Detection of ESBLs Amp C David Livermore Health

Detection of ESBLs & Amp. C David Livermore Health Protection Agency, Colindale, London

Some premises • Growing resistance to 3 -gen cephs • Mostly ESBLs in E. coli & Klebsiella; Amp. C in Enterobacter, Citrobacter, Serratia… but not always • Identification of mechanism aids – Epidemiological investigation / control – Treatment choice – Recognition of the exceptional e. g. MBLs
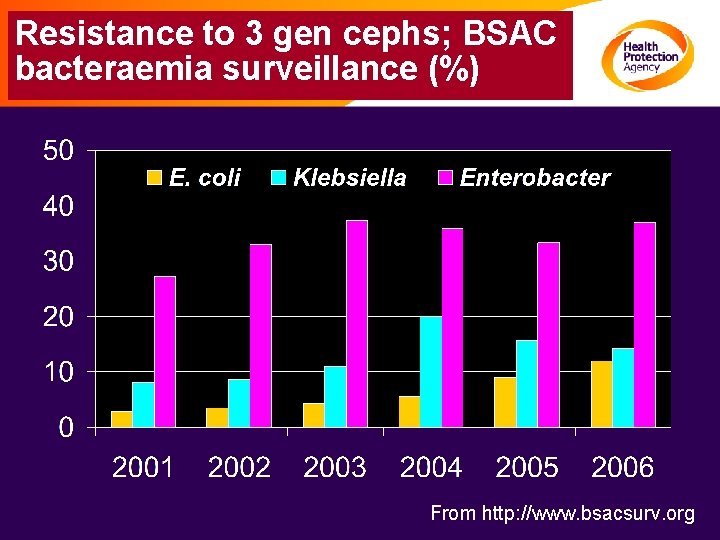
Resistance to 3 gen cephs; BSAC bacteraemia surveillance (%) From http: //www. bsacsurv. org
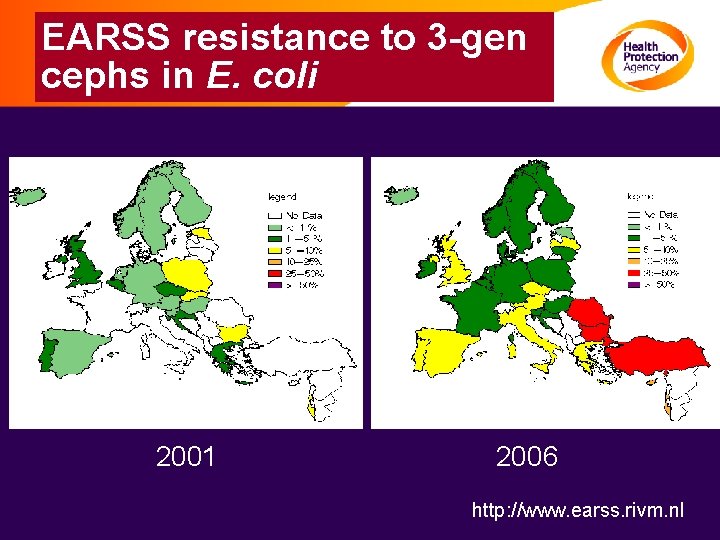
EARSS resistance to 3 -gen cephs in E. coli 2001 2006 http: //www. earss. rivm. nl

