol text at the end of the protocol-use NIH format or endnote Attachments and Appendices Budget , PI’s–CV, certification, Data collection forms, request to access data, Site agreement, ICF, HIPAA waiver, amendments

Step 3: Regulatory Submission and Approval n n Department Review/ Scientific Review IRB review-process Site approval COI Review

Scientific Review Focus n n n n n Research question Study design/methodology Systematic review Research team is qualified Background and rationale Sample size Subject selection Statistical plan Ensure the protection of human rights and welfare through study design

IRB Review Focus n n n Protection of rights and welfare of human subjects Risk/ benefit analysis Adverse event reporting plan Human subject selection/vulnerable population Privacy and confidentiality Informed consent process Monitoring and data safety management Subjects participation with payments Continuing reviews Consistency across study Conflict of interest

Top Ten Problems which IRB Finds in your Application n n Inconsistency-numbers, Inclusion/exclusion, funding, study procedures Shared responsibility- documents, application not read by dept chair Writing and language- copying and pasting from grants or other templates Too much or too less info-differentiate b/w SOC and research, Poorly written consent forms- not using templates Risk/benefit Compensation for t/m of adverse events Conflict of interest Research team Continuing reviews

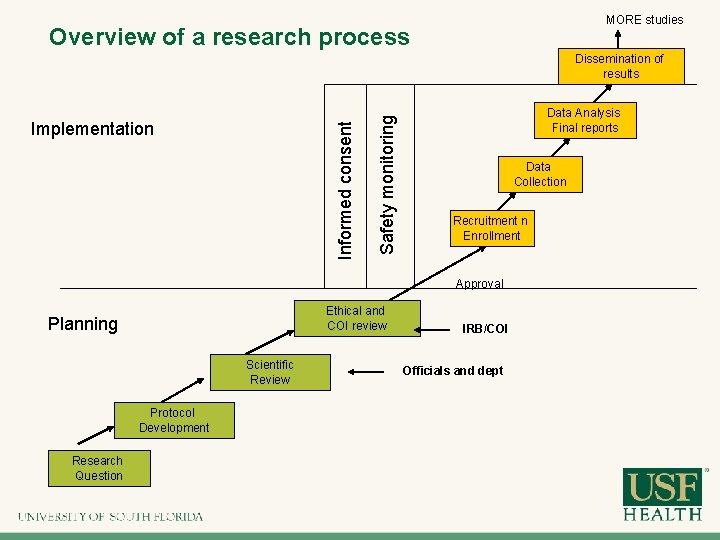
MORE studies Overview of a research process Informed consent Implementation Safety monitoring Dissemination of results Data Analysis Final reports Data Collection Recruitment n Enrollment Approval Ethical and COI review Planning Scientific Review Protocol Development Research Question IRB/COI Officials and dept

Finally! points to remember n Writing a protocol is a team effort n Takes time and would lead to many drafts initially n n n Involve your mentor, statistician, medical writer, data analyst from the start Review all relevant literature critically Expect multiple reviews from OCR, sponsors, and IRB Your draft version and date are very important, use footer with short title and date If any amendments to the IC or protocol, change to either requires IRB review and approval Follow templates posted on the websites

Few points to consider Do you still need IRB review? n If you are using existing data n If you are collecting biological specimens for research through a non invasive procedures n If you are writing for publication n If your research involves greater than minimal risk, what kind of IRB review is required? Is there any study population that needs special consideration when planning the recruitment of human subjects?

Few More Points to Consider What reports are you expected to submit to the IRB once your study is approved? n Any unanticipated problem needs reporting to the IRB chair and sponsor n Immediate reporting using AE reporting form in case of AE n Annual progress report/continuing reviews n Final report after the study is complete n Major deviation from the protocol

USF Regulatory/ Ethics/COI Research support Sadaf Aslam-813 -250 -2205 Scientific Review Committee under CTSI Benjamin Djulbegovic-813 -974 -9178 Sadaf Aslam-813 -250 -2205 USF Health Office of Clinical Research (OCR) Catherine Jahrsdorfer-813 -396 -9172 TGH OCR Jennifer Cooper 813 -844 -4236 Conflict Of Interest Camille Mc. Whirter- 813 -974 -6676

Finding Funding sources USF Office of Research DSR- USF Internal Grants http: //www. research. usf. edu/sr/internal_awards/summaries. htm USF Health Office of Clinical Research NIH, NSF, non profit organizations http: //www. grants. gov/ National Science Foundation http: //www. nsf. gov/funding/ Congressionally Directed Medical Research Program http: //cdmrp. army. mil/ Centers for Disease Control http: //www. cdc. gov/od/pgo/funding/grants/foamain. shtm Department of Defense Do. D information and links

Education and Training • COM courses on Clinical Research Methods GMS-6875 -Ethical and Regulatory aspect of clinical research • Online certificate course on Clinical Research Methods • COPH -Clinical Research related course PHC 6017 -Design and conduct of clinical trials • IRB offers several training courses for new investigators • Health Sciences Shimberg library http: //library. hsc. usf. edu/ • Look for monthly COM events

But why think, why not try the experiment? John Hunter, 1775