Determining the Empirical Formula of Copper Chloride Purpose



























![Copper is a Transition Metal. Ground state electron configuration: [Ar]. 3 d 10. 4 Copper is a Transition Metal. Ground state electron configuration: [Ar]. 3 d 10. 4](https://slidetodoc.com/presentation_image/a46c2eb156d1b7e54e3ae6189b2de696/image-32.jpg)








- Slides: 42

Determining the Empirical Formula of Copper Chloride Purpose of the Experiment To determine the empirical formula of a compound containing only copper and chlorine.

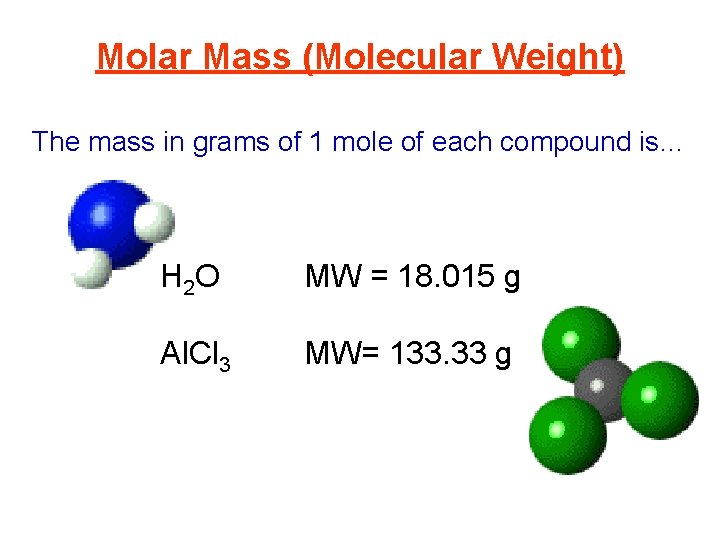
Molar Mass (Molecular Weight) The mass in grams of 1 mole of a compound is equal to the mass in grams of the components. H 2 O MW = 2(1. 008) + 15. 999 g Al. Cl 3 MW = 26. 98 + 3(35. 45) g (Note: The Significant Figures. ) So the mass in grams of 1 mole of each compound is…

Molar Mass (Molecular Weight) The mass in grams of 1 mole of each compound is… H 2 O MW = 18. 015 g Al. Cl 3 MW= 133. 33 g

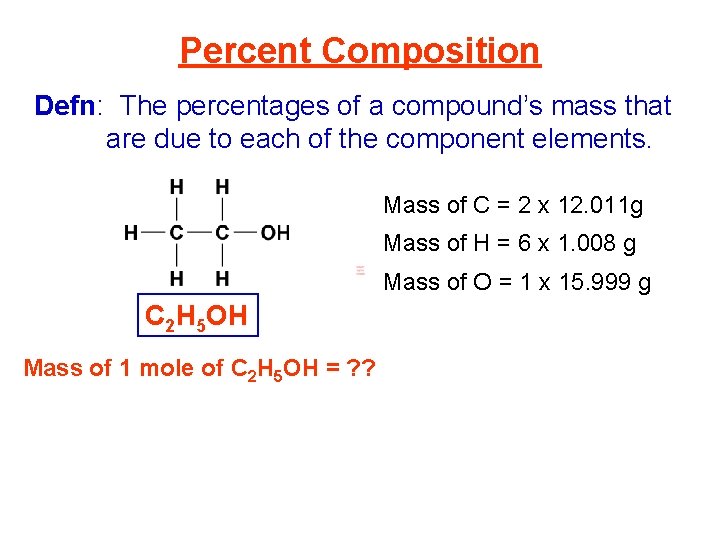
Percent Composition Defn: The percentages of a compound’s mass that are due to each of the component elements. Mass of C = 2 x 12. 011 g Mass of H = 6 x 1. 008 g Mass of O = 1 x 15. 999 g C 2 H 5 OH Mass of 1 mole of C 2 H 5 OH = ? ?

