Determines Number Common the element of protons CANNOT



� Determines � Number � Common the element of protons CANNOT change for an element charge notation is +1 � Mass (g) = 1. 673 x 10 -24 � Mass (amu)= 1. 0073 = 1

� Common charge notation is 0. No charge! � Mass (g) 1. 675 x 10 -24 (same mass as proton) � Mass (amu)= 1. 0082 = 1 � Number of neutrons CAN change for an element �ISOTOPES � Stabilizes nucleus by nuclear force

� Common Charge notation is -1 � Mass (g) of e- = 9. 109 x 10 -28 � Mass (amu)=. 0006 = 0 � Number of electrons CAN change for an element �IONS � Responsible for chemical reactivity, forms bonds
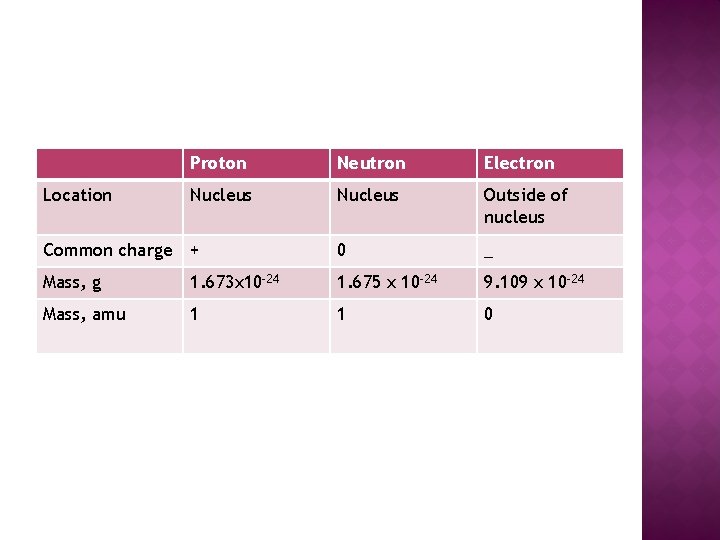
Proton Neutron Electron Nucleus Outside of nucleus Common charge + 0 _ Mass, g 1. 673 x 10 -24 1. 675 x 10 -24 9. 109 x 10 -24 Mass, amu 1 1 0 Location

� Which subatomic particle can NOT change for an element and gives it’s identity? � Which subatomic particle stabilizes the nucleus?

� Atomic Number: The number of protons in each atom of that element. � Unique to EACH element, never changes!! � Neutral atoms: proton #= electron # � Example: 1. 2. How many protons does Beryllium (Be) have? How many electrons? How many protons does Gold (Au) have?
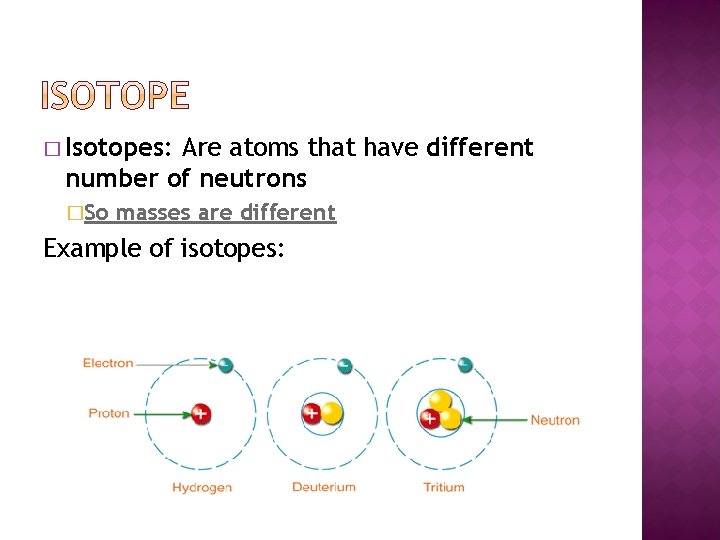
� Isotopes: Are atoms that have different number of neutrons �So masses are different Example of isotopes:

� Mass Number: The total number of protons and neutrons in the nucleus

� Element- � What Mass number is the hyphen notation of lithium if the mass number is 4?

Common isotopes of carbon Carbon-12 Carbon-13 Carbon-14 How many protons do all the carbon isotopes have? How many neutrons do each of the carbon isotopes have?

�Atomic #= Proton #= Electron # (because we assume atoms are neutral) �Mass #= Atomic # + Neutron #
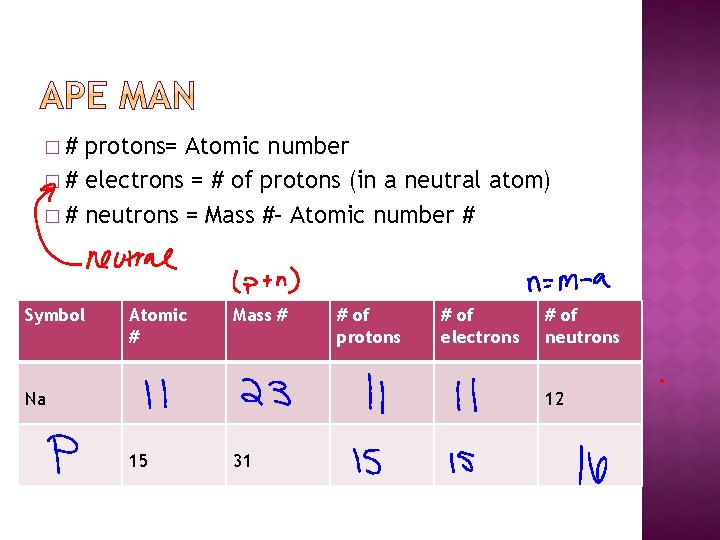
�# protons= Atomic number � # electrons = # of protons (in a neutral atom) � # neutrons = Mass #– Atomic number # Symbol Atomic # Mass # Na # of protons # of electrons # of neutrons 12 15 31

� Atoms are Neutral because they have the same number of electrons and protons �You will assume this unless indicated otherwise

� Ions : are when the electron number and proton number are different leading to a net charge. � NOTE: YOU WILL ALWAYS ASSUME ATOMS ARE NEUTRAL (protons = electrons) UNLESS TOLD OTHERWISE

� Average atomic mass is the weighted average of the atomic masses of the naturally occurring isotopes �DO NOT look at this number when trying to find mass #.

You must answer the question by yourself When you have it, write it down on a piece of paper Bring the worksheet you picked up in the back 1. 2. 3. 1. 4. Winning team gets 5 points Don’t single one person out on the other team each time.
- Slides: 18