DESIGN OF THE PERIODIC TABLE DMITRI MENDELEEV Russian

DESIGN OF THE PERIODIC TABLE • DMITRI MENDELEEV: – Russian chemist who first arranged the elements according to their increasing atomic mass. • HENRY MOSELEY: – British scientist who arranged the elements of the modern day periodic table according to increasing atomic numbers.

COLUMNS • 18 Main numbered columns • Columns of elements are called FAMILIES. • Elements within a family have similar properties.

ROWS • 7 Main numbered rows • Horizontal rows are called PERIODS. • Properties of the elements change greatly across a period • Active solid inactive gas
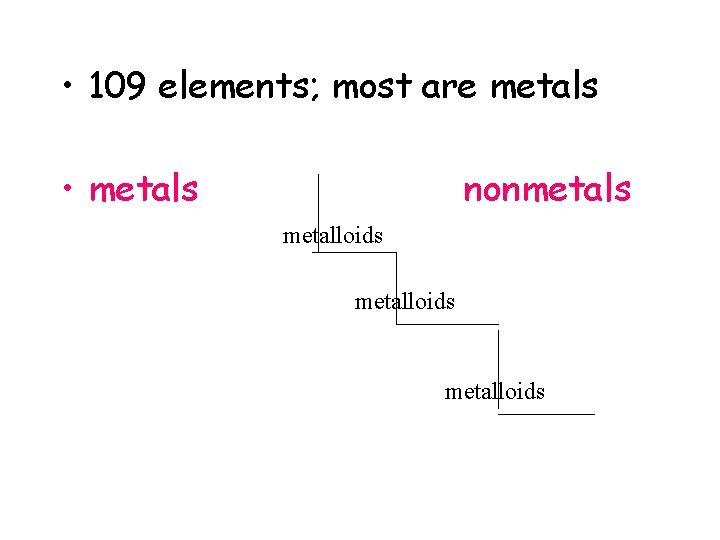
• 109 elements; most are metals • metals nonmetals metalloids

METALS • LUSTER = shiny • Corrosive (react with water) • Good conductors (heat and • Tend to lose electricity) electrons • High density • DUCTILE (wires) • MALLEABLE (hammered into thin sheets)

NON-METALS • No luster; dull • Not good conductors of heat or electricity • Brittle; break easily • Not ductile or malleable • Low density • Can bond with a metal (Na. Cl) • Tend to gain electrons • Elements with full energy levels are non-reactive

METALLOIDS • “Metal-like” • Have properties of both metals and non-metals • Are all solids (shiny or dull) • Are better conductors than nonmetals • Are ductile and malleable
- Slides: 7