Detection of Biological Material and Integration into Casework

Detection of Biological Material and Integration into Casework Workflow – how to get research implemented into casework Rachel Fleming 03/05/2018
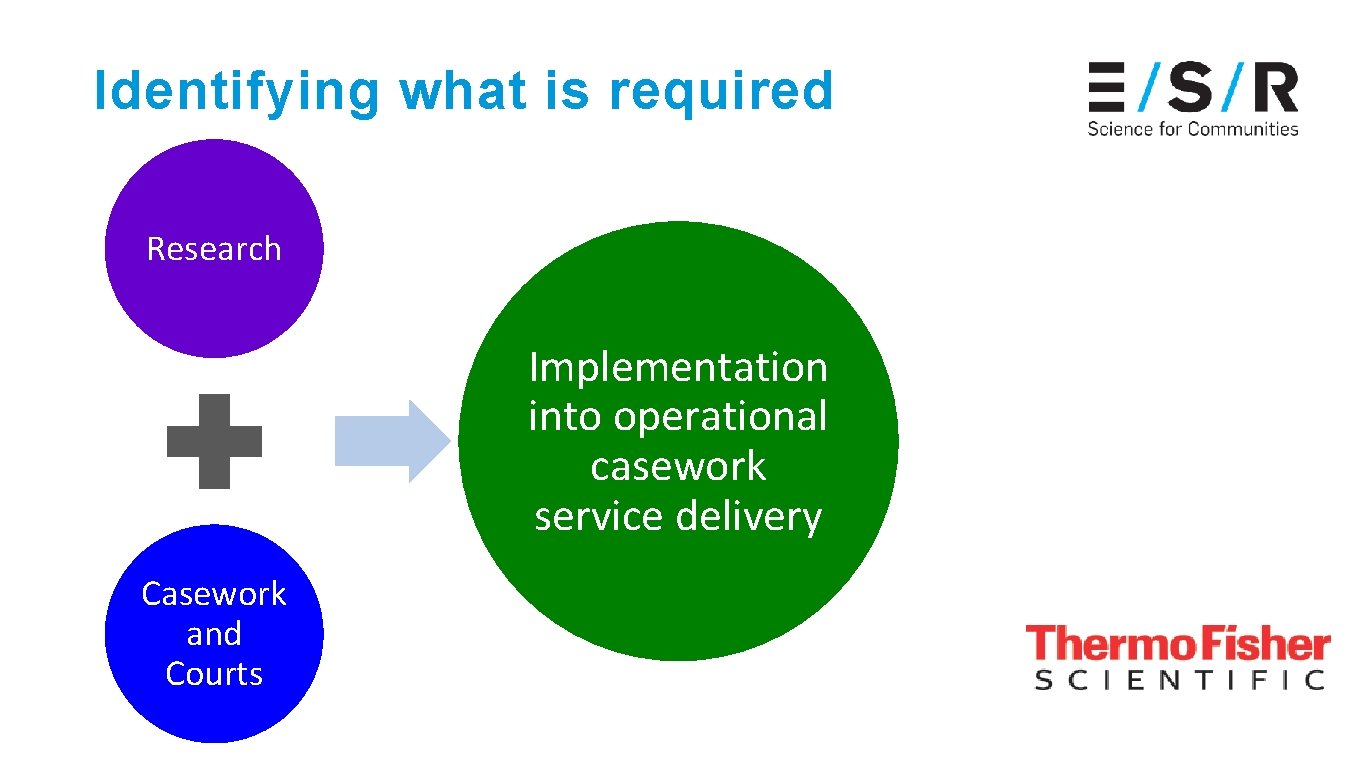
Identifying what is required Research Implementation into operational casework service delivery Casework and Courts
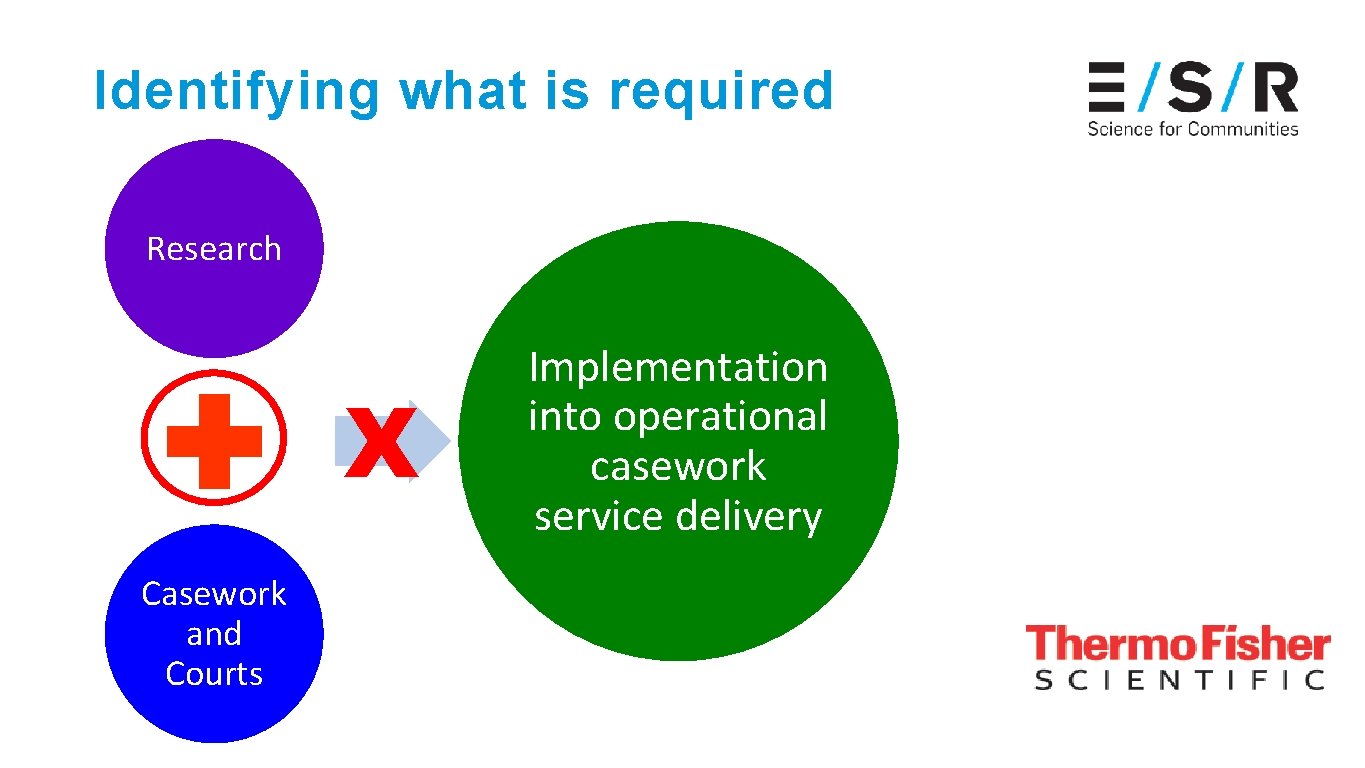
Identifying what is required Research X Casework and Courts Implementation into operational casework service delivery

Research
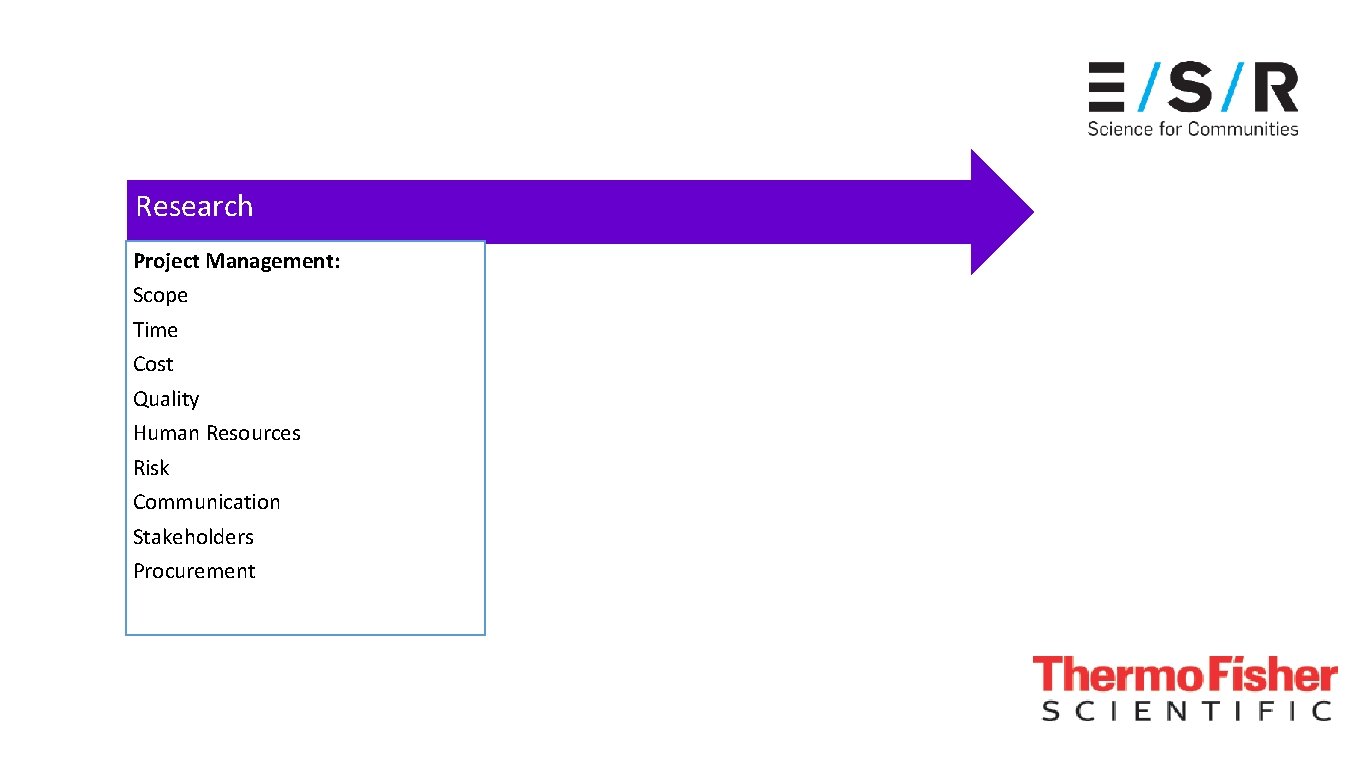