Detecting ESBL producers 2 steps: • Screen for resistance with an indicator ceph • Do confirmatory test on those found resistant
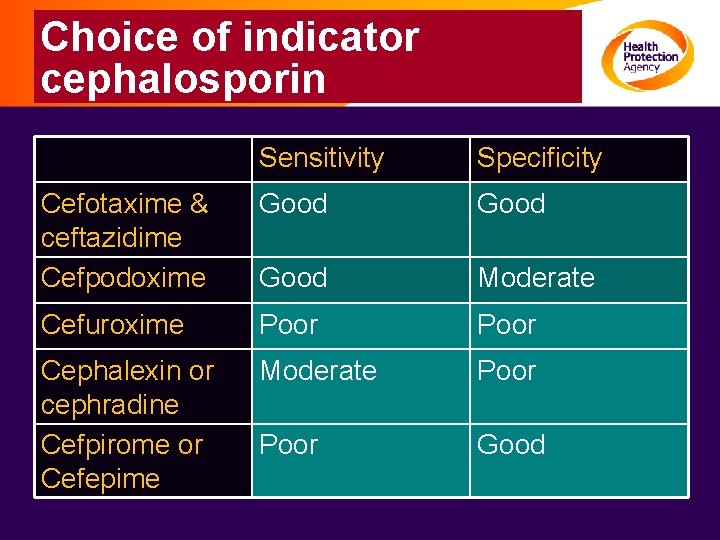
Choice of indicator cephalosporin Sensitivity Specificity Cefotaxime & ceftazidime Cefpodoxime Good Moderate Cefuroxime Poor Cephalexin or cephradine Cefpirome or Cefepime Moderate Poor Good

Detection of ESBLs: step 2 Seek ceph/clav synergy in ceph R isolates • Double disc • Combination disc • Etest See http: //www. hpa. org. uk


Combination discs Disc with cephalosporin + clavulanic acid Disc with cephalosporin alone
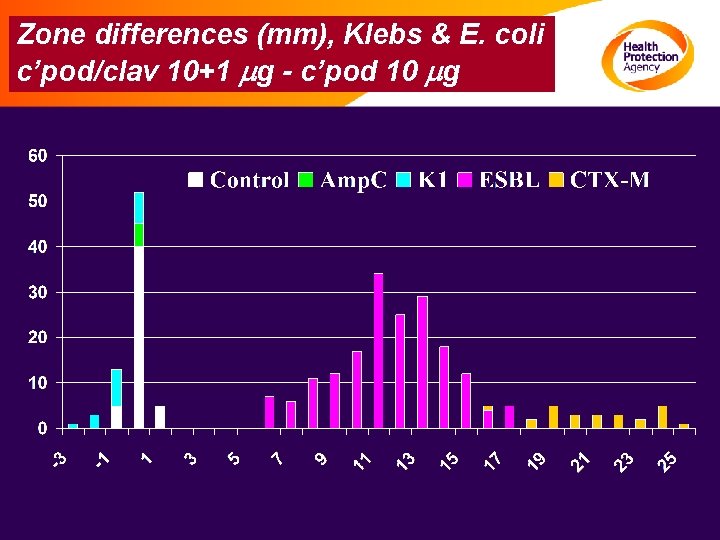
Zone differences (mm), Klebs & E. coli c’pod/clav 10+1 mg - c’pod 10 mg
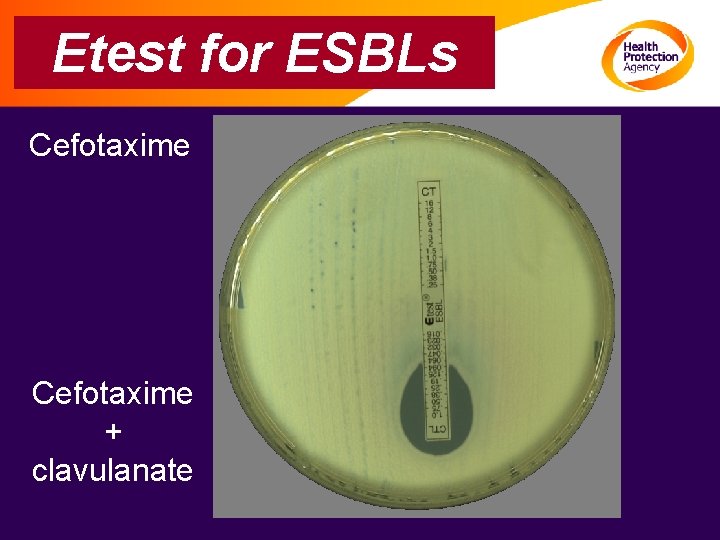
Etest for ESBLs Cefotaxime + clavulanate
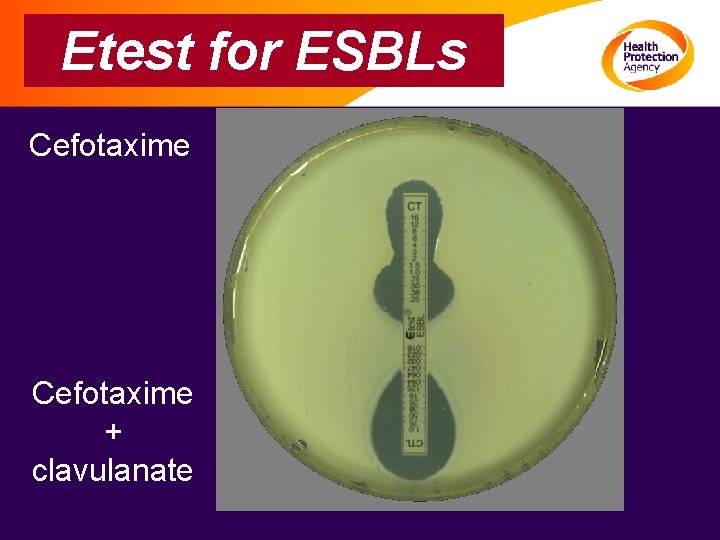
Etest for ESBLs Cefotaxime + clavulanate

