Description of Phase Transformation i Equilibrium phase transformations

Description of Phase Transformation (i) Equilibrium phase transformations – occurring at the transition temperature ( e. g. freezing at Tm) (ii) Non-equilibrium phase transformations (freezing below Tm) Equilibrium phase transitions: Recall the Ehrenfest (“classical”) classification scheme. An nth order transition is defined by the lowest- order derivative which becomes discontinuous at the transition temperature.
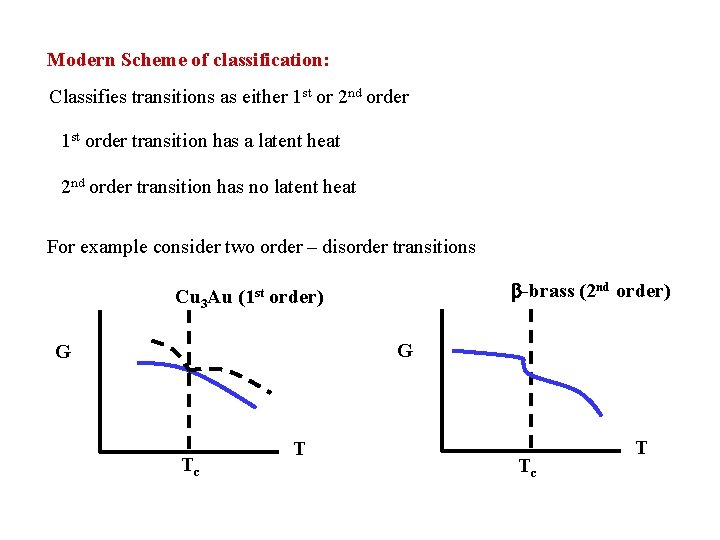
Modern Scheme of classification: Classifies transitions as either 1 st or 2 nd order 1 st order transition has a latent heat 2 nd order transition has no latent heat For example consider two order – disorder transitions -brass (2 nd order) Cu 3 Au (1 st order) G G Tc T
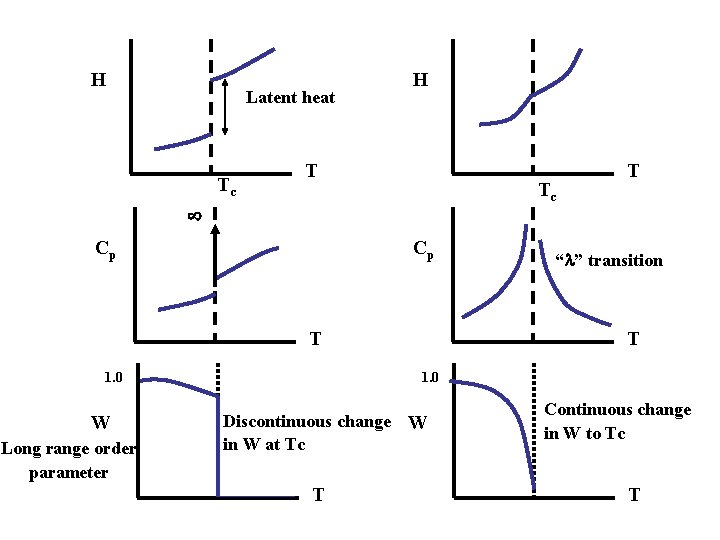
H Latent heat Tc H T Tc Cp Cp T Long range order parameter “ ” transition T 1. 0 W T Discontinuous change W in W at Tc T Continuous change in W to Tc T

Tid bits • 1 st order transition has infinite Cp at Tc • Cp has a lambda point for 2 nd order transition • 2 nd order (continuous) transitions are studied as the “physics of critical phenomena” Non – equilibrium Transformations: Many important phase transformations do not occur at equilibrium. (e. g. undercooling during solidification) Since these transitions do not occur at Tc, we can’t use the previous classification schemes.