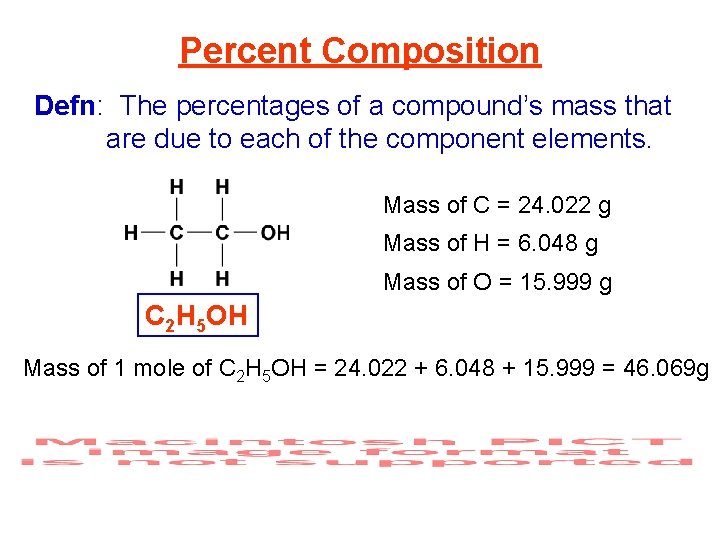
Percent Composition Defn: The percentages of a compound’s mass that are due to each of the component elements. Mass of C = 24. 022 g Mass of H = 6. 048 g Mass of O = 15. 999 g C 2 H 5 OH Mass of 1 mole of C 2 H 5 OH = 24. 022 + 6. 048 + 15. 999 = 46. 069 g Mass percent of C = ? ?

Percent Composition Defn: The percentages of a compound’s mass that are due to each of the component elements. Mass of C = 24. 022 g Mass of H = 6. 048 g Mass of O = 15. 999 g C 2 H 5 OH Mass of 1 mole of C 2 H 5 OH = 24. 022 + 6. 048 + 15. 999 = 46. 069 g

Empirical Formula Represents the simplest whole-number ratio of the various types of atoms in a compound. Examples What is the Empirical Formula for the following compounds? Drying Agent Scent of Bananas P 4 O 10 C 14 H 28 O 4 Caffeine C 8 H 10 N 4 O 2

Empirical Formula Represents the simplest whole-number ratio of the various types of atoms in a compound. Examples What is the Empirical Formula for the following compounds? Drying Agent Scent of Bananas Caffeine P 4 O 10 C 14 H 28 O 4 C 8 H 10 N 4 O 2 P 2 O 5 C 7 H 14 O 2 C 4 H 5 N 2 O

Empirical Formula Example 1 A white compound is analyzed and found to contain 43. 64% phosphorous and 56. 36% oxygen by mass. What is the empirical formula? 1 st Step:

Empirical Formula Example 1 A white compound is analyzed and found to contain 43. 64% phosphorous and 56. 36% oxygen by mass. What is the empirical formula? 1 st Step: Because it is a percent we can choose any mass we want and multiply that mass by the percents of the components. What is the easiest mass to choose?

Empirical Formula Example 1 A white compound is analyzed and found to contain 43. 64% phosphorous and 56. 36% oxygen by mass. What is the empirical formula? 1 st Step: Because it is a percent we can choose any mass we want and multiply that mass by the percents of the components. What is the easiest mass to choose? 100. 00 grams

Empirical Formula Example 1 A white compound is analyzed and found to contain 43. 64% phosphorous and 56. 36% oxygen by mass. So since we chose 100. 00 g of compound we have 43. 64 g P & 56. 36 g O. Can we compare grams directly? 2 nd Step:

Empirical Formula Example 1 A white compound is analyzed and found to contain 43. 64% phosphorous and 56. 36% oxygen by mass. So since we chose 100. 00 g of compound we have 43. 64 g P & 56. 36 g O. Can we compare grams directly? 2 nd Step: Convert grams to moles. 43. 64 g P x ? ? ? 56. 36 g O x ? ? ?