ESBL confirmatory tests Pro Contra Double disc Cheap Best disc spacing varies with strain Combination disc Etest Cheap, sensitive & specific Batch variation; Sensitive More expensive; False +ve's with K 1 in K. oxytoca Internally controlled Controls critical

Controls for ESBL tests • +ve E. coli with TEM-3, -10 & CTX-M-15 available as NCTC 13351, 13352, 13353 No one control is perfect… and these have high levels of enzyme whilst some clinical isolate have very low levels • –ve E. coli (e. g. NCTC 10418) Critical for combination discs; should give equal zones irrespective of clavulanate

Further investigating ESBLs • Multiplex PCR for 5 bla. CTX-M groups* • TEM & SHV mutants require sequencing • Beware…. Isolates may have >1 enzyme –e. g. Classical TEM / SHV + TEM / SHV ESBL –Many with CTX-M-15 also have OXA-1 & TEM-1 • Isoelectric focusing gives fullest picture

ESBL tests for Amp. C inducible species • Methods optimised for E. coli & Klebsiella • More difficult with Enterobacter – clavulanate induces Amp. C; hides ESBL • Advice is to do synergy test (NOT SCREEN) with 4 th gen ceph; specificity good, sensitivity moderate BSAC bacteraemia: c. 25% Ceph. R Enterobacter have ESBL, not Amp. C…. . Probably an underestimate

Bacteria not to test for ESBLs Acinetobacters – Often S to clavulanate alone S. maltophilia – +ve result by inhibition of L-2 chromosomal b-lactamase, ubiquitous in the species

Suspect derepressed / plasmid Amp. C if: • Resistant 3 -gen cephs, NOT cefepime & cefpirome • Resistant to cefoxitin (but more ESBL producers R, too, nowadays) • No ceph/clav synergy
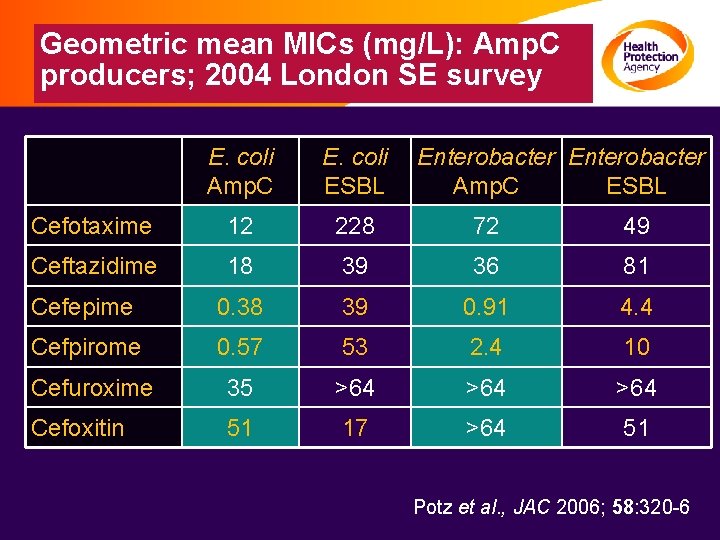
Geometric mean MICs (mg/L): Amp. C producers; 2004 London SE survey E. coli Amp. C E. coli ESBL Enterobacter Amp. C ESBL Cefotaxime 12 228 72 49 Ceftazidime 18 39 36 81 Cefepime 0. 38 39 0. 91 4. 4 Cefpirome 0. 57 53 2. 4 10 Cefuroxime 35 >64 >64 Cefoxitin 51 17 >64 51 Potz et al. , JAC 2006; 58: 320 -6