Research Project Management: Scope Time Cost Quality Human Resources Risk Communication Stakeholders Procurement
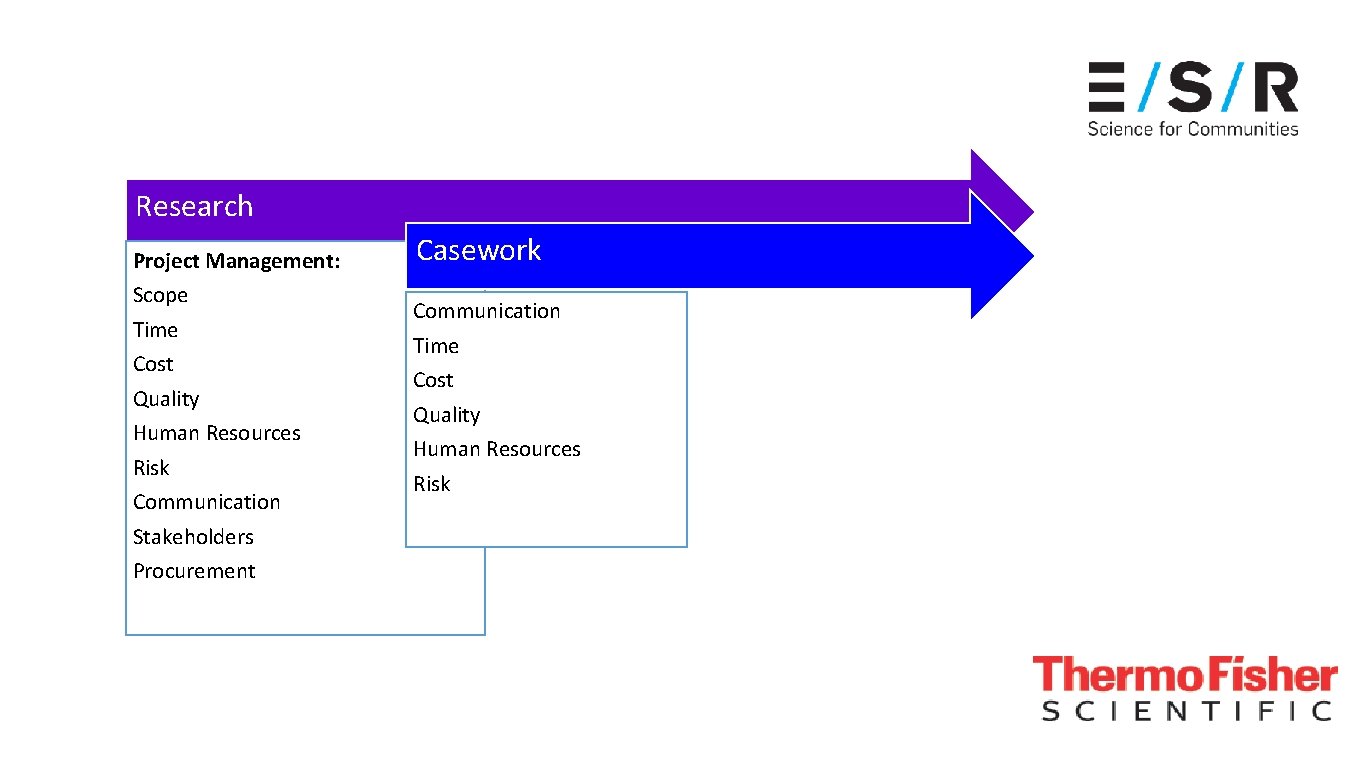
Research Project Management: Scope Time Cost Quality Human Resources Risk Communication Stakeholders Procurement Casework Communication Time Cost Quality Human Resources Risk
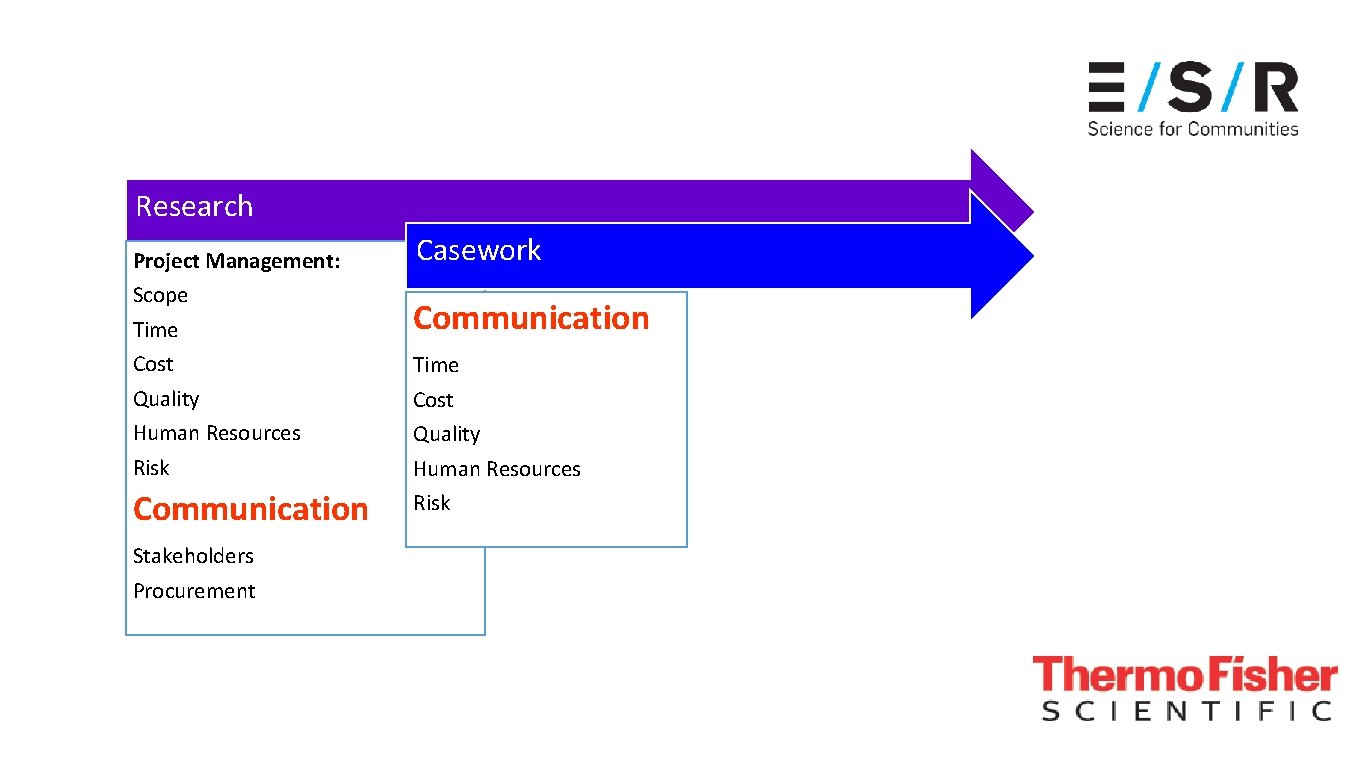
Research Project Management: Scope Time Cost Quality Human Resources Risk Communication Stakeholders Procurement Casework Communication Time Cost Quality Human Resources Risk
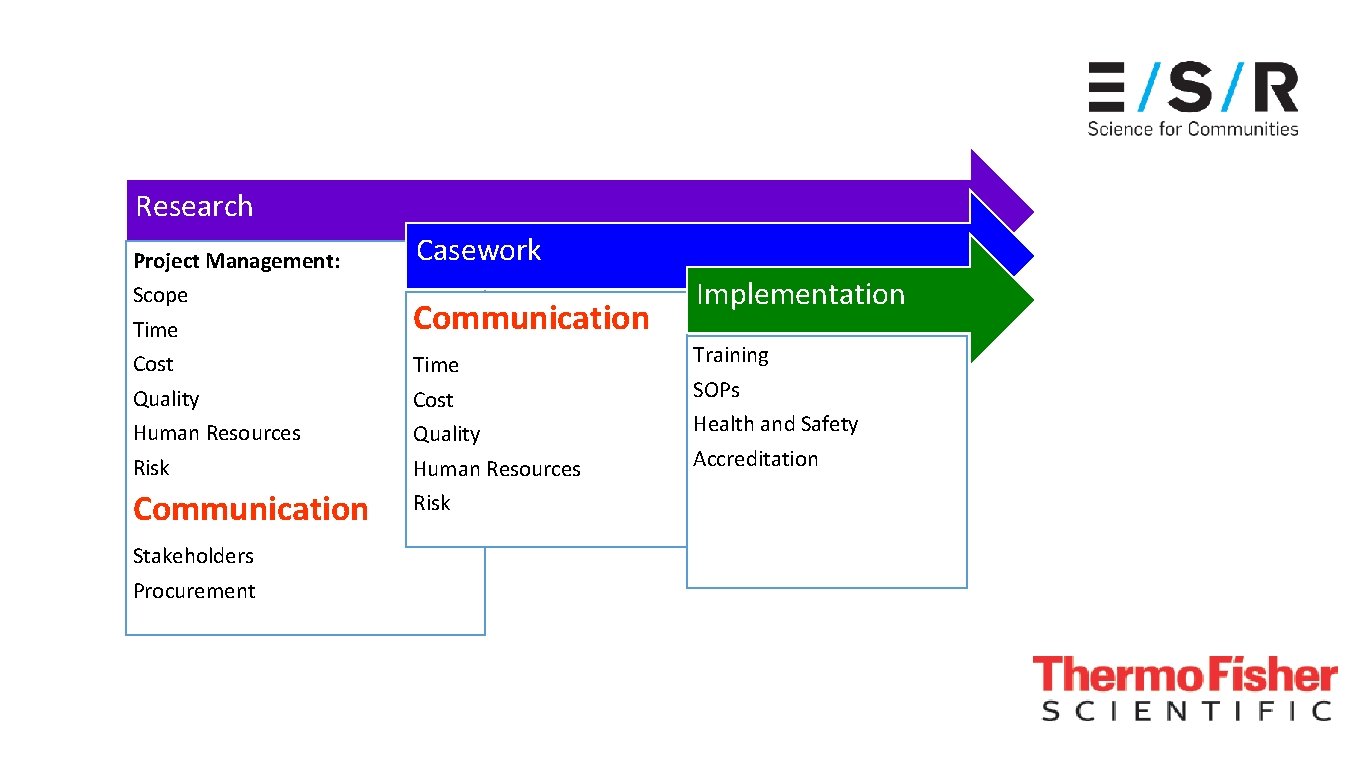
