Designing peptides from nature to bench Crazy about

Designing peptides: from nature to bench Crazy about biomedicine Júlia García Pindado
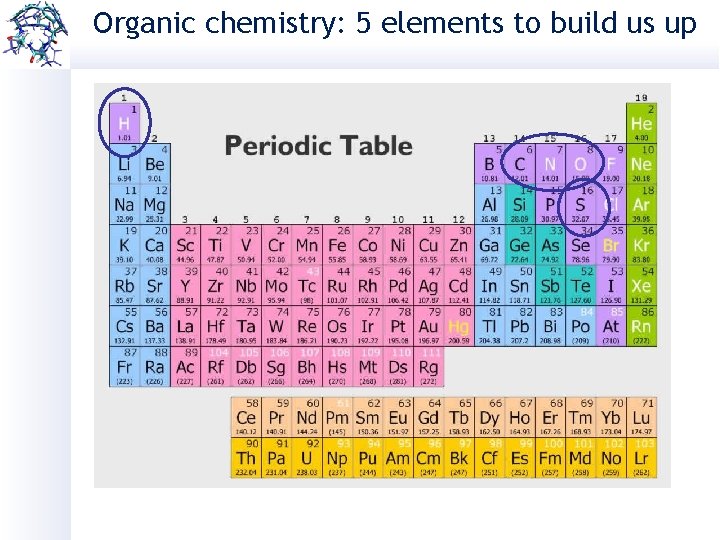
Organic chemistry: 5 elements to build us up
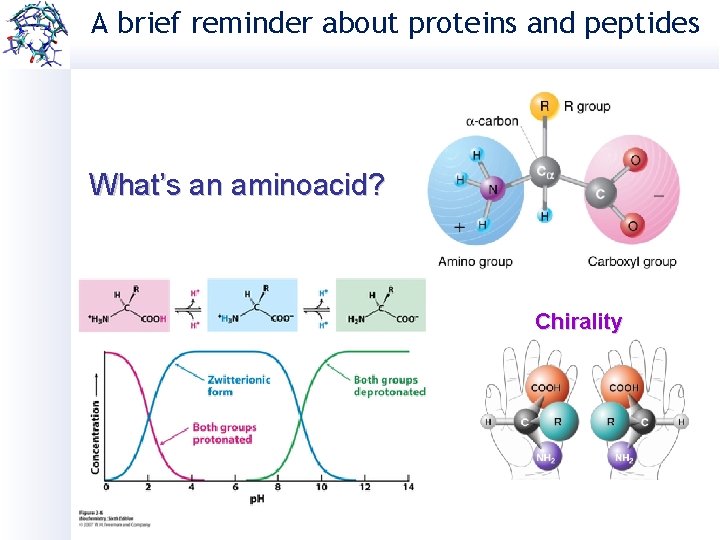
A brief reminder about proteins and peptides What’s an aminoacid? Chirality
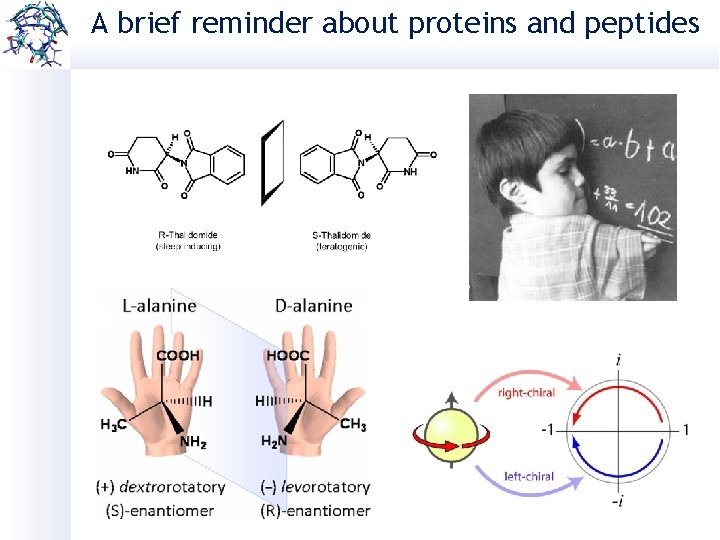
A brief reminder about proteins and peptides
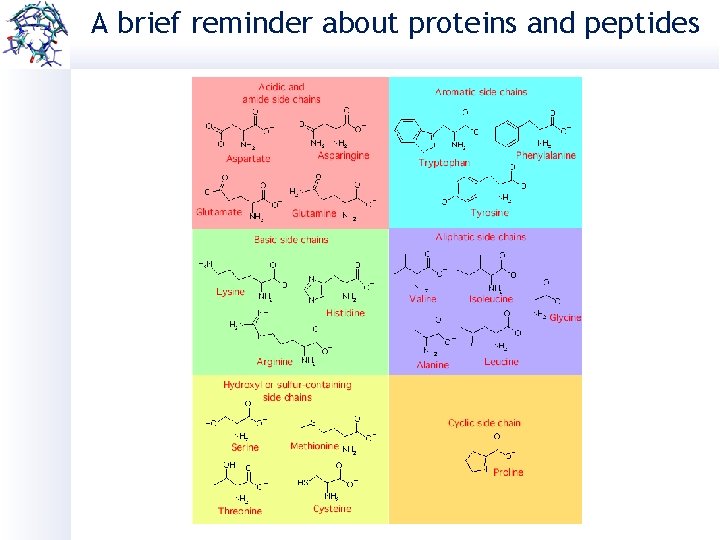
A brief reminder about proteins and peptides
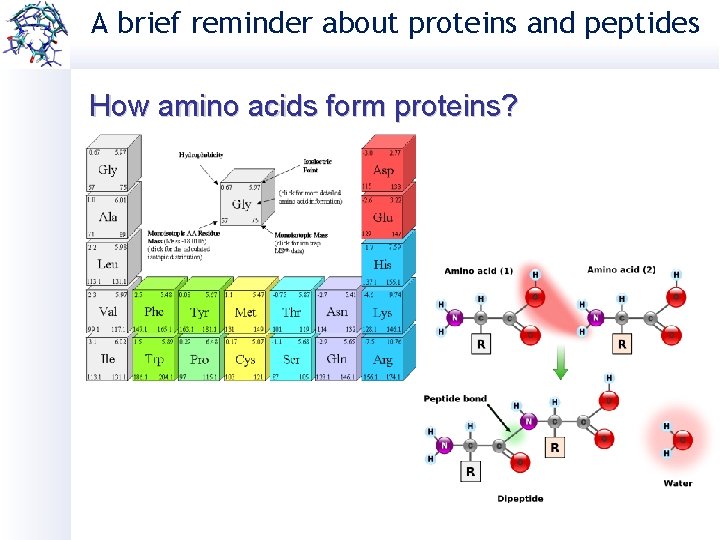
A brief reminder about proteins and peptides How amino acids form proteins?
A brief reminder about proteins and peptides Proteins
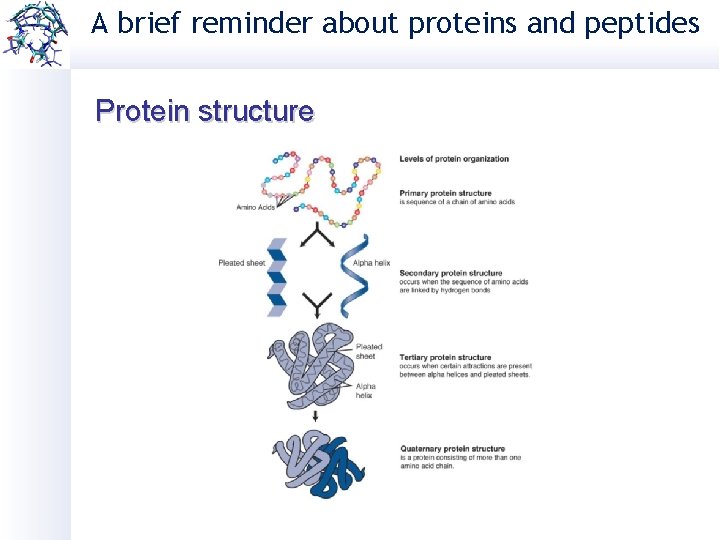