Some wrinkles… • Amp. C-derepressed M. morganii are S to pip/tazo • Amp. C derepressed Serratia are S to ceftazidime • Cefoxitin R an unreliable marker for Providencia, Morganella & Serratia spp. – Inducible & derepressed strains may appear I or S • Amp. C derepressed P. aeruginosa tend to be S to carbenicillin / efflux mutants are R

Confirmatory tests for Amp. C • Seek cefotaxime/cloxacillin synergy – Cefotaxime MIC +100 mg/L cloxacillin – Zones of cefotaxime 30 mg discs on agar + 100 mg/L cloxacillin – No agreed interpretive standards • Can also use phenylboronic acid as inhibitor
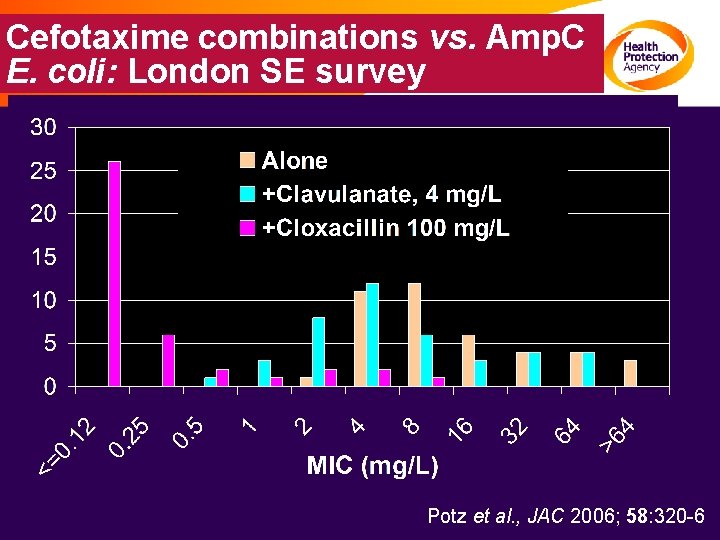
Cefotaxime combinations vs. Amp. C E. coli: London SE survey Potz et al. , JAC 2006; 58: 320 -6
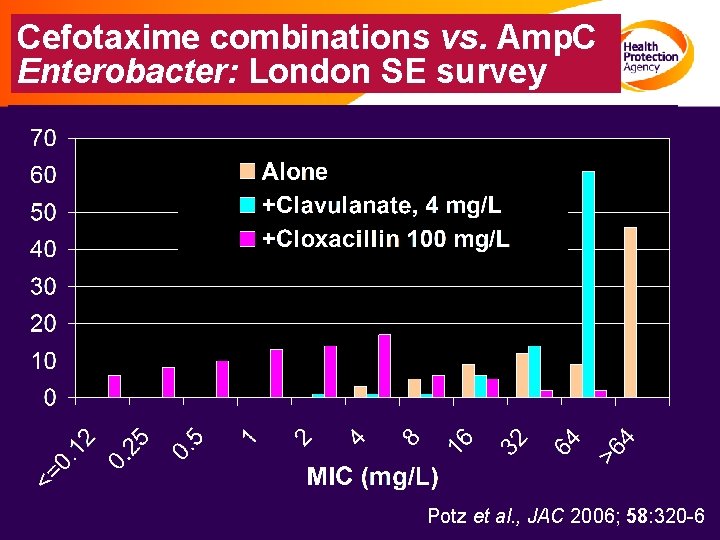
Cefotaxime combinations vs. Amp. C Enterobacter: London SE survey Potz et al. , JAC 2006; 58: 320 -6