Research Project Management: Scope Time Cost Quality Human Resources Risk Communication Stakeholders Procurement Casework Communication Time Cost Quality Human Resources Risk Implementation Training SOPs Health and Safety Accreditation
Next generation of Cell. Typer Changes based on: • Casework experience • New research New equipment/technology (future-proofing)

Genetic analyzers Casework laboratory were starting their transition from 3130 x. L to 3500 x. L genetic analyzers

Genetic analyzers Cell. Typer 2
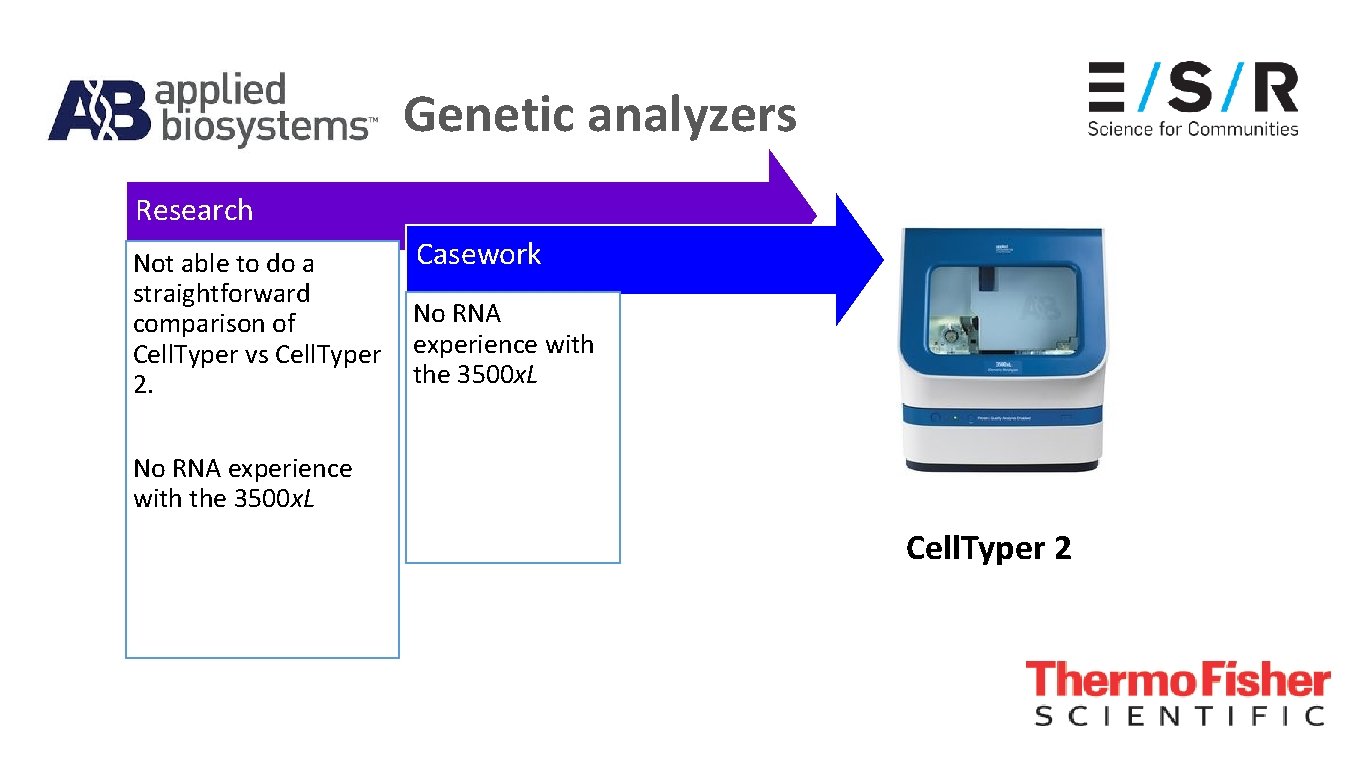