A brief reminder about proteins and peptides Protein structure
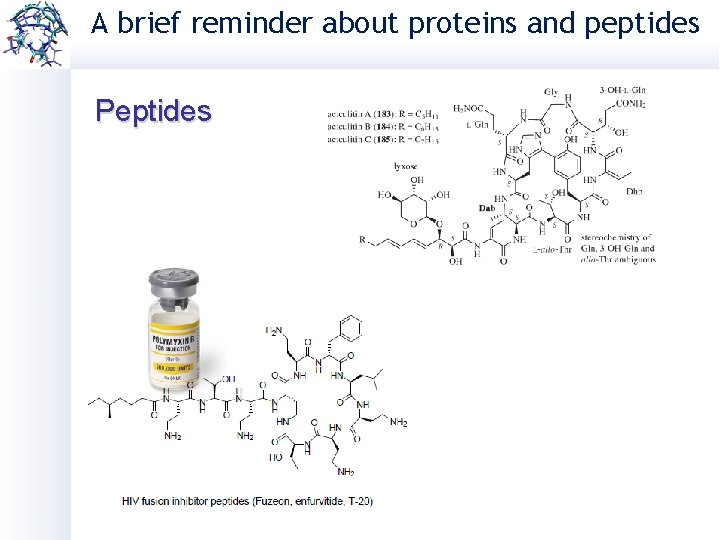
A brief reminder about proteins and peptides Peptides
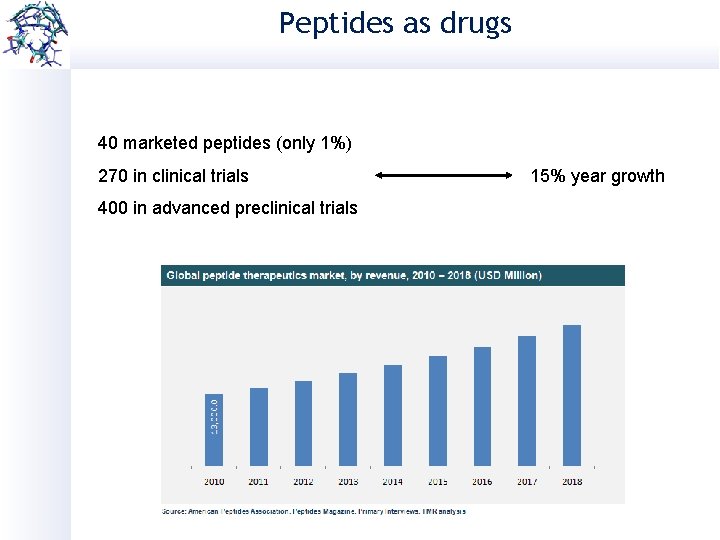
Peptides as drugs 40 marketed peptides (only 1%) 270 in clinical trials 400 in advanced preclinical trials 15% year growth
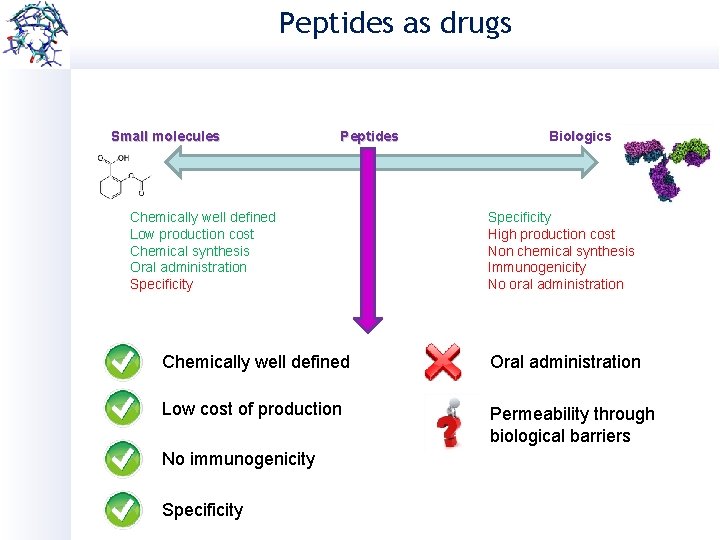
Peptides as drugs Small molecules Peptides Chemically well defined Low production cost Chemical synthesis Oral administration Specificity Biologics Specificity High production cost Non chemical synthesis Immunogenicity No oral administration Chemically well defined Oral administration Low cost of production Permeability through biological barriers No immunogenicity Specificity