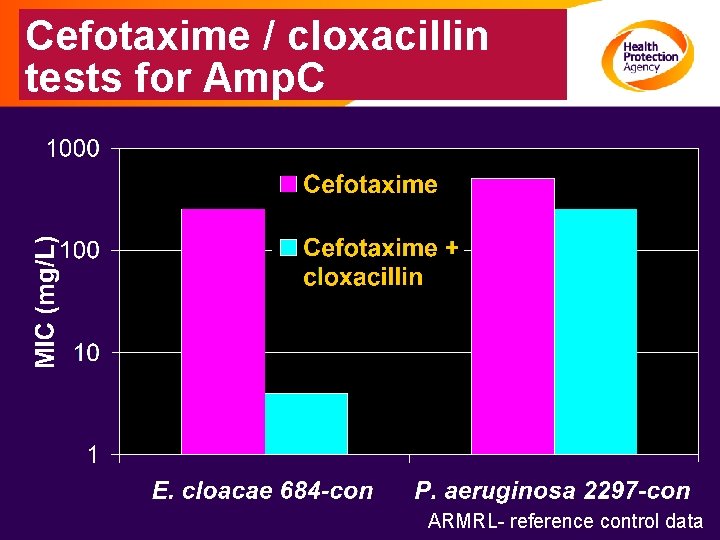
Cefotaxime / cloxacillin tests for Amp. C ARMRL- reference control data
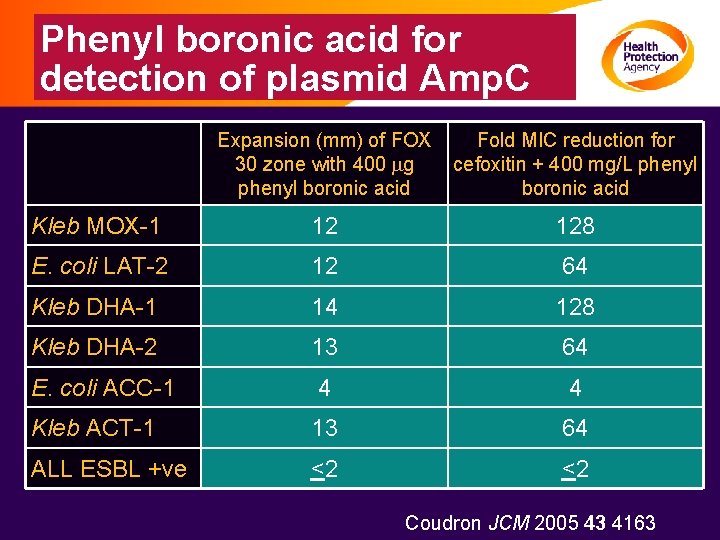
Phenyl boronic acid for detection of plasmid Amp. C Expansion (mm) of FOX 30 zone with 400 mg phenyl boronic acid Fold MIC reduction for cefoxitin + 400 mg/L phenyl boronic acid Kleb MOX-1 12 128 E. coli LAT-2 12 64 Kleb DHA-1 14 128 Kleb DHA-2 13 64 E. coli ACC-1 4 4 Kleb ACT-1 13 64 ALL ESBL +ve <2 <2 Coudron JCM 2005 43 4163
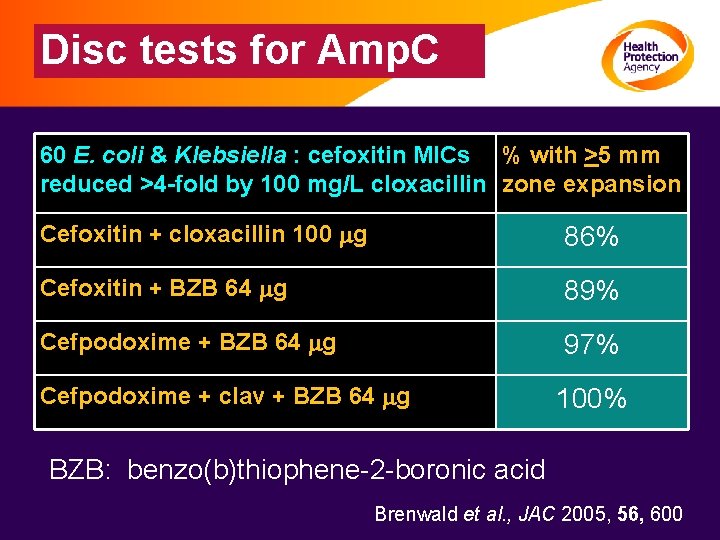
Disc tests for Amp. C 60 E. coli & Klebsiella : cefoxitin MICs % with >5 mm reduced >4 -fold by 100 mg/L cloxacillin zone expansion Cefoxitin + cloxacillin 100 mg 86% Cefoxitin + BZB 64 mg 89% Cefpodoxime + BZB 64 mg 97% Cefpodoxime + clav + BZB 64 mg 100% BZB: benzo(b)thiophene-2 -boronic acid Brenwald et al. , JAC 2005, 56, 600

