Determining Absolute Time Absolute Time numerical time using

Determining Absolute Time

� Absolute Time: numerical time using a specific units like years � Isotopes: Form of an element with more or fewer neutrons than other forms of the same element � Decay product: Element produced by the decay of a radioactive isotope � Decay-product ratio: Ratio between the mass of a radioactive element and the mass of the decay product

� If an isotope is radioactive, it will break down naturally into a lighter element called a decay product. � This process occurs at a predictable rate and can be used to determine how old an object is.

� Half-life: Time required for half of an element's atoms in a sample to change to the decay product. � In each half-life only half of the remaining radioactive atoms decay, no matter how large the sample is.
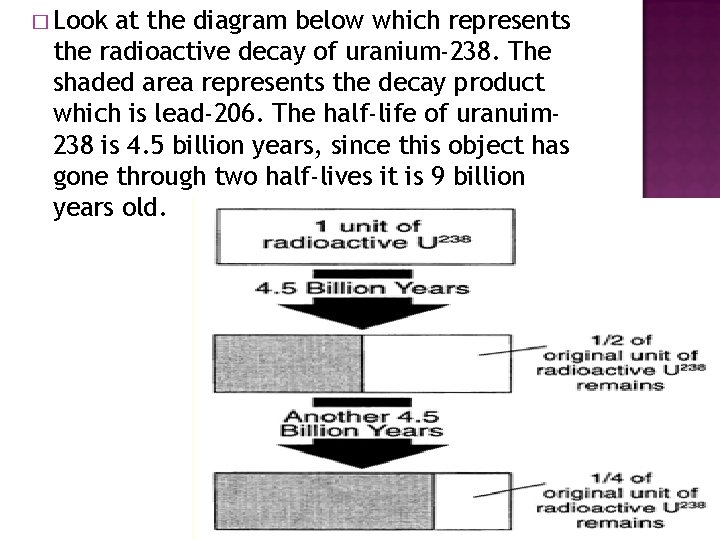
� Look at the diagram below which represents the radioactive decay of uranium-238. The shaded area represents the decay product which is lead-206. The half-life of uranuim 238 is 4. 5 billion years, since this object has gone through two half-lives it is 9 billion years old.

� Look at the decay-product ratio � This will help calculate the number of halflives that have occurred since the sample was formed. � Once you know that you can find the age of the sample.

� If an object is made up of 50 % decay product then the object has been through 1 half-life. � 75% decay product equals 2 half-lives � 87. 5% decay product equals 3 half-lives � 93. 76% decay product equals 4 half-lives, and so on. � Once you know the number of half-lives you can determine the age by using the half-life in years listed on the back of the Reference Tables.

� The chart on the next page gives information about the radioactive decay of carbon-14. � You could make a chart like this one for any radioactive decay question. � It will show the half-lives, radioactive element (or decay product) and the age of the sample are related. � Try doing this to see if it will help you. � You will want to have the front page of your ERST handy
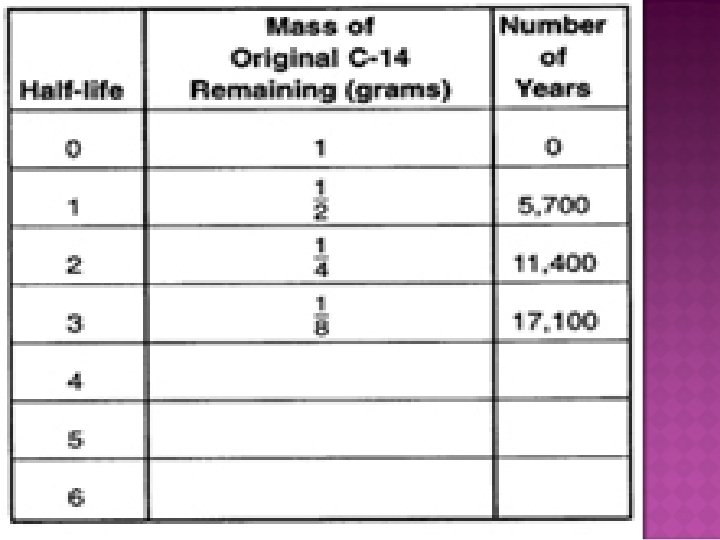

� The sample to be dated must contain a measurable amount of a radioactive element and its decay product � A sample with the remains of living things will likely contain carbon-14 � A geologist must choose the radioactive isotope whose half-life best measure the age of the sample � Example: Using carbon-14 can only get you back 50, 000 years, which is relatively very recent � More recent rocks wouldn’t have enough uranium -238 which can measure the oldest rocks on Earth

� These notes are based on pages 132 -133 in the RRB
- Slides: 11