Genetic analyzers Research Not able to do a straightforward comparison of Cell. Typer vs Cell. Typer 2. Casework No RNA experience with the 3500 x. L Cell. Typer 2

Research • MPS research • Stable regions of RNA • Patricia Albani – Ph. D project Forensic Sci Int-Gen 20 (2016) 61 -70

Primer placement – stable RNA region vs non-stable region SLC 4 A 1
![Primer placement – stable RNA region vs non-stable region SLC 4 A 1 [RFU] Primer placement – stable RNA region vs non-stable region SLC 4 A 1 [RFU]](http://slidetodoc.com/presentation_image_h2/8b4acd7abd5360c0ffe98a000a0033af/image-15.jpg)
Primer placement – stable RNA region vs non-stable region SLC 4 A 1 [RFU] SLC 4 A 1 v 1 10000 SLC 4 A 1 v 3 8000 6000 4000 2000 0 BA 2 BC 2 BD 1 BE 1 BH 1 SLC 4 A 1 v 2
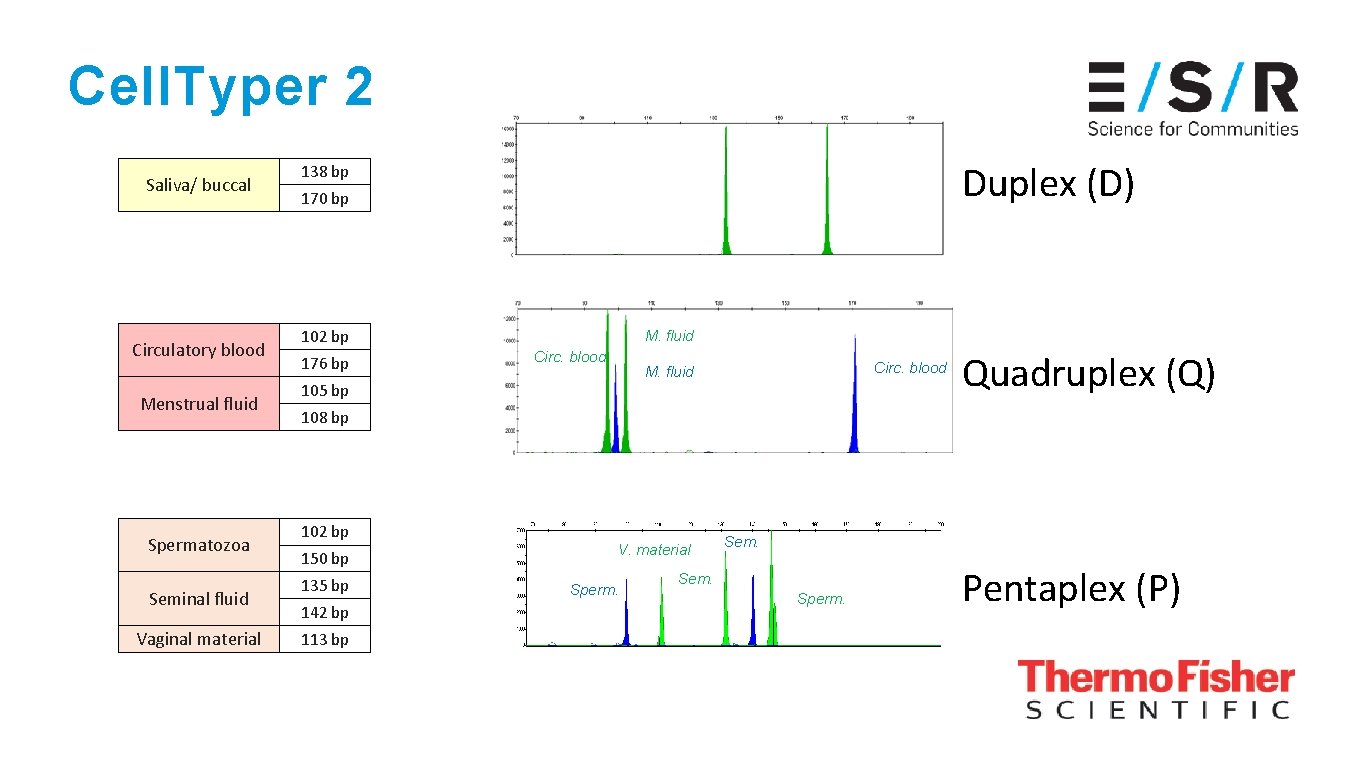
Cell. Typer 2 Saliva/ buccal Circulatory blood Menstrual fluid Spermatozoa Seminal fluid Vaginal material Duplex (D) 138 bp 170 bp 102 bp 176 bp M. fluid Circ. blood M. fluid 105 bp Quadruplex (Q) 108 bp 102 bp 150 bp 135 bp 142 bp 113 bp V. material Sperm. Sem. Sperm. Pentaplex (P)
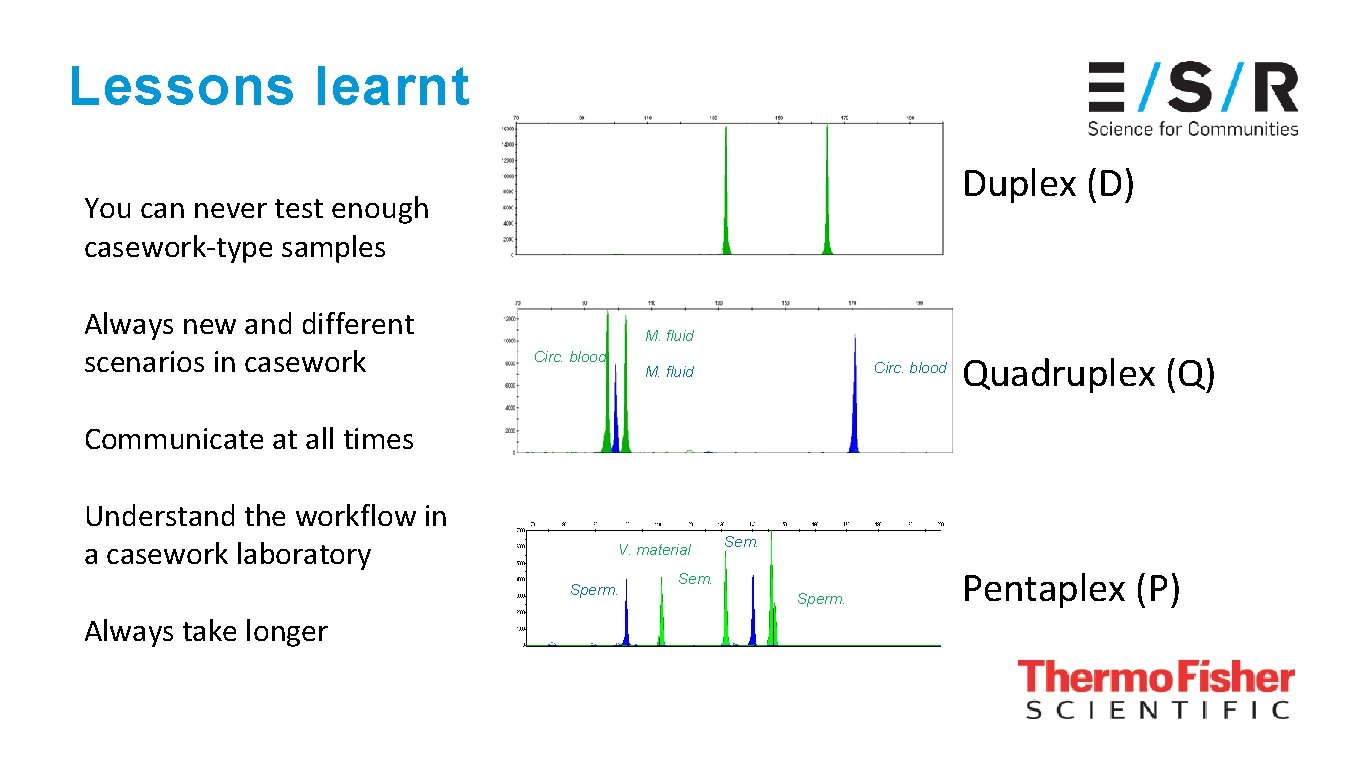
