Determination of Trace Metals Volatile Organic Compounds and


















- Slides: 31

Determination of Trace Metals, Volatile Organic Compounds, and Other Water Standards in WMU Drinking Water By: Tyler Walter Thesis Chair: Dr. Carla M. Koretsky Thesis Committee: Dr. Michael Barcelona, Dr. Steven Bertman

Background • U. S. Water Footprint = 2100 – 2500 m 3/capita/year • Among the highest in the world • EPA (Environmental Protection Agency) • DEQ (Department of Environmental Quality) • CDC (Center for Disease Control) • Kalamazoo City Water Department • Treatment processes (Pulsipher, 2011; Department of Public Service, 2011)

Objectives • Determine the concentrations of various contaminants in on campus drinking water • Trace metals (Cu, Zn, Fe, Mn, Ni, Pb and As) • Average hardness, p. H, E. coli levels, and concentrations of select pesticides (atrazine/simazine), NO 3 -, NO 2 - (as N), and Cl • A suite of volatile organic compounds (toluene, ethylbenzene, xylene, and chlorobenzene) • Compare the contaminant concentrations among select building • Influence of building age on concentration • Influence of water source distance from the buildings water supply entrance on concentration • Compare measured concentrations to EPA standards

Hypothesis • Concentrations of trace metals, volatile organic compounds, pesticides, NO 3 -, NO 2 -, and Cl will increase with increasing distance of the water source from the initial water supply point • Concentrations will also increase with increased building age • Levels of hardness, p. H, and bacteria will not be affected by distance from the initial water supply point • Levels will increase with building age

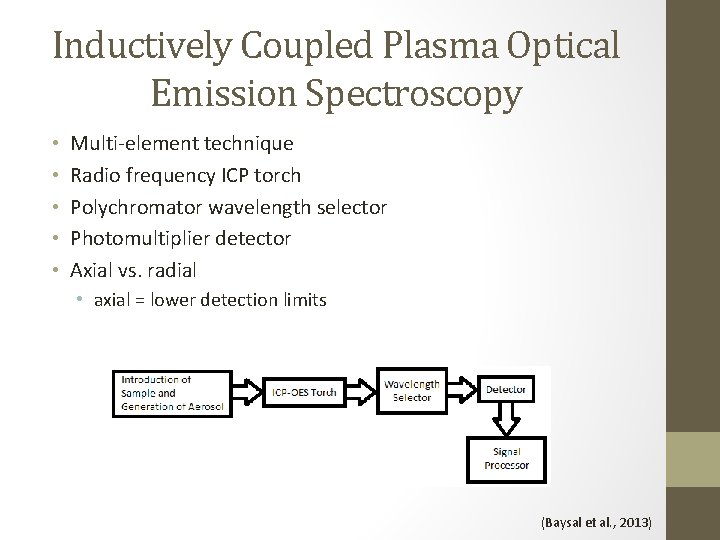
Inductively Coupled Plasma Optical Emission Spectroscopy • • • Multi-element technique Radio frequency ICP torch Polychromator wavelength selector Photomultiplier detector Axial vs. radial • axial = lower detection limits (Baysal et al. , 2013)

Watersafe® Drinking Water Test kits • Silver Lake Research Corporation (discovertesting. com) • Cheap, yet effective • Child safety • Lead levels in drinking water • Center for Disease Control (CDC) • Escherichia coli (City Water Test Kit, 2013)

Solid Phase Microextraction (SPME) • Direct (immersion) SPME vs. Headspace SPME • Fused Silica fiber • Absorbs target compounds • Split vs. Splitless (Pecoraino et al. , 2008)