Empirical Formula Example 1 A white compound is analyzed and found to contain 43. 64% phosphorous and 56. 36% oxygen by mass. So since we chose 100. 00 g of compound we have 43. 64 g P & 56. 36 g O. Can we compare grams directly? 2 nd Step: Convert grams to moles. 43. 64 g P x (1 mol P / 30. 974 g P) = 1. 409 mol P 56. 36 g O x (1 mol O / 15. 999 g O) = 3. 523 mol O

Empirical Formula Example 1 A white compound is analyzed and found to contain 43. 64% phosphorous and 56. 36% oxygen by mass. So we now have 1. 409 moles of P & 3. 523 moles of O. What can we do to compare the moles? 3 rd Step:

Empirical Formula Example 1 A white compound is analyzed and found to contain 43. 64% phosphorous and 56. 36% oxygen by mass. So we now have 1. 409 moles of P & 3. 523 moles of O. What can we do to compare the moles? 3 rd Step: Convert the smaller number to one. Do this by dividing both mole values by the smaller one. 1. 409 = ? P and 1. 409 3. 523 =? O 1. 409

Empirical Formula Example 1 A white compound is analyzed and found to contain 43. 64% phosphorous and 56. 36% oxygen by mass. So we now have 1. 409 moles of P & 3. 523 moles of O. What can we do to compare the moles? 3 rd Step: Convert the smaller number to one. Do this by dividing both mole values by the smaller one. 1. 409 = 1 P and 1. 409 3. 523 = 2. 5 O 1. 409

Empirical Formula Example 1 A white compound is analyzed and found to contain 43. 64% phosphorous and 56. 36% oxygen by mass. So by dividing both mole values by the smaller one we deduced: 1. 409 = 1 P and 1. 409 3. 523 = 2. 5 O 1. 409 This yields the formula PO 2. 5 Is this an acceptable empirical formula?

Empirical Formula Example 1 A white compound is analyzed and found to contain 43. 64% phosphorous and 56. 36% oxygen by mass. So by dividing both mole values by the smaller one we deduced: 1. 409 = 1 P and 1. 409 3. 523 = 2. 5 O 1. 409 This yields the formula PO 2. 5 Is this an acceptable empirical formula? No.

Empirical Formula Example 1 A white compound is analyzed and found to contain 43. 64% phosphorous and 56. 36% oxygen by mass. Since compounds must contain whole numbers of atoms, the empirical formula should contain only whole numbers. What must we do to fix the formula PO 2. 5?

Empirical Formula Example 1 A white compound is analyzed and found to contain 43. 64% phosphorous and 56. 36% oxygen by mass. Since compounds must contain whole numbers of atoms, the empirical formula should contain only whole numbers. What must we do to fix the formula PO 2. 5? 4 th Step: Convert to whole numbers. Do this by multiplying both mole values to get whole numbers.

Empirical Formula Example 1 A white compound is analyzed and found to contain 43. 64% phosphorous and 56. 36% oxygen by mass. Since compounds must contain whole numbers of atoms, the empirical formula should contain only whole numbers. What must we do to fix the formula PO 2. 5? 4 th Step: Convert to whole numbers. Do this by multiplying both mole values to get whole numbers. 1 P x 2 = 2 P 2. 5 O x 2 = 5 O Empirical Formula = P 2 O 5

Empirical Formula Example 2 A student burns 0. 353 g of magnesium ribbon in an open crucible. A white powder forms and is found to weigh 0. 585 g. What is the empirical formula? Atmospheric oxygen is in excess heat Mg (solid – silvery white) + O 2 (gas) → Mgx. Oy (solid – white) (0. 353 g) limiting reagent (0. 585 g) resulting mass How do we determine the empirical formula?

Empirical Formula Example 2 A student burns 0. 353 g of magnesium ribbon in an open crucible. A white powder forms and is found to weigh 0. 585 g. What is the empirical formula? Atmospheric oxygen is in excess heat Mg (solid – silvery white) + O 2 (gas) → Mgx. Oy (solid – white) (0. 353 g) limiting reagent (0. 585 g) resulting mass How do we determine the empirical formula? 1 st Step: Calculate mass percents.