3 -D test for Amp. Cs Plate seeded with cefoxitin S indicator strain Cut cross in agar, heavily inoculated with test strain Cefoxitin disc Looks for distortion where cross intersects the cefoxitin zone

Clover leaf (3 dimensional) test for Amp. C Test strain E. cloacae, Amp. C derepressed Indicator E. coli NCTC 10418 Disc Cefoxitin 30 mg
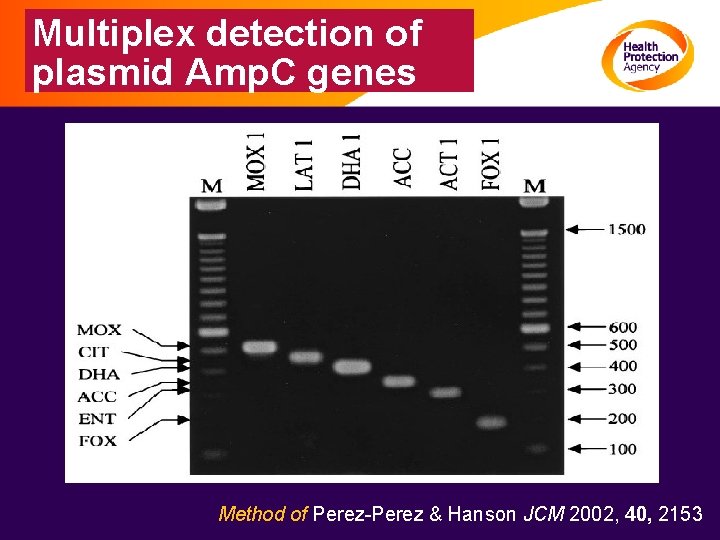
Multiplex detection of plasmid Amp. C genes Method of Perez-Perez & Hanson JCM 2002, 40, 2153

Amp. C commercial tests ROSCO- ‘research only’ : high content (500 mg) cloxacillin & boronic acid discs for double disc synergy tests AB Biodisk- evaluating double-ended cefotetan or cefoxitin plus cloxacillin or boronic acid ETests