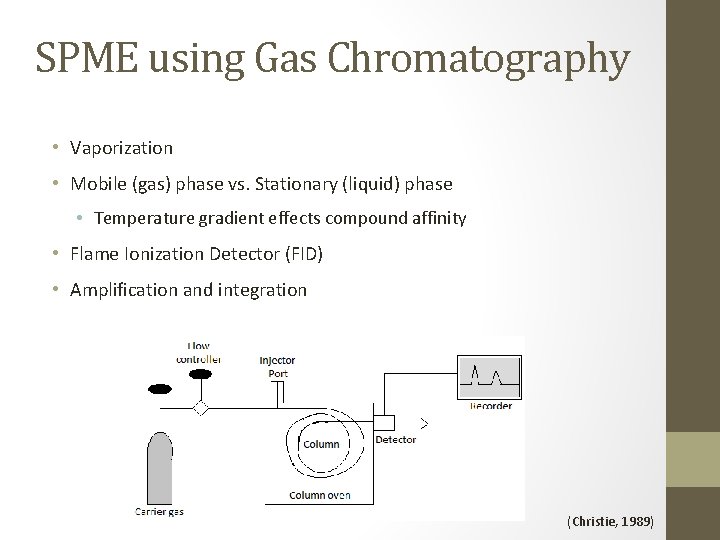
SPME using Gas Chromatography • Vaporization • Mobile (gas) phase vs. Stationary (liquid) phase • Temperature gradient effects compound affinity • Flame Ionization Detector (FID) • Amplification and integration (Christie, 1989)

Sample Collection Sources • Locations: • Davis Hall (1954) • • ground floor drinking fountain e* first floor dorm room faucet d* second floor lounge faucet third floor dorm room faucet d • The Chemistry Building (2007) • First floor drinking fountain ed* • Second floor drinking fountain • Third floor drinking fountain d* • The Dalton Center (1982) • First floor drinking fountain (NE) e • First floor drinking fountain (SE)* • Second floor drinking fountain (NE) d* e = entry point d = duplicate * = Watersafe® Source

Sample Collection • ICP-OES • 20 m. L polyvinyl vials • Acidified with 5% nitric acid • Watersafe Drinking Water Test • On site sample collection and analysis • SPME using Gas Chromatography • 20 m. L vials • Neoprene-containing cap

ICP-OES Analysis • Trace metal solutions – 0, 25, 50, 100, 250, 500, 1000 ppb • Cu, Zn, Fe, Mn, Ni, and As • Yttrium used as internal standard • Acidified (5% nitric acid) • Diluted with Ultrapure water • Axial Mode

Watersafe® Drinking Water Test • Bacteria Test – growth medium test • Lead/Pesticides Test – indicator test • Nitrate/Nitrite Test – colorimetric test • p. H/hardness/chlorine Test – colorimetric test (City Water Test Kit, 2013)

SPME Calibration Standards • 20 ppm stock solutions: • Toluene (99. 97% purity) • Ethylbenzene/Total xylene (≥ 98. 5 % purity) • 25 ppm stock solution: • Chlorobenzene (100% purity) • ~1 m. L of solution added to each vial • Magnetic stir bar added

SPME and GC Parameters • • • 40°C thermostatic bath (5 min) 85 m Carboxen/polydimethylsiloxane fiber 20 min absorption (Headspace SPME method) GC inlet (250°C) for 5 min desorption in splitless mode GC column: • • 30 m long Coated with 0. 32 mm I. D. (internal diameter) 1. 5 µm thick Maximum temperature: 260°C • Temperature Program: • 40°C initial temperature (2 min) • Rises 8°C/min until reaching 210°C • Carrier gas: • Helium with 50. 8 m. L/min flow rate • Average velocity of 29 cm/s (Pecoraino et al. , 2008)

ASME/ASTM Piping Standards • ASTM Piping Regulations: • Galvanized Pipe (A 53/A 53 M-12): • Contains Fe, C, Mn, P, S, Cu, Ni, Cr, Mo, V • Zn-coating • Older building = more corrosion (higher Zn concentration) • Copper Pipe (B 88 -09): • Contains Cu (w/Au) and trace amounts of P • Plastic Piping: • CPVC (chlorinated polyvinyl chloride) • Schedule 40 rating (ASTM, 2009; ASTM, 2012)