Ideal drug candidate Ø Permeability through biological barriers reach the target Ø Stability Ø Solubility Ø Active (n. M range) Ø Non toxic
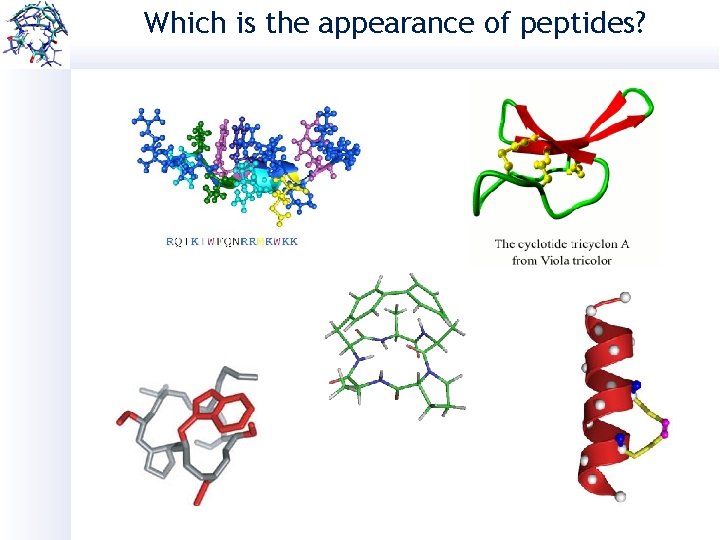
Which is the appearance of peptides?