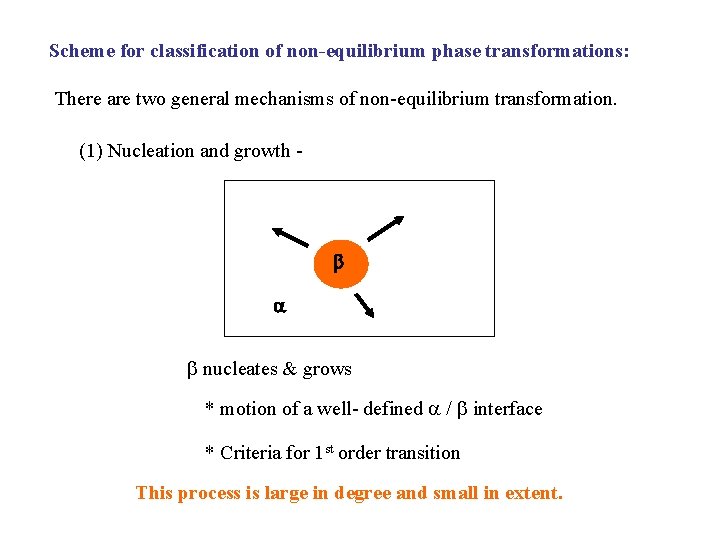
Scheme for classification of non-equilibrium phase transformations: There are two general mechanisms of non-equilibrium transformation. (1) Nucleation and growth - nucleates & grows * motion of a well- defined / interface * Criteria for 1 st order transition This process is large in degree and small in extent.
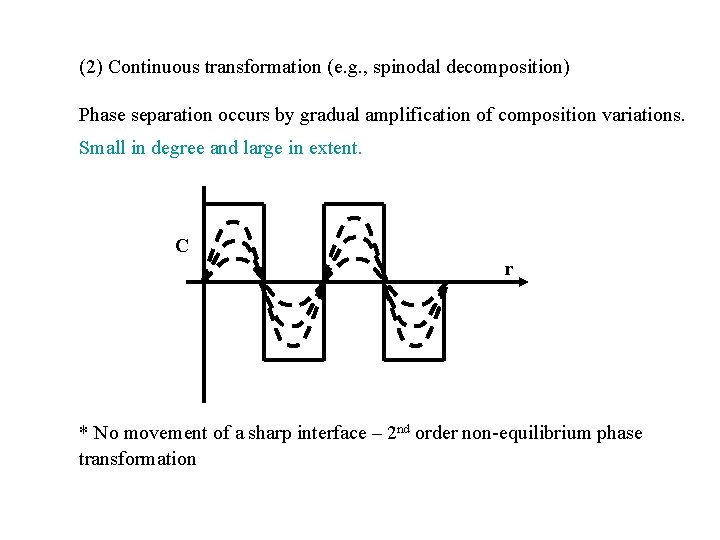
(2) Continuous transformation (e. g. , spinodal decomposition) Phase separation occurs by gradual amplification of composition variations. Small in degree and large in extent. C r * No movement of a sharp interface – 2 nd order non-equilibrium phase transformation
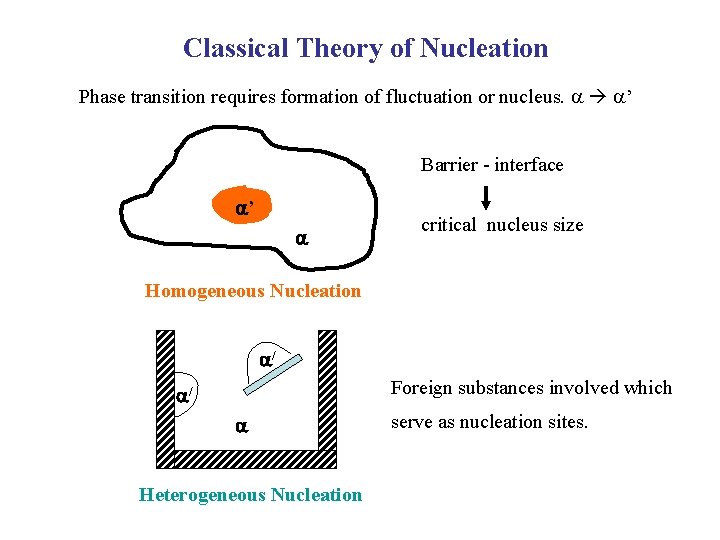
Classical Theory of Nucleation Phase transition requires formation of fluctuation or nucleus. ’ Barrier - interface ’ critical nucleus size Homogeneous Nucleation / Foreign substances involved which / Heterogeneous Nucleation serve as nucleation sites.