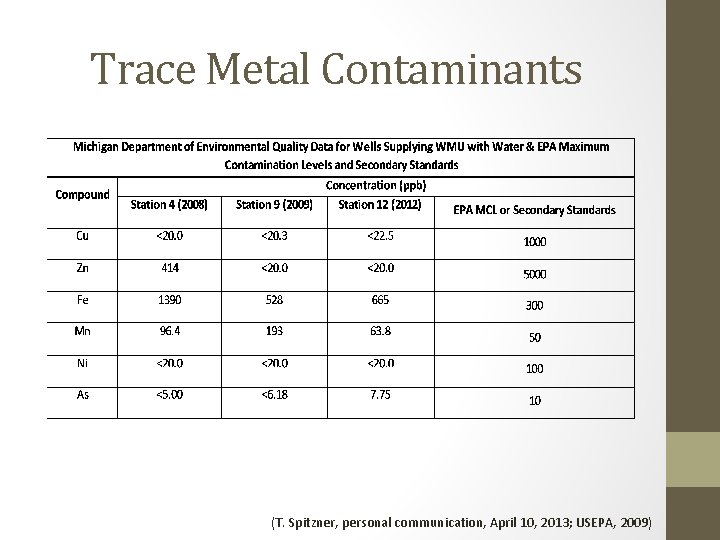
Trace Metal Contaminants (T. Spitzner, personal communication, April 10, 2013; USEPA, 2009)

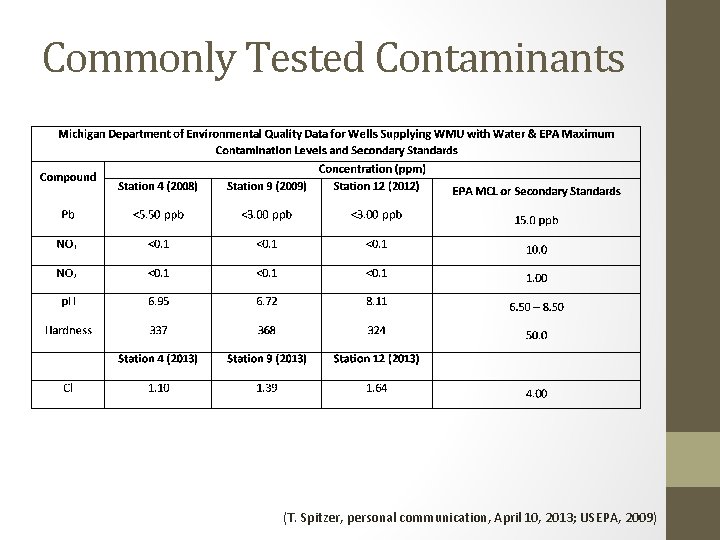
Commonly Tested Contaminants (T. Spitzer, personal communication, April 10, 2013; USEPA, 2009)

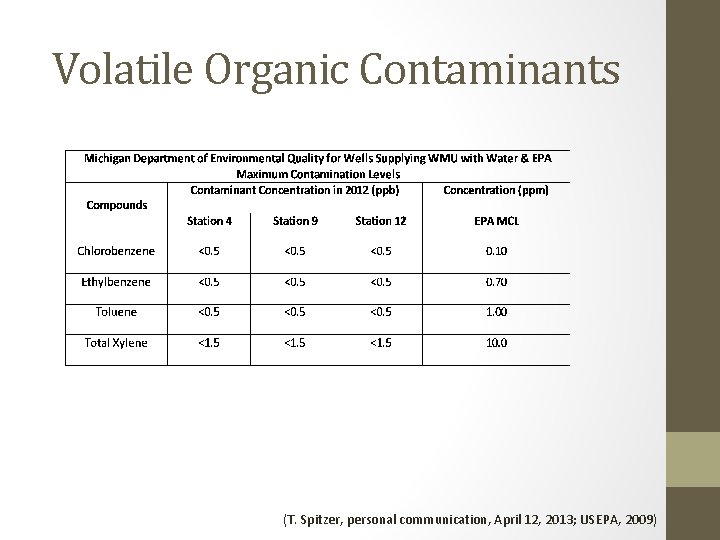
Volatile Organic Contaminants (T. Spitzer, personal communication, April 12, 2013; USEPA, 2009)