Hyperproduction of K 1 enzyme • Unique to K. oxytoca, chromosomal • Indole +ve Klebsiella – R cefuroxime, aztreonam, pip / tazo, ceftriaxone – Borderline (S/I/R) to cefotaxime – S to ceftazidime & carbapenems – Weak cefotaxime or cefepime/clav synergy
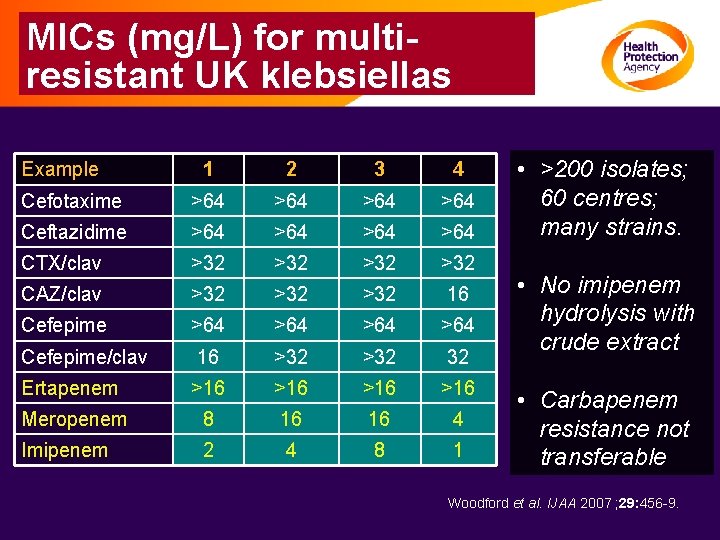
MICs (mg/L) for multiresistant UK klebsiellas Example 1 2 3 4 Cefotaxime >64 >64 Ceftazidime >64 >64 CTX/clav >32 >32 CAZ/clav >32 >32 16 Cefepime >64 >64 Cefepime/clav 16 >32 32 Ertapenem >16 >16 Meropenem 8 16 16 4 Imipenem 2 4 8 1 • >200 isolates; 60 centres; many strains. • No imipenem hydrolysis with crude extract • Carbapenem resistance not transferable Woodford et al. IJAA 2007 ; 29: 456 -9.
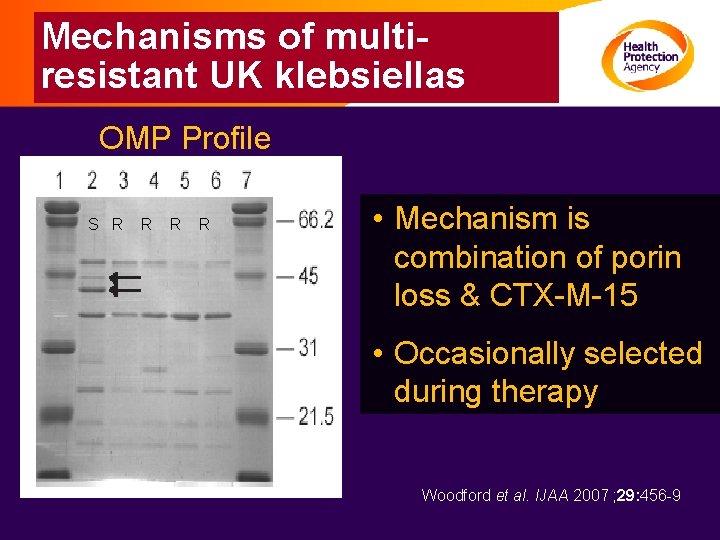
Mechanisms of multiresistant UK klebsiellas OMP Profile S R R • Mechanism is combination of porin loss & CTX-M-15 • Occasionally selected during therapy Woodford et al. IJAA 2007 ; 29: 456 -9

Acquired carbapenemases • KPC – Class A, 4 variants – Spreading world-wide, 2 cases in UK…. so far – Often clonal, mostly Klebsiella, Enterobacter • Metallo’s – Class B, VIM, IMP families, also SPM, SIM, GIM – Scattered, mostly non-fermenters – >100 UK in since 2001, mostly VIM+ P. aeruginosa

Carbapenemase or not. . . • KPC…. clearest R to ertapenem; no synergy in clavulanate, cloxacillin, boronic acid or EDTA tests – Easy to confuse with combination of ESBL + impermeability • Metallo’s…. Suspect if isolate has reduced carbapenem susceptibility, reversed by ESBL… But – Frank carbapenem resistance not always seen – EDTA tests not specific… many false +ves – Spare aztreonam, may be affected by other mechanisms

A problem in Bolzano • 209 Ceph R Enterobacteriaceae, most had ESBLs • 24 lacked ceph / clav synergy- mix of E. coli, pneumoniae, K. oxytoca, Citrobacter K. • Imipenem MICs 2 - >32 mg/L, mostly 4 -8 mg/L – Meropenem, ertapenem MICs lower than imipenem – Imipenem + EDTA MICs 0. 12 -1 mg/L • All had bla. VIM; mix of clonal & non-clonal!!! • 19 R to aztreonam--- had CTX-M ----5 susceptible Aschbacher et al, submitted