Thermodynamics of Nucleation : (i) Homogeneous Nucleation – (1925 Volmer-Weber) Series of bimolecular reactions I (A) Here, i* is the critically sized nucleus. Other methods of cluster formation such as simultaneous collisions are less probable. Volmer’s kinetic analysis considered only the forward rate of reaction (A). The back reacting rate was considered small and was ignored

Thermodynamic balance of reactions up to (A) Mixing of ni clusters of size i increases the entropy of the system some cluster populations are always present. Dilute solution theory for mixture of n 1 ……. ni clusters: The chemical potential of an i atom cluster : # per unit volume in equilibrium : (*chemical potential per mole the same in all clusters likely an incorrect assumption!)
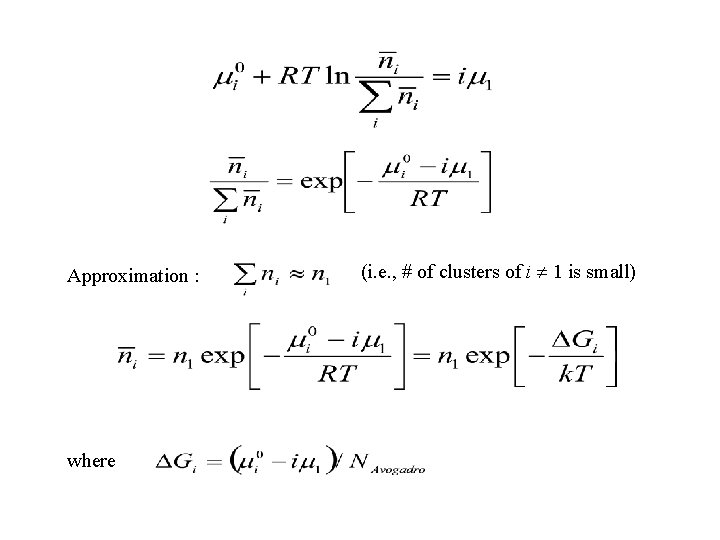
Approximation : where (i. e. , # of clusters of i 1 is small)
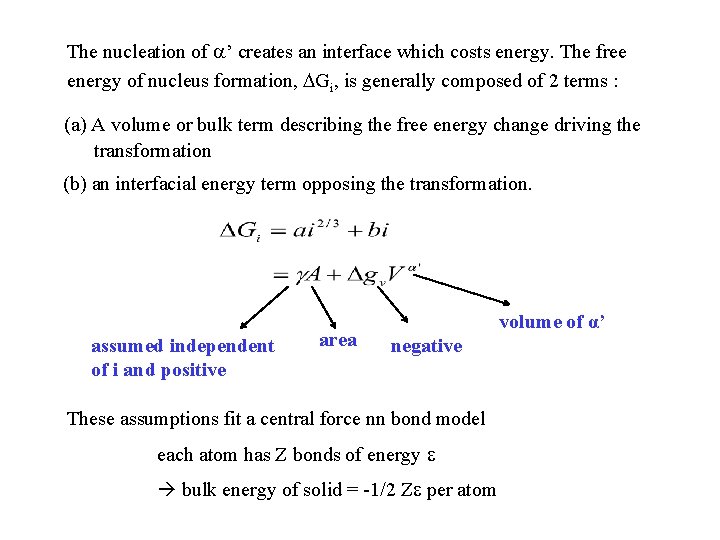
The nucleation of ’ creates an interface which costs energy. The free energy of nucleus formation, Gi, is generally composed of 2 terms : (a) A volume or bulk term describing the free energy change driving the transformation (b) an interfacial energy term opposing the transformation. assumed independent of i and positive area volume of α’ negative These assumptions fit a central force nn bond model each atom has Z bonds of energy bulk energy of solid = -1/2 Z per atom
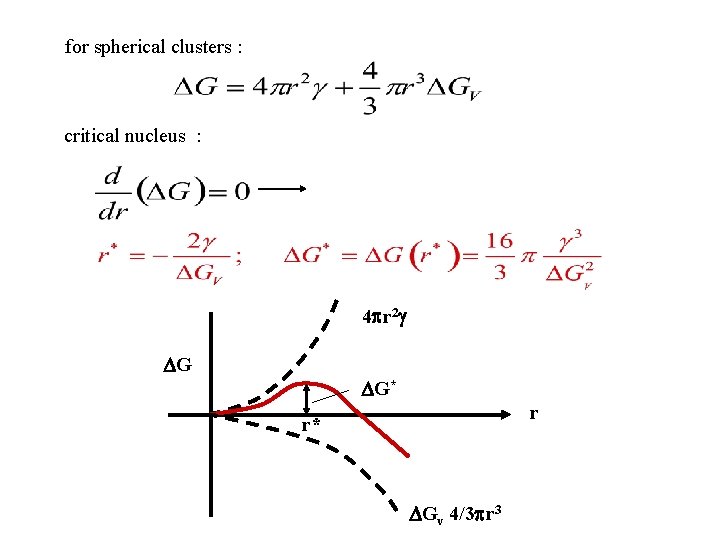
for spherical clusters : critical nucleus : 4 r 2 G G* r r* Gv 4/3 r 3

The equilibrium conc. of critical nuclei What to use for Gv? Pure materials (1) Gas Liquid Gas Solid Gas pressure Equil. vap pressure (2) Condensed phases Recall: liq sol Entropy of transformation ; Te = equil. transition temp.