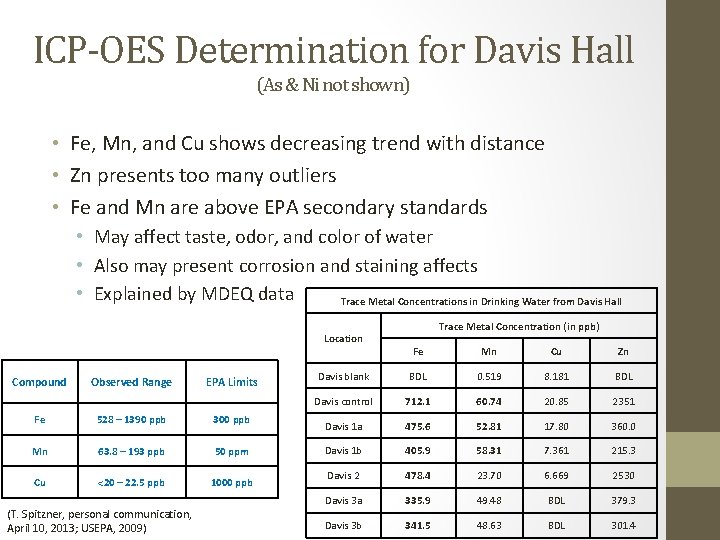
ICP-OES Determination for Davis Hall (As & Ni not shown) • Fe, Mn, and Cu shows decreasing trend with distance • Zn presents too many outliers • Fe and Mn are above EPA secondary standards • May affect taste, odor, and color of water • Also may present corrosion and staining affects • Explained by MDEQ data Trace Metal Concentrations in Drinking Water from Davis Hall Location Compound Observed Range EPA Limits Fe 528 – 1390 ppb 300 ppb Mn 63. 8 – 193 ppb 50 ppm Cu <20 – 22. 5 ppb 1000 ppb (T. Spitzner, personal communication, April 10, 2013; USEPA, 2009) Trace Metal Concentration (in ppb) Fe Mn Cu Zn Davis blank BDL 0. 519 8. 181 BDL Davis control 712. 1 60. 74 20. 85 2351 Davis 1 a 475. 6 52. 81 17. 80 360. 0 Davis 1 b 405. 9 58. 31 7. 361 215. 3 Davis 2 478. 4 23. 70 6. 669 2530 Davis 3 a 335. 9 49. 48 BDL 379. 3 Davis 3 b 341. 5 48. 63 BDL 301. 4

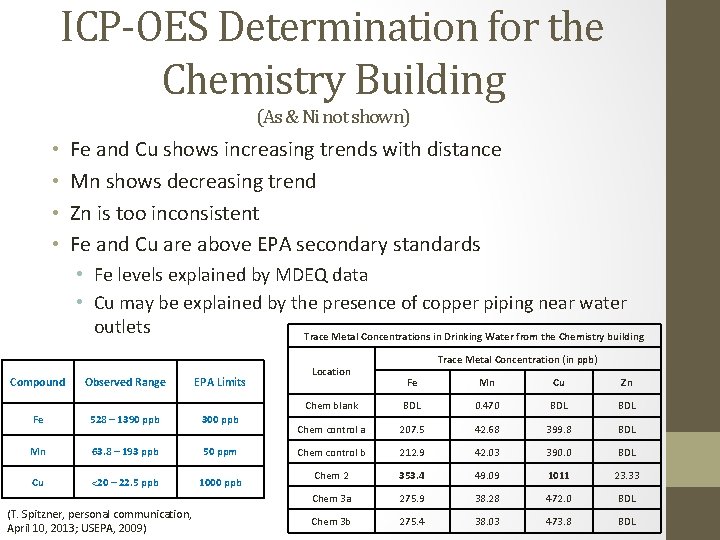
ICP-OES Determination for the Chemistry Building (As & Ni not shown) • • Fe and Cu shows increasing trends with distance Mn shows decreasing trend Zn is too inconsistent Fe and Cu are above EPA secondary standards • Fe levels explained by MDEQ data • Cu may be explained by the presence of copper piping near water outlets Trace Metal Concentrations in Drinking Water from the Chemistry building Compound Observed Range EPA Limits Fe 528 – 1390 ppb 300 ppb Mn 63. 8 – 193 ppb 50 ppm Cu <20 – 22. 5 ppb 1000 ppb (T. Spitzner, personal communication, April 10, 2013; USEPA, 2009) Location Trace Metal Concentration (in ppb) Fe Mn Cu Zn Chem blank BDL 0. 470 BDL Chem control a 207. 5 42. 68 399. 8 BDL Chem control b 212. 9 42. 03 390. 0 BDL Chem 2 353. 4 49. 09 1011 23. 33 Chem 3 a 275. 9 38. 28 472. 0 BDL Chem 3 b 275. 4 38. 03 473. 8 BDL