Peptide synthesis

Solid phase peptide synthesis (SPPS) Developed by Merrifield in 1963 o Fast development o Complex peptides can be reached o 60 -70% efficiency Manual SPPS Microwave assisted SPPS

Solid phase peptide synthesis (SPPS)
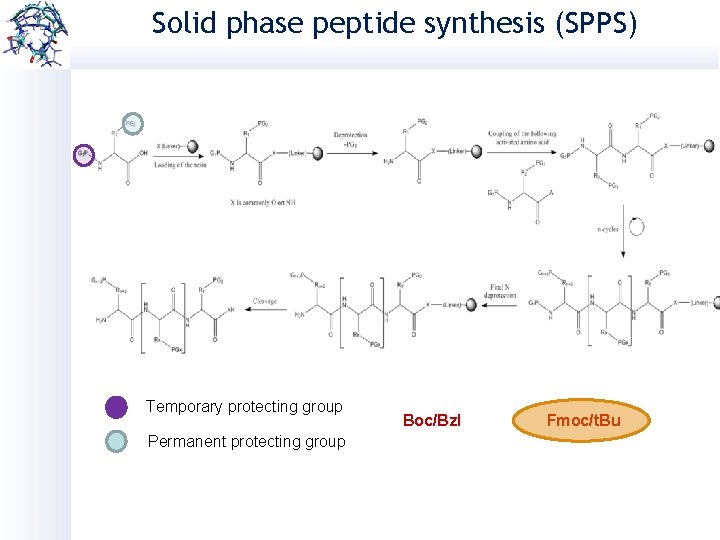
Solid phase peptide synthesis (SPPS) Temporary protecting group Permanent protecting group Boc/Bzl Fmoc/t. Bu

