Design and Analysis of Crossover Study Designs Bhargava

Design and Analysis of Crossover Study Designs Bhargava Kandala Department of Pharmaceutics College of Pharmacy , UF

Crossover Study Treatments administered in a sequence to each experimental unit over a set of time periods. Comparison of treatments on a within-subject level. Increased precision of treatment comparisons. A treatment given in one period might influence the response in the following treatment period – residual/carryover effect Baseline values – Can be included as covariates to increase the precision
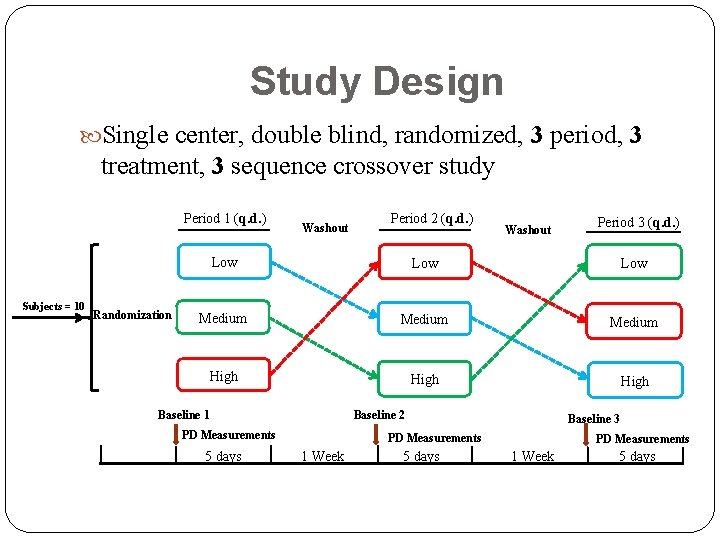
Study Design Single center, double blind, randomized, 3 period, 3 treatment, 3 sequence crossover study Period 1 (q. d. ) Subjects = 10 Randomization Washout Period 2 (q. d. ) Washout Period 3 (q. d. ) Low Low Medium High Baseline 1 Baseline 2 PD Measurements 5 days Baseline 3 PD Measurements 1 Week 5 days

Model for Crossover Design Period 1 2 3 4 5 6 I A B C II B C A C III C A B B A B C A

proc glm data = allperiodanaly; class sequence subject period trt; model fenoav = sequence subject(sequence) period trt/solution; random subject(sequence); run;

proc mixed data = allperiodanaly; class sequence subject period trt; model fenoav = sequence period trt; random subject(sequence); lsmeans trt/ pdiff cl; run;

Baseline
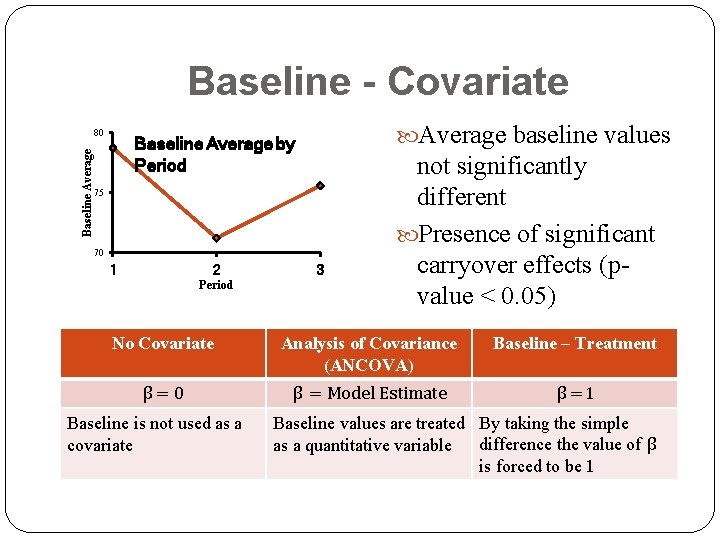
Baseline - Covariate Baseline Average 80 Average baseline values Baseline Average by Period 75 70 1 2 Period 3 not significantly different Presence of significant carryover effects (pvalue < 0. 05) No Covariate Analysis of Covariance (ANCOVA) Baseline – Treatment β=0 β = Model Estimate β =1 Baseline is not used as a covariate Baseline values are treated By taking the simple as a quantitative variable difference the value of β is forced to be 1