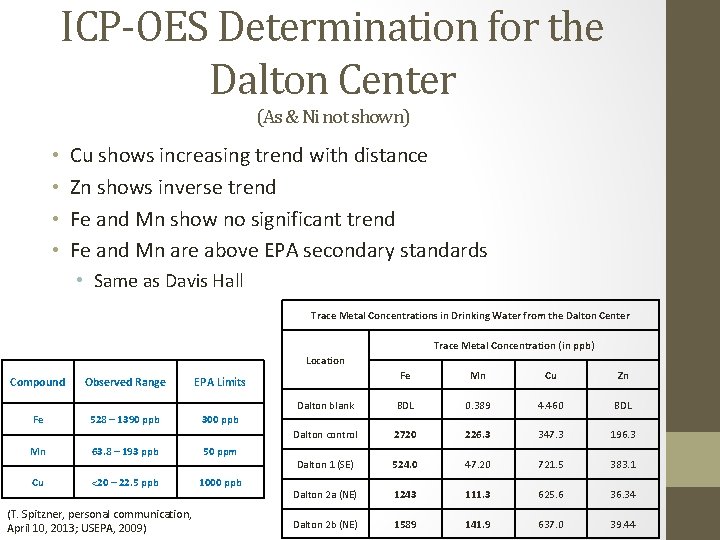
ICP-OES Determination for the Dalton Center (As & Ni not shown) • • Cu shows increasing trend with distance Zn shows inverse trend Fe and Mn show no significant trend Fe and Mn are above EPA secondary standards • Same as Davis Hall Trace Metal Concentrations in Drinking Water from the Dalton Center Trace Metal Concentration (in ppb) Location Compound Observed Range EPA Limits Fe 528 – 1390 ppb 300 ppb Mn 63. 8 – 193 ppb 50 ppm Cu <20 – 22. 5 ppb 1000 ppb (T. Spitzner, personal communication, April 10, 2013; USEPA, 2009) Fe Mn Cu Zn Dalton blank BDL 0. 389 4. 460 BDL Dalton control 2720 226. 3 347. 3 196. 3 Dalton 1 (SE) 524. 0 47. 20 721. 5 383. 1 Dalton 2 a (NE) 1243 111. 3 625. 6 36. 34 Dalton 2 b (NE) 1589 141. 9 637. 0 39. 44

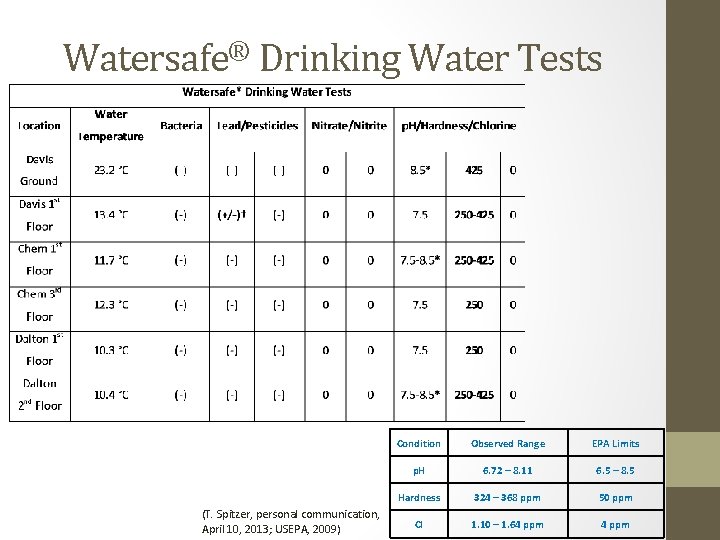
Watersafe® Drinking Water Tests (T. Spitzer, personal communication, April 10, 2013; USEPA, 2009) Condition Observed Range EPA Limits p. H 6. 72 – 8. 11 6. 5 – 8. 5 Hardness 324 – 368 ppm 50 ppm Cl 1. 10 – 1. 64 ppm