Solid phase peptide synthesis (SPPS) Resins Size of the resin beads (80 -200 µm) Mechanical and thermal stability Price Swelling Types: Ø Cross-linked polysterene Ø Polyamide Ø Tentagel Ø Soluble
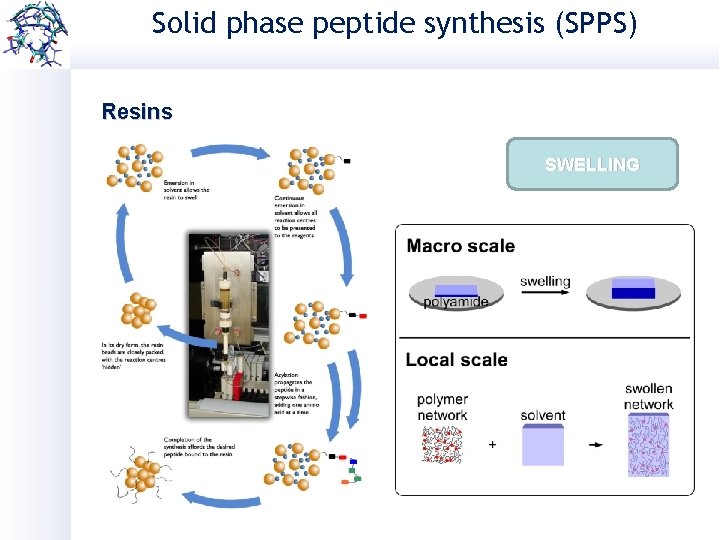
Solid phase peptide synthesis (SPPS) Resins Wang SWELLING 2 -Chlorotrityl chloride Rink amide Chemmatrix
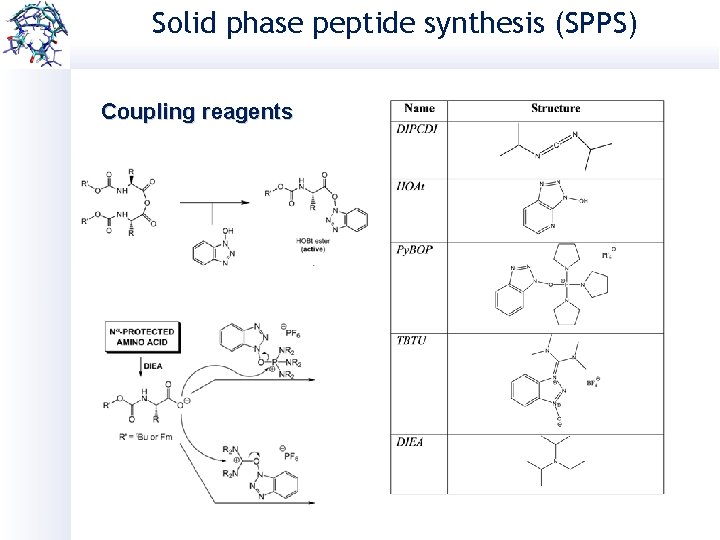
Solid phase peptide synthesis (SPPS) Coupling reagents
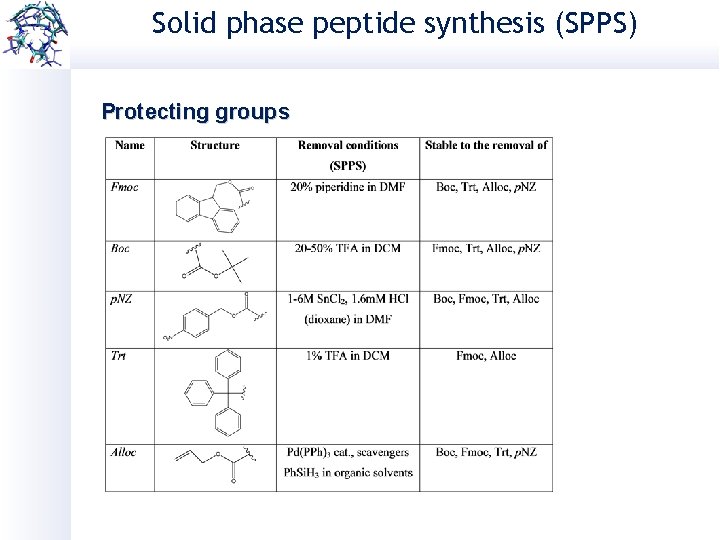
Solid phase peptide synthesis (SPPS) Protecting groups