Empirical Formula Example 2 1 st Step: Calculate mass percents. Mass of O = total mass of compound – mass of Mg = 0. 232 g Mg = 60. 3% and O = 39. 7%

Empirical Formula Example 2 2 nd Step: Formula Masses and their Percent Composition. Formula masses and percent composition of three theoretical compounds of Mg and O Mg = 24. 31 g/mole O = 15. 999 g/mole Formula of Oxide Mgx. Oy Formula Weight Mg. O 24. 31+15. 999 = Mg. O 2 24. 31 + 2(15. 999) = Mg 2 O 2(24. 31) + 15. 999 = Mg = 60. 3% and O = 39. 7% %Mg %O

Empirical Formula Example 2 2 nd Step: Formula Masses and their Percent Composition. Formula masses and percent composition of three theoretical compounds of Mg and O Mg = 24. 31 g/mole O = 15. 999 g/mole Formula of Oxide Mgx. Oy Formula Weight Mg. O 24. 31+15. 999 = 40. 309 Mg. O 2 24. 31 + 2(15. 999) = 56. 308 Mg 2 O 2(24. 31) + 15. 999 = 64. 619 Mg = 60. 3% and %Mg O = 39. 7% %O

Empirical Formula Example 2 2 nd Step: Formula Masses and their Percent Composition. Formula masses and percent composition of three theoretical compounds of Mg and O Mg = 24. 31 g/mole Formula of Oxide Mgx. Oy Mg. O 2 Mg 2 O O = 15. 999 g/mole Formula Weight 24. 31+15. 999 = 24. 31 + 2(15. 999) = 2(24. 31) + 15. 999 = Mg = 60. 3% and %Mg %O 40. 309 24. 31/ 40. 309 15. 999/ 40. 309 56. 308 24. 31/ 56. 308 2 x 15. 999/ 56. 308 64. 619 2 x 24. 31/ 64. 619 15. 999/ 64. 619 O = 39. 7%

Empirical Formula Example 2 2 nd Step: Formula Masses and their Percent Composition. Formula masses and percent composition of three theoretical compounds of Mg and O Mg = 24. 31 g/mole O = 15. 999 g/mole Formula of Oxide Mgx. Oy Formula Weight %Mg %O Mg. O 24. 31+15. 999 = 40. 309 60. 3 39. 7 Mg. O 2 24. 31 + 2(15. 999) = 56. 308 43. 2 56. 8 Mg 2 O 2(24. 31) + 15. 999 = 64. 619 75. 2 24. 8 Mg = 60. 3% and O = 39. 7%

Empirical Formula Example 2 2 nd Step: Formula Masses and their Percent Composition. Formula masses and percent composition of three theoretical compounds of Mg and O Mg = 24. 31 g/mole O = 15. 999 g/mole Formula of Oxide Mgx. Oy Formula Weight %Mg %O **Mg. O 24. 31+15. 999 = 40. 309 60. 3 39. 7 Mg. O 2 24. 31 + 2(15. 999) = 56. 308 43. 2 56. 8 Mg 2 O 2(24. 31) + 15. 999 = 64. 619 75. 2 24. 8 Mg = 60. 3% and O = 39. 7%