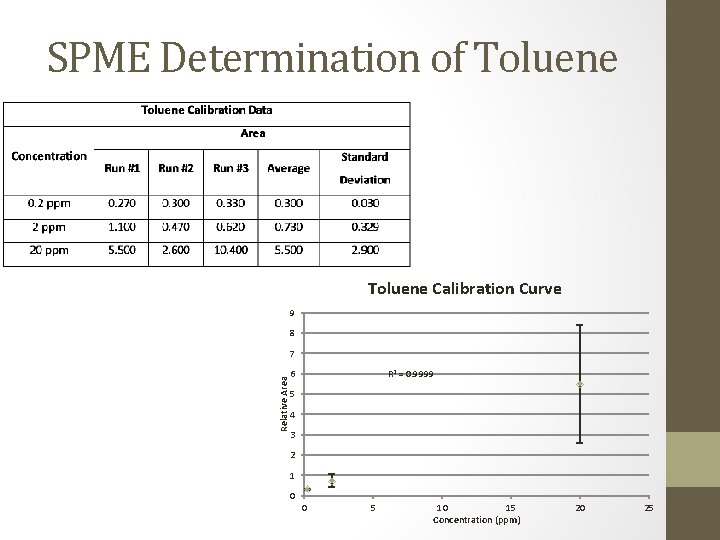
SPME Determination of Toluene Calibration Curve 9 8 Relative Area 7 R 2 = 0. 9999 6 5 4 3 2 1 0 0 5 10 15 Concentration (ppm) 20 25

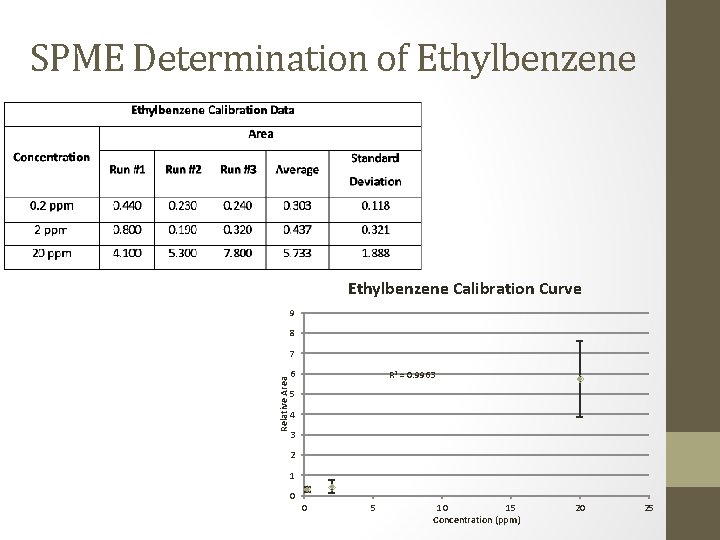
SPME Determination of Ethylbenzene Calibration Curve 9 8 Relative Area 7 6 R 2 = 0. 9963 5 4 3 2 1 0 0 5 10 15 Concentration (ppm) 20 25