Solid phase peptide synthesis (SPPS) Protecting groups
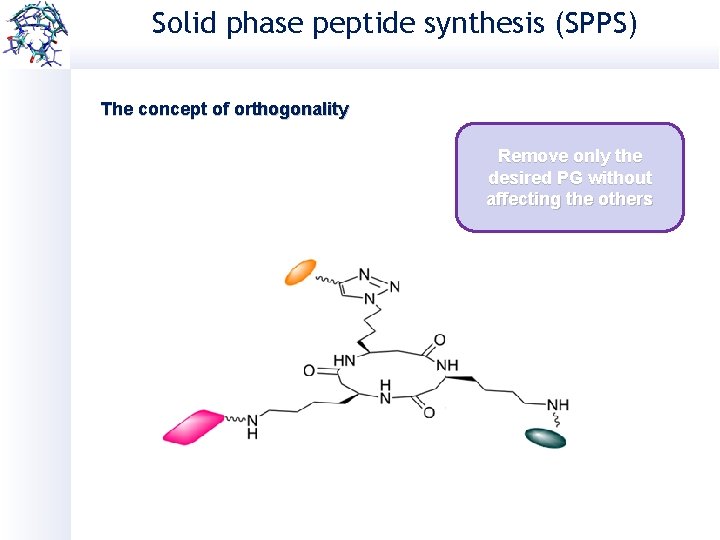
Solid phase peptide synthesis (SPPS) The concept of orthogonality Remove only the desired PG without affecting the others
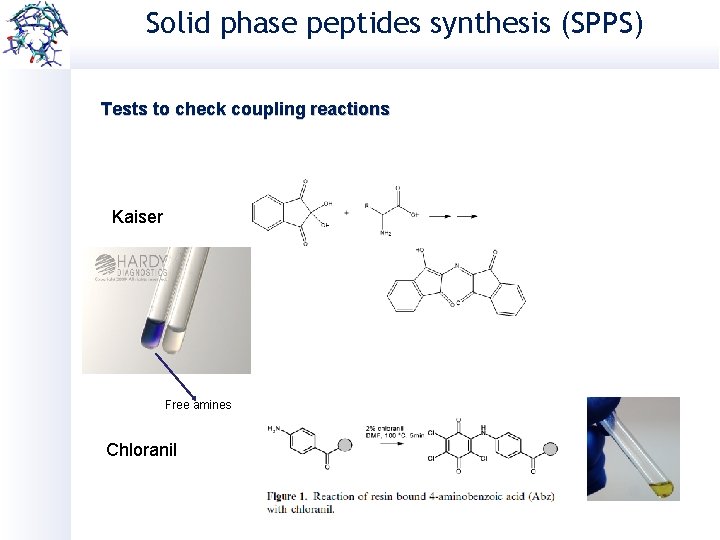
Solid phase peptides synthesis (SPPS) Tests to check coupling reactions Kaiser Free amines Chloranil
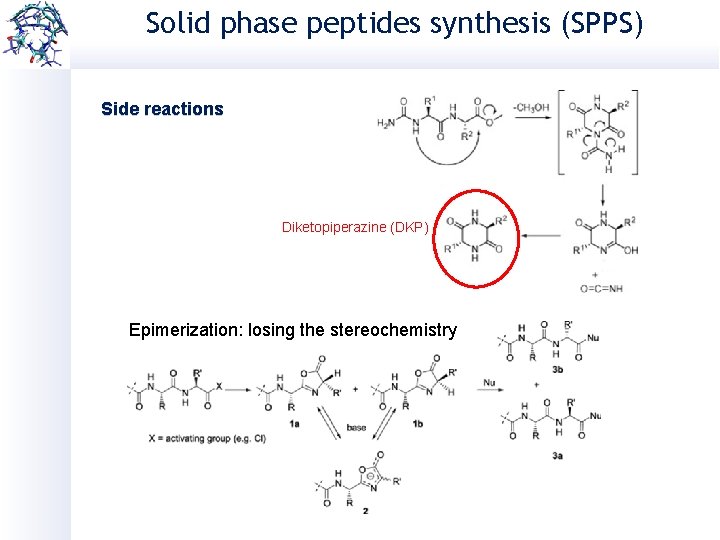
Solid phase peptides synthesis (SPPS) Side reactions Diketopiperazine (DKP) Epimerization: losing the stereochemistry

Solid phase peptides synthesis (SPPS) Other inconveniences Aggregation Deletion of residues Problems during the cleavage process

Solid phase peptides synthesis (SPPS) Purification Traditional chromatographic purification

Solid phase peptides synthesis (SPPS) Purification SPPS enables us to avoid performing several purification steps Reagents Solvent Impurities Beads with the peptide Beads of resin wash Reagents and byproducts

Solid phase peptides synthesis (SPPS) Purification HPLC can be used to separate the drug from all the other impurities