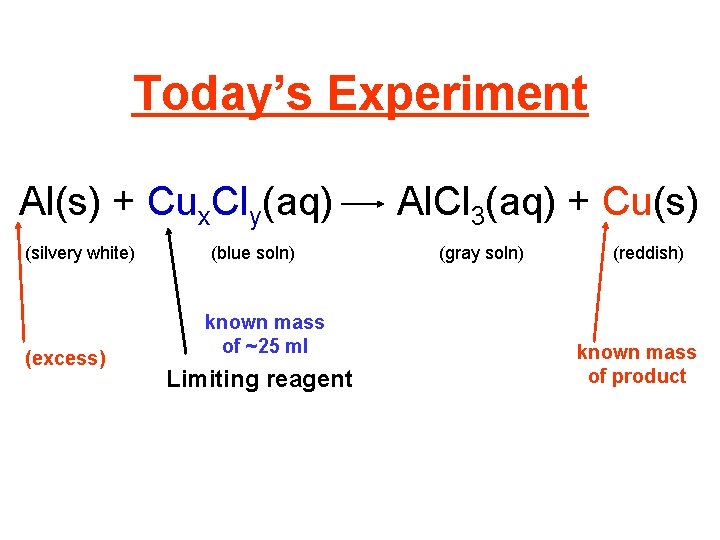
Today’s Experiment Al(s) + Cux. Cly(aq) (silvery white) (excess) (blue soln) known mass of ~25 ml Limiting reagent Al. Cl 3(aq) + Cu(s) (gray soln) (reddish) known mass of product
![Copper is a Transition Metal Ground state electron configuration Ar 3 d 10 4 Copper is a Transition Metal. Ground state electron configuration: [Ar]. 3 d 10. 4](https://slidetodoc.com/presentation_image/a46c2eb156d1b7e54e3ae6189b2de696/image-32.jpg)
Copper is a Transition Metal. Ground state electron configuration: [Ar]. 3 d 10. 4 s 1 Shell structure: 2. 8. 1 Transition metals may exhibit multiple oxidation states (+1, +2, +3, etc…). These are not easily predicted by position in the periodic table. Transition metals ions in aqueous solutions frequently are brightly colored, also due to d orbitals (e. g. Cu ions are blue).

Last week we worked with Zinc. Zn and Al are both stronger reducing agents than copper. (Note: the redox potentials on next slide. ) Because of this either one would work to produce metallic copper from a solution of a copper salt. We chose aluminum to work with because the reaction between Zinc and Copper Chloride is quite exothermic and extreme caution would have had to have been observed to avoid burns.

The oxidation of aluminum is more likely than the oxidation of zinc. * * * These potentials indicate the relative thermodynamic tendency for the indicated half-reaction to occur. Al <--> Al+3 + 3 e– Zn <--> Zn+2 + 2 e– Cu <--> Cu+2 + 2 e– E = -0. 166 volts E = -0. 763 volts E = +0. 34 volts

Removal of Excess Reducing Agent 2 Al(s) + 6 HCl(aq) --> 2 Al. Cl 3(aq) + 3 H 2(g) Cu(s) + n HCl(aq) -x-> No Reaction Once you have ensured that the aluminum is in excess (some aluminum will be floating on the surface), then it is time to proceed with the next step: removal of the aluminum. This is accomplished by adding HCl in excess. The HCl reacts with the aluminum and not the copper.

Checkout 2 pc Al foil – Add in excess. Do not fold. 1 - pair of Beaker Tongs Reagents in Lab Cu. Clx solution in 4 L spigot jugs - Take ~25 ml for each run (using 100 ml Grad Cylinder) Record data: (0. 08067 g Cu. Clx / ml, d=1. 074 g/ml) 10% HCl in 1 L wash bottles - Take ~5 ml for each run (using 10 ml Grad Cylinder) (N. B. solid Na. HCO 3 is to be used for acid spills. )