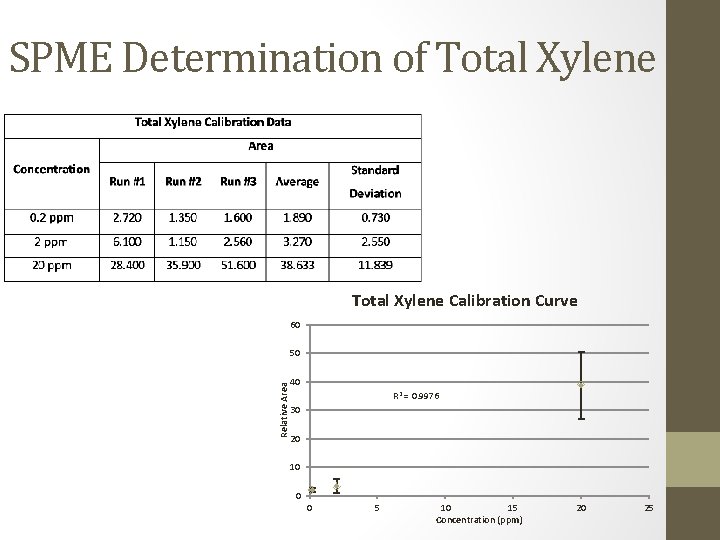
SPME Determination of Total Xylene Calibration Curve 60 Relative Area 50 40 R 2 = 0. 9976 30 20 10 0 0 5 10 15 Concentration (ppm) 20 25

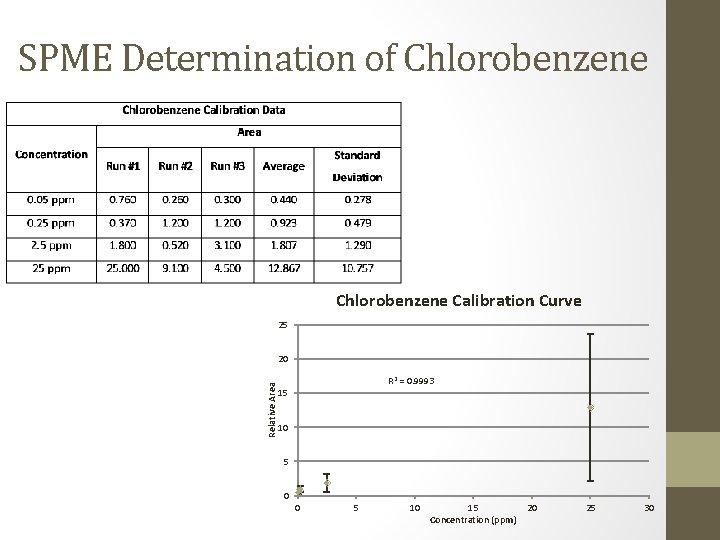
SPME Determination of Chlorobenzene Calibration Curve 25 Relative Area 20 R 2 = 0. 9993 15 10 5 0 0 5 10 15 Concentration (ppm) 20 25 30

SPME/GC Determination • Ethylbenzene, total xylene, and chlorobenzene • All samples were below detection limits • Ethylbenzene: below 0. 2 ppm • Total Xylene: below 0. 2 ppm • Chlorobenzene: below 0. 05 ppm • Toluene • Most samples were below detection limits • Toluene: below 0. 2 ppm • 4 samples produced peaks

SPME: Toluene Determination • 4 samples produced peaks • 2 were calculated to below detection limits • 1 was calculated to 0. 622 ppm (Davis 1) • Below EPA limits • 1 was calculated to 1. 115 ppm (Dalton 1 NE) • Above EPA limits, warranted further testing • Second test produced no peaks • Possibly explained by evaporation over time • Chronic consumption produces serious health affects

Conclusions • Some slight trends observed between distance from initial water supply and concentration • Most samples showed inverse trends • Contradicts hypothesis • Zinc showed strong trend between concentration and building age • Increased age = increased corrosion • No other contaminants showed any trends • Contradicts hypothesis • Several compounds were above EPA limits and standards • Most were secondary standards explained by MDEQ data • Presence of toluene above EPA limits • Absence in second test may be due to evaporation over time

Recommendations • Monitor water temperature more closely • Water temperature for ICP-OES and SPME analysis was never measured • Work with a partner • Most of the Watersafe Drinking Water Test kit procedures involve comparative judgment • A second person could alleviate result bias • Fresh Samples for SPME analysis • Evaporation may have produced different data • Nearby GC detected target chemicals • Use more calibration points for SPME analysis

Are there any questions?