Cica b-Test (Mast) • Examine hydrolysis of chromogenic oxyimino ceph, HMRZ-86 - yellow to red • If +ve, test inhibition IN SEQUENCE by: – Sodium mercaptoacetic acid – MBL – Clavulanic acid – Class A / ESBL – Benzo-thiophene-2 -boronic acid – Amp. C • Count first positive result
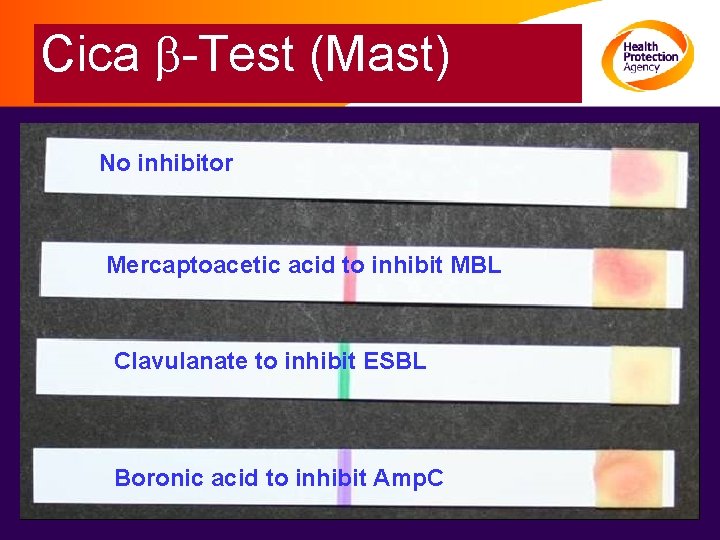
Cica b-Test (Mast) No inhibitor Mercaptoacetic acid to inhibit MBL Clavulanate to inhibit ESBL Boronic acid to inhibit Amp. C
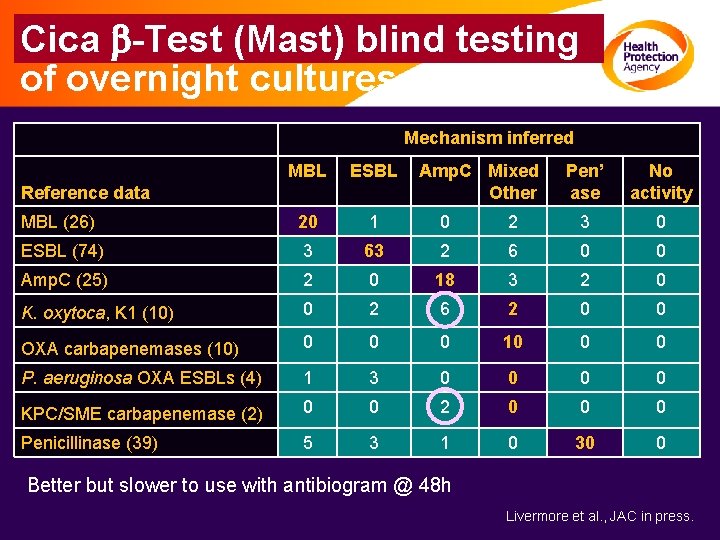
Cica b-Test (Mast) blind testing of overnight cultures Mechanism inferred MBL ESBL Pen’ ase No activity MBL (26) 20 1 0 2 3 0 ESBL (74) 3 63 2 6 0 0 Amp. C (25) 2 0 18 3 2 0 K. oxytoca, K 1 (10) 0 2 6 2 0 0 OXA carbapenemases (10) 0 0 0 10 0 0 P. aeruginosa OXA ESBLs (4) 1 3 0 0 KPC/SME carbapenemase (2) 0 0 2 0 0 0 Penicillinase (39) 5 3 1 0 30 0 Reference data Amp. C Mixed Other Better but slower to use with antibiogram @ 48 h Livermore et al. , JAC in press.

Summary • Labs should be able to recognise ESBL producers – Even among Enterobacters – Ref lab will look at difficult cases • Labs should be able to recognise Amp. C derepressed strains & those with plasmid Amp. C • Enterobacteriaceae with reduced carbapenem susceptibility need reference investigation • New tests being developed
- Slides: 40