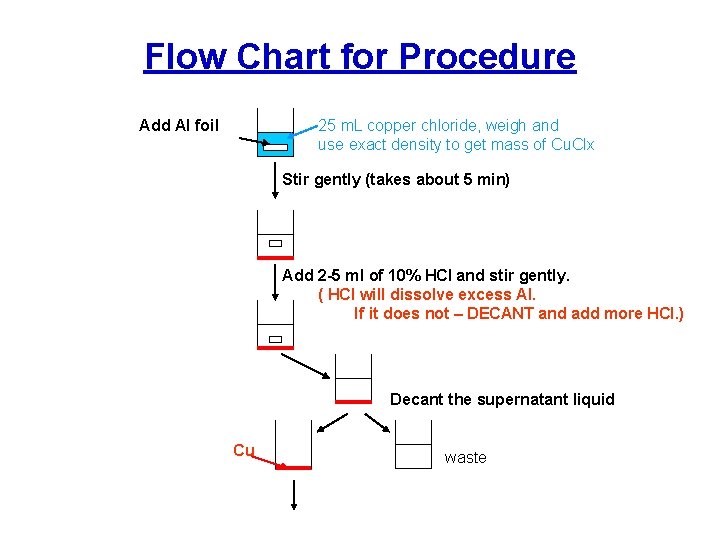
Flow Chart for Procedure Add Al foil 25 m. L copper chloride, weigh and use exact density to get mass of Cu. Clx Stir gently (takes about 5 min) Add 2 -5 ml of 10% HCl and stir gently. ( HCl will dissolve excess Al. If it does not – DECANT and add more HCl. ) Decant the supernatant liquid Cu waste

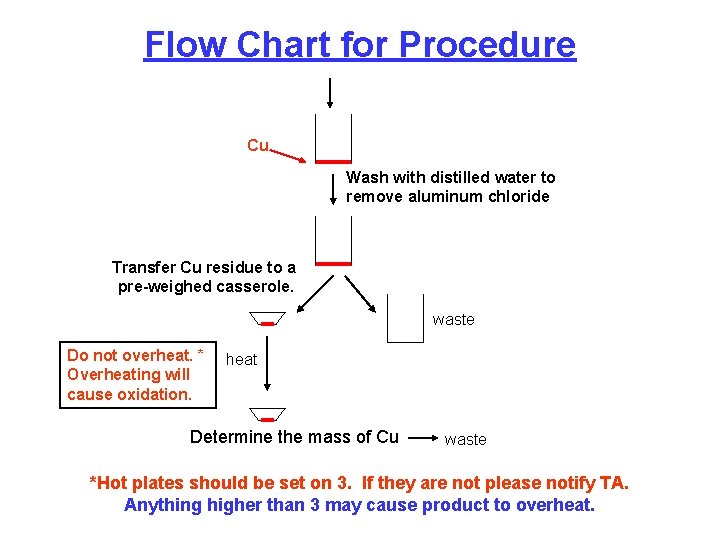
Flow Chart for Procedure Cu Wash with distilled water to remove aluminum chloride Transfer Cu residue to a pre-weighed casserole. waste Do not overheat. * Overheating will cause oxidation. heat Determine the mass of Cu waste *Hot plates should be set on 3. If they are not please notify TA. Anything higher than 3 may cause product to overheat.

Procedure Notes v. Record all weights to 0. 001 g. v. Weigh 25 ml of Cu. Clx solution, use exact density to calculate exact volume, then calculate the mass of Cu. Clx. v. Do not use metal forceps or spatulas when stirring solution. v. Add Al foil until blue color is gone, allow excess foil to dissolve also. v. Allow container to cool before weighing. v. Speed up cooling by placing in front of hood sash raised 4 -6”.

Hazards v 10% HCl-strong acid, corrosive v. Cux. Cly solution-heavy metal, irritant v. Hot surfaces - hotplates, glassware (Be Careful: Hot glassware looks just like cold glassware. ) Waste v. Liquid Waste: Al+3 / HCl v. Used Solids: Cu

Summary of Data & Calculations* Results (calculations) Collected data Mass of Cu. Clx Mass of Cu Mass percent of Cu Mass of Cl Mass percent of Cl Empirical formula *Note that on the bottom of page 79 it states that the calculations which need to be shown on a separate sheet of paper are indicated by asterisks (*).

Homework Assignment - Due September 22 -25. Read: Dimensional Analysis (pp. 22 -26) Do: Problem Set #2 (p. 23) 2. 1 -2. 4 & Set #3 (26) 3. 1 -3. 2 (On a separate sheet of paper, show your calculations. ) For September 22 -25 Read: Separating the Components of a Ternary Mixture p. 83. Turn-In: EF pp. 79 -82; Calculations Page (explained on p. 79) & DA Homework.