How can we improve synthetic peptides? Y? H W To… - Increase stability - Enhance cell uptake and permeability through biological barriers - Improve resistance against proteases ü ü N-methylated peptides Non natural aminoacids Cyclic peptides Stapled peptides
How can we improve synthetic peptides? Proteases
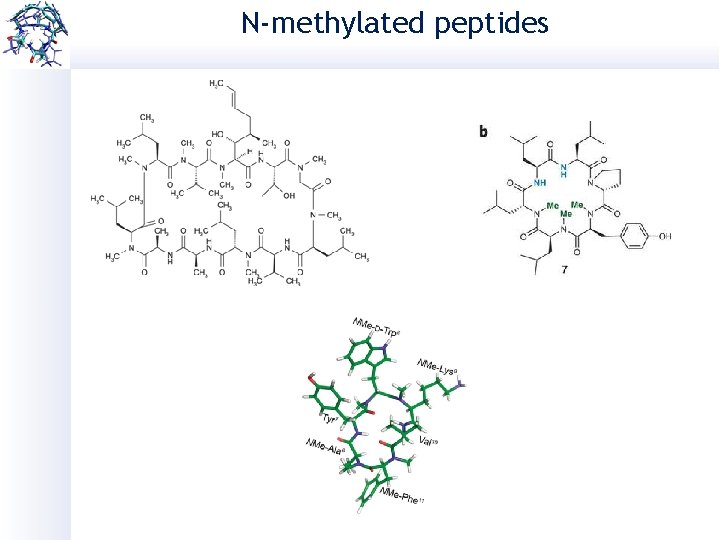
N-methylated peptides
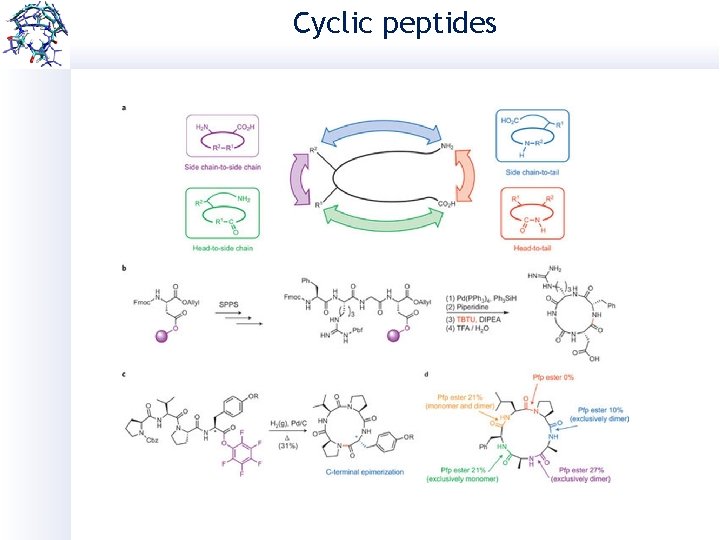
Cyclic peptides

Stapled peptides

Designing peptides v Synthesis of natural peptides v Synthesis of novel peptides
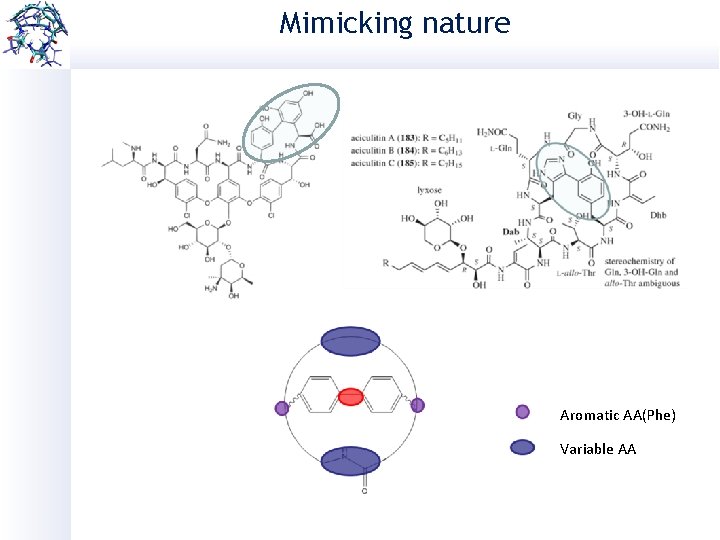
Mimicking nature Aromatic AA(Phe) Variable AA