Lessons learnt Duplex (D) You can never test enough casework-type samples Always new and different scenarios in casework M. fluid Circ. blood M. fluid Quadruplex (Q) Communicate at all times Understand the workflow in a casework laboratory V. material Sperm. Always take longer Sem. Sperm. Pentaplex (P)
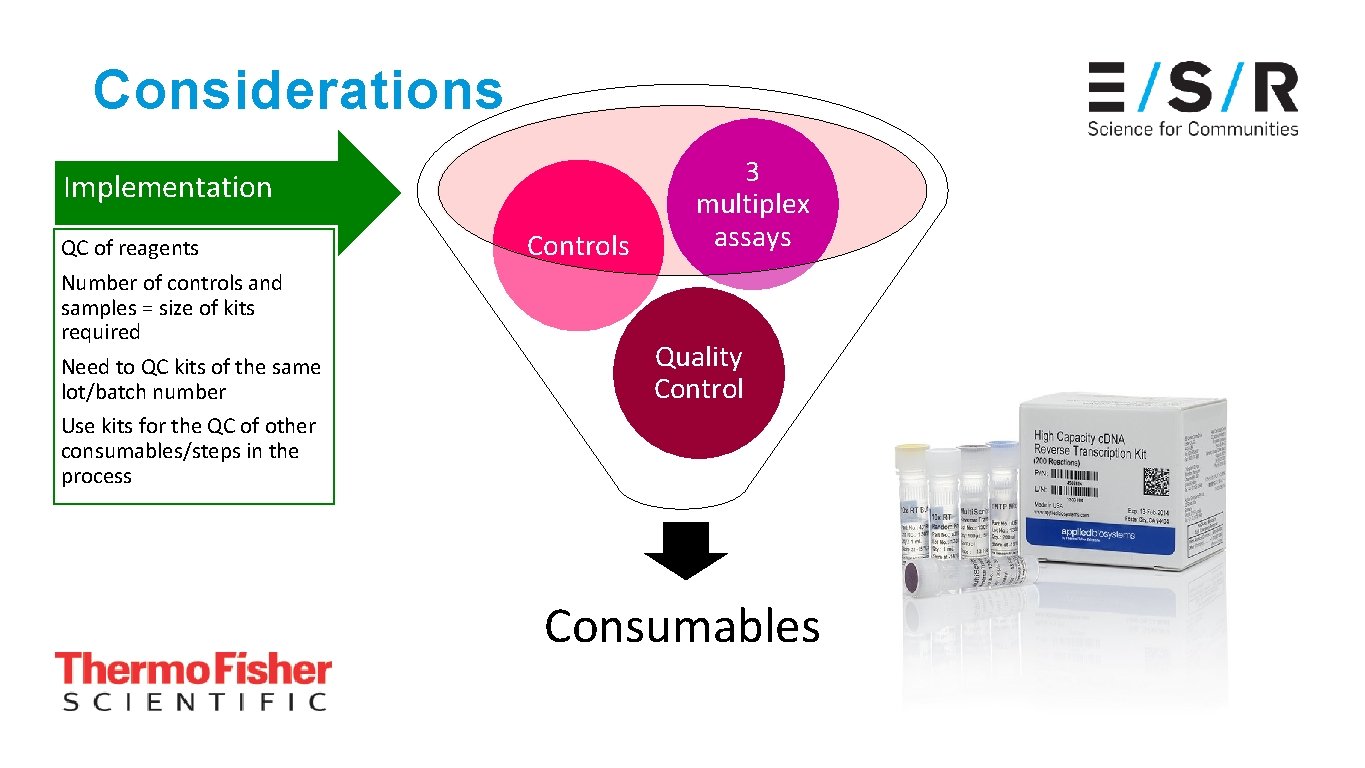
Considerations Implementation QC of reagents Number of controls and samples = size of kits required Need to QC kits of the same lot/batch number Use kits for the QC of other consumables/steps in the process Controls 3 multiplex assays Quality QC of reagents Control Consumables
Realtime PCR CE MPS Different platforms for different laboratories/uses
Casework Research Companies Technology RNA in forensic science at ESR Courts Other laboratories
Casework Research Communication Companies Technology Courts Other laboratories

Acknowledgements Patricia Albani Jayshree Patel Heidi Baker Sally. Ann Harbison Johanna Veth Keshni Rasanayagam Tracey Haldane Alicia Haines Thermo Fisher Scientific Rachel. Fleming@esr. cri. nz “Speaker was provided travel and hotel support by Thermo Fisher Scientific for this presentation, but no remuneration. When used for purposes other than Human Identification or Paternity Testing the instruments and software modules cited are for Research Use Only. Not for use in diagnostic procedures. Thermo Fisher Scientific and its affiliates are not endorsing, recommending, or promoting any use or application of Thermo Fisher Scientific products presented by third parties during this seminar. Information and materials presented or provided by third parties are provided as-is and without warranty of any kind, including regarding intellectual property rights and reported results. Parties presenting images, text and material represent they have the rights”
- Slides: 22