Detection of atmospherically relevant hydrocarbons by diode laser

Detection of atmospherically relevant hydrocarbons by diode laser cavity ringdown spectroscopy P. Cias, C. Wang, T. S. Dibble 60 -th International Symposium on Molecular Spectroscopy 06/21/2005 Diagnostic Instrumentation & Analysis Laboratory at Mississippi State University

Motivation Ø Importance of VOCs in atmospheric chemistry Ø Significance of organic peroxy radicals Ø Crucial role of isoprene-OH peroxy radicals Ø Environmental significance

First stage of studies Ø Detection of low concentrations of VOCs that are present in the atmosphere: 1, 3 -butadiene isoprene 2, 3 -dimethyl-1, 3 -butadiene Ø Experimental determination of their absorption cross-sections

The main problems of the VOCs and their radicals studies Ø Can they be quantitatively detected? (detection limit, sensitivity) Ø Can the particular isomers be distinguished? (resolution, selectivity)

Why Cavity Ringdown? Ø Trace levels of VOCs detection or the kinetic studies of their radicals cannot be done by conventional absorption spectroscopy (a detection limit issue) Ø Cavity Ringdown Spectroscopy as a very sensitive method makes these studies possible

Why Near Infrared region? Ø The absorption spectra of organic peroxy radicals obtained in near UV are broad and without well defined structure. The shapes and intensities themselves cannot provide isomeric selective information. Ø The spectra of organic peroxy radicals in NIR region exhibit well defined structure. This makes the identification of specific isomers possible. Ø Availability of appropriate diode lasers in the NIR region
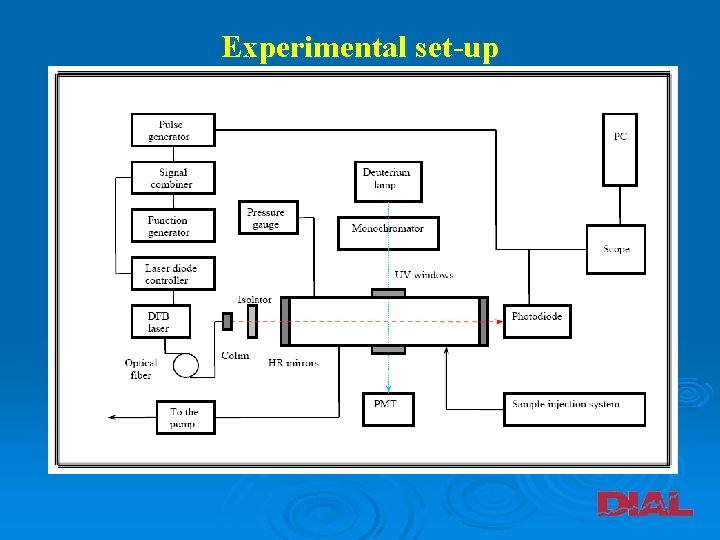
Experimental set-up

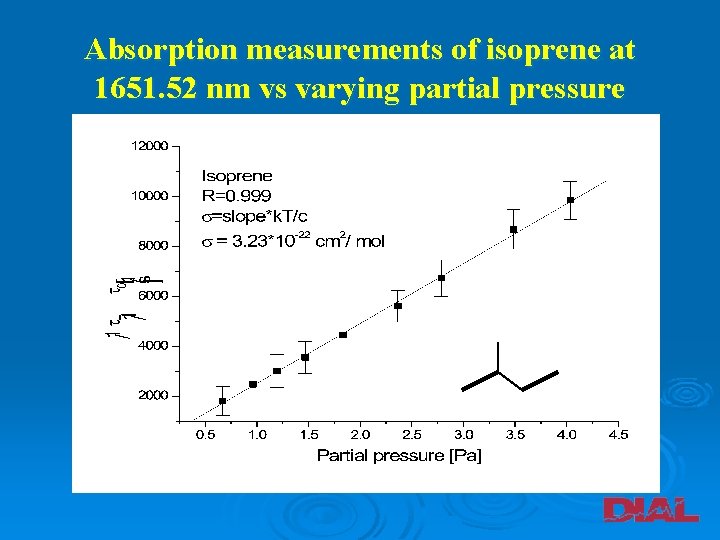
Absorption measurements of isoprene at 1651. 52 nm vs varying partial pressure

Absorption cross-sections and detection limits for 1, 3 -butadiene, isoprene and 2, 3 -dimethyl-1, 3 -butadiene at 1651. 52 nm Baseline stability [%] s Detection limit [ppmv] [cm 2 / mol] 2. 70*10 -21 (a) 1, 3 -butadiene 0. 32 2. 50 ± 0. 01*10 -22 3. 75 Isoprene 0. 43 3. 23 ± 0. 02*10 -22 2. 76 2, 3 -dimethyl 1, 3 -butadiene 0. 39 2. 82 ± 0. 02*10 -22 3. 45 (a) Absorption cross-section of 1, 3 -butadiene measured at 1624. 57 nm. A. M. Parkes et al. , Analyst 128 (2003) 960

Conclusions Ø Absorption cross-sections of isoprene and 2, 3 dimethyl-1, 3 -butadiene were determined in the NIR for the first time Ø This experimental data will be helpful in the further studies of isoprene peroxy radicals

Future plans Ø The NIR absorption spectra of isoprene-OH peroxy radicals Ø Absorption cross-section determination Ø Rate constants for the reactions of isoprene-OH peroxy radicals Ø Understanding of the role of isoprene in global atmospheric chemistry


Acknowledgments Dr. Chuji Wang Diagnostic Instrumentation & Analysis Laboratory Starkville, MS Susan T. Scherrer Diagnostic Instrumentation & Analysis Laboratory Starkville, MS Dr. Theodore S. Dibble State University of New York, College of Environmental Science and Forestry, Syracuse, NY National Science Foundation # ATM 0352926 Cooperation between MSU-DIAL and SUNY-ESF
- Slides: 14