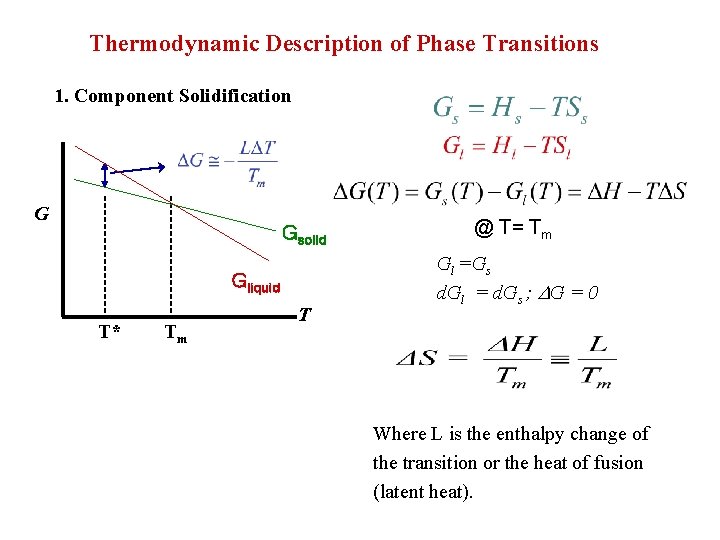
Thermodynamic Description of Phase Transitions 1. Component Solidification G Gsolid Gliquid T* Tm T @ T= Tm Gl =Gs d. Gl = d. Gs ; G = 0 Where L is the enthalpy change of the transition or the heat of fusion (latent heat).

Alloys: ’ nucleus can have a composition different from . e. g. , binary system, x is atom fraction of one element.
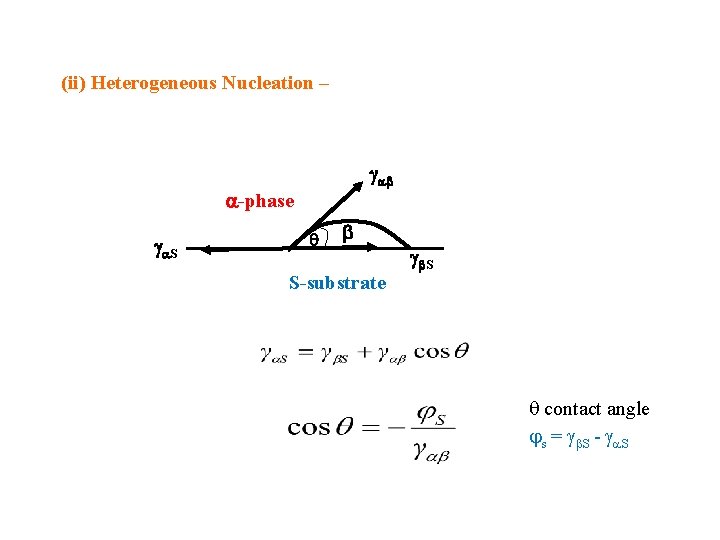
(ii) Heterogeneous Nucleation – -phase S S-substrate S contact angle s = S - S

Superhydrophobic surface – Lotus leaf