Carryover Effect

* Covariates tested for carryover; proc mixed data = allperiodanaly; class sequence subject period trt; model fenoav = sequence period fenob trt carry 1 carry 2; random subject(sequence); lsmeans trt/ pdiff cl e; run;
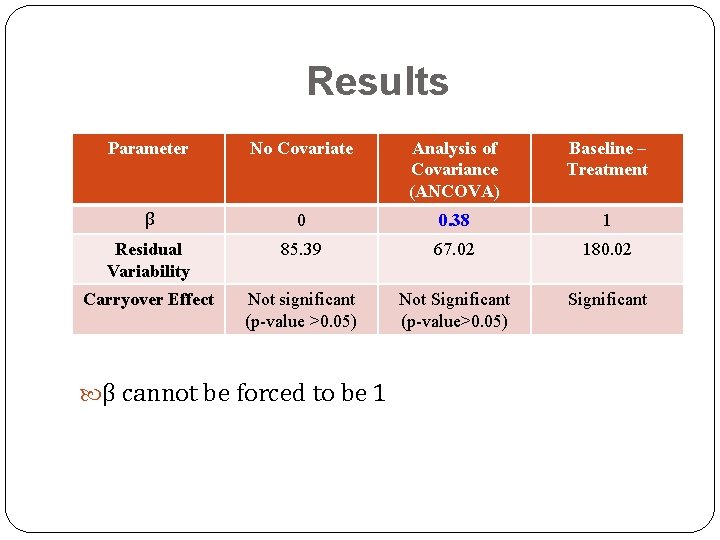
Results Parameter No Covariate Analysis of Covariance (ANCOVA) Baseline – Treatment β 0 0. 38 1 Residual Variability 85. 39 67. 02 180. 02 Carryover Effect Not significant (p-value >0. 05) Not Significant (p-value>0. 05) Significant β cannot be forced to be 1
Results Parameter No Covariate Analysis of Covariance (ANCOVA) Baseline – Treatment β 0 0. 38 1 Residual Variability 85. 39 67. 02 180. 02 Carryover Effect Not significant (p-value >0. 05) Not Significant (p-value>0. 05) Significant
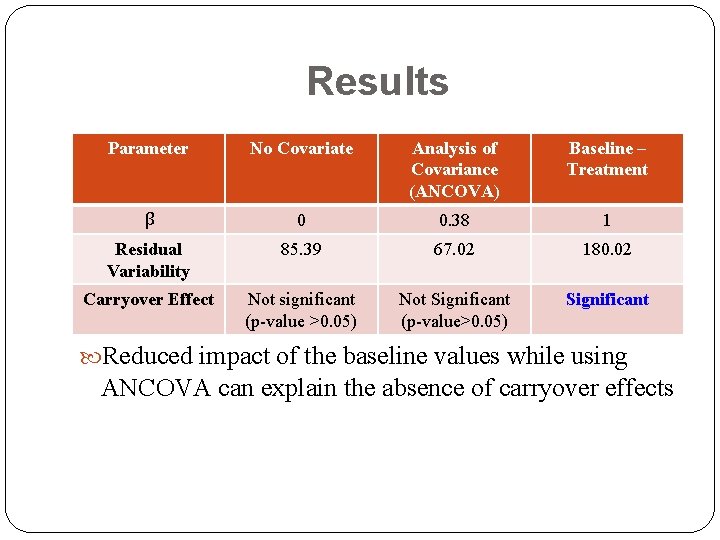
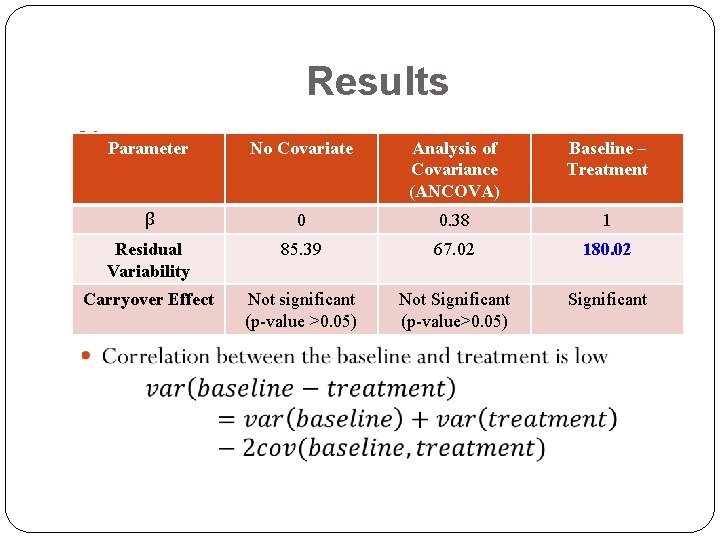
Results Parameter No Covariate Analysis of Covariance (ANCOVA) Baseline – Treatment β 0 0. 38 1 Residual Variability 85. 39 67. 02 180. 02 Carryover Effect Not significant (p-value >0. 05) Not Significant (p-value>0. 05) Significant Reduced impact of the baseline values while using ANCOVA can explain the absence of carryover effects

Conclusions Day 5 data suitable for analysis Maximum dose resolution No carryover effect Baseline adjustment Simple difference increases the variability and introduces carryover effects ANCOVA is the preferred method Crossover design model with baseline values as covariates will be used for future simulations
- Slides: 14