Creating new sequences 1) Envisaging the desired product 2) Proper selection of the strategy 1) Resin 2) PG 3) Final removal of all the PG? 4) Further reactions and/or deprotections after cleavage 3) Carrying out the synthesis
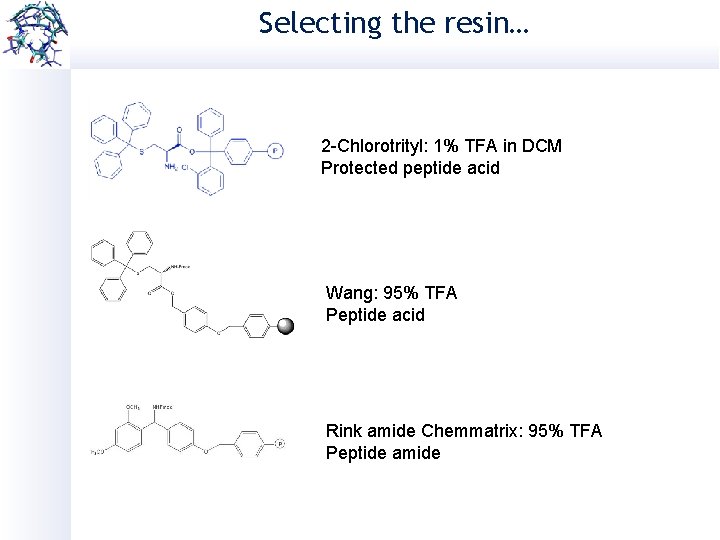
Selecting the resin… 2 -Chlorotrityl: 1% TFA in DCM Protected peptide acid Wang: 95% TFA Peptide acid Rink amide Chemmatrix: 95% TFA Peptide amide
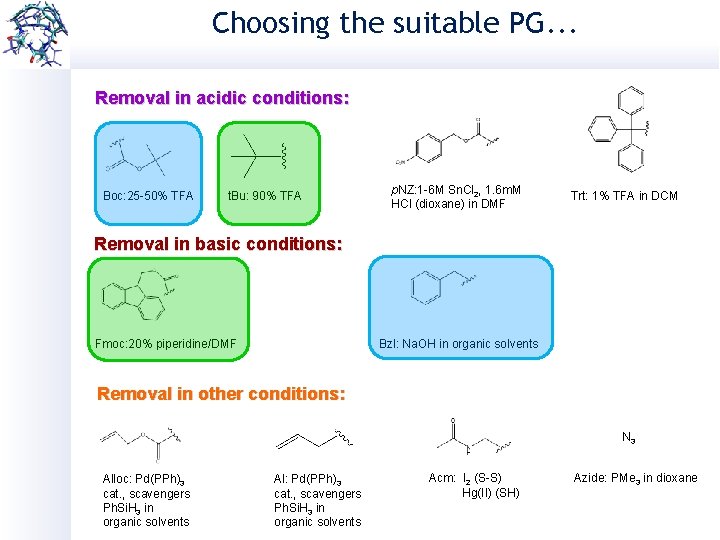
Choosing the suitable PG. . . Removal in acidic conditions: Boc: 25 -50% TFA t. Bu: 90% TFA p. NZ: 1 -6 M Sn. Cl 2, 1. 6 m. M HCl (dioxane) in DMF Trt: 1% TFA in DCM Removal in basic conditions: Fmoc: 20% piperidine/DMF Bzl: Na. OH in organic solvents Removal in other conditions: N 3 Alloc: Pd(PPh)3 cat. , scavengers Ph. Si. H 3 in organic solvents Al: Pd(PPh)3 cat. , scavengers Ph. Si. H 3 in organic solvents Acm: I 2 (S-S) Hg(II) (SH) Azide: PMe 3 in dioxane

Choosing the suitable PG. . .
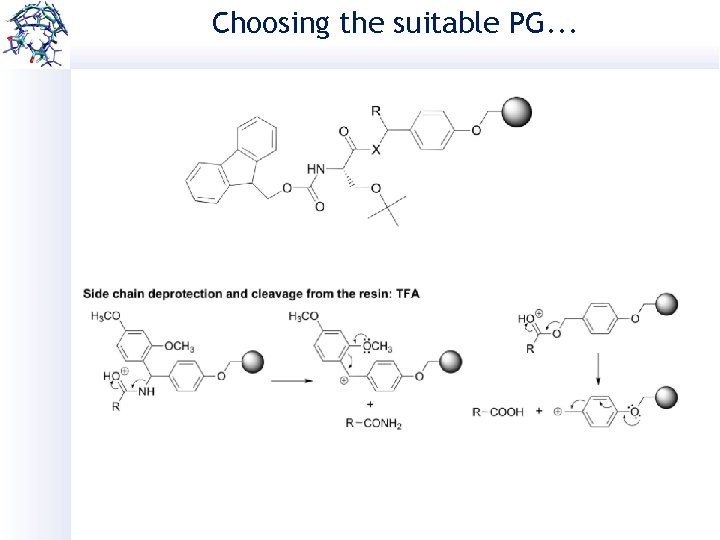
Choosing the suitable PG. . .

Analysis of the obtained product High Performance Liquid Chromatography (HPLC)

Analysis of the obtained product Mass spectrometry (MS)

Our lab (Giralt’s group) Design, synthesis and structure of peptides and proteins
- Slides: 44