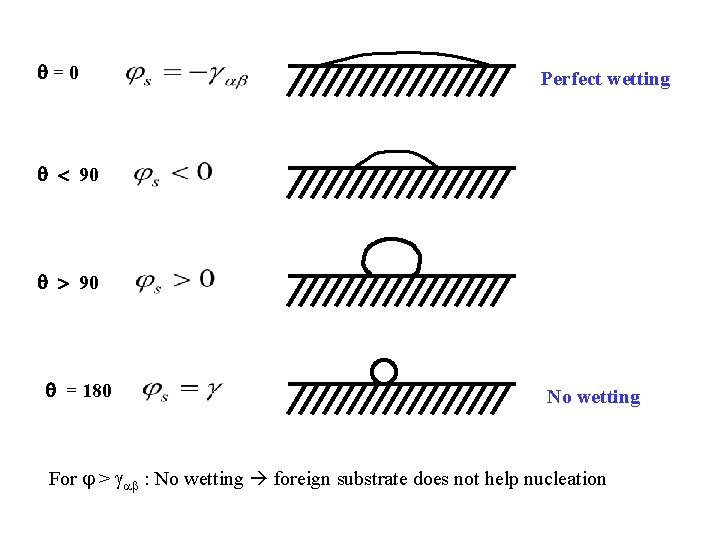
=0 Perfect wetting 90 90 = 180 No wetting For > : No wetting foreign substrate does not help nucleation
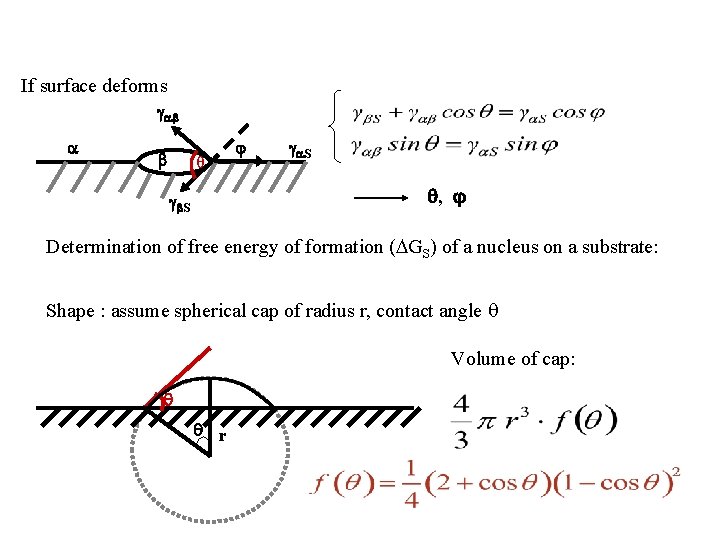
If surface deforms θ S , S Determination of free energy of formation ( GS) of a nucleus on a substrate: Shape : assume spherical cap of radius r, contact angle Volume of cap: r

Cap volume Cap area Contact circle area S - S
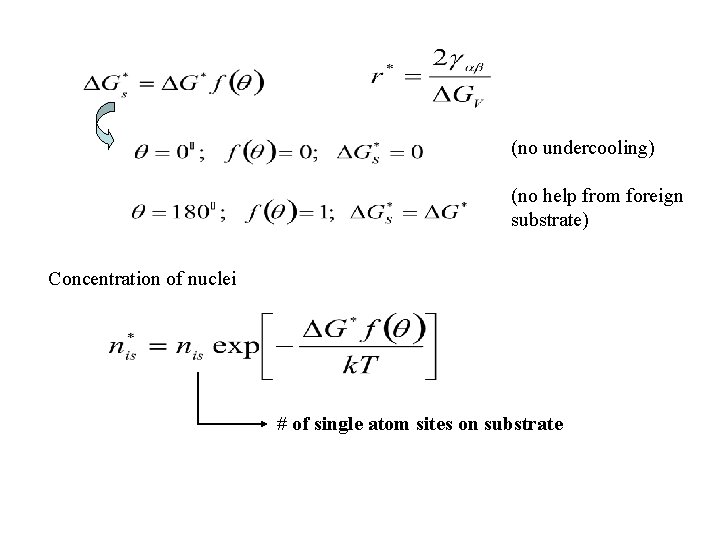
(no undercooling) (no help from foreign substrate) Concentration of nuclei # of single atom sites on substrate
- Slides: 21