Describing Substances Unit 4 Review So Far Weve

Describing Substances Unit 4

Review So Far… ▪ We’ve learned that matter makes up everything ▪ Matter has mass and takes up space (volume) ▪ Matter can exist in three states/phases (solid, liquid, and gas) ▪ With the loss or gain of energy matter can transition between these three phases. ▪ We can calculate the amount of energy gained or lost using Q=mc∆T or Q=m. Hv or Q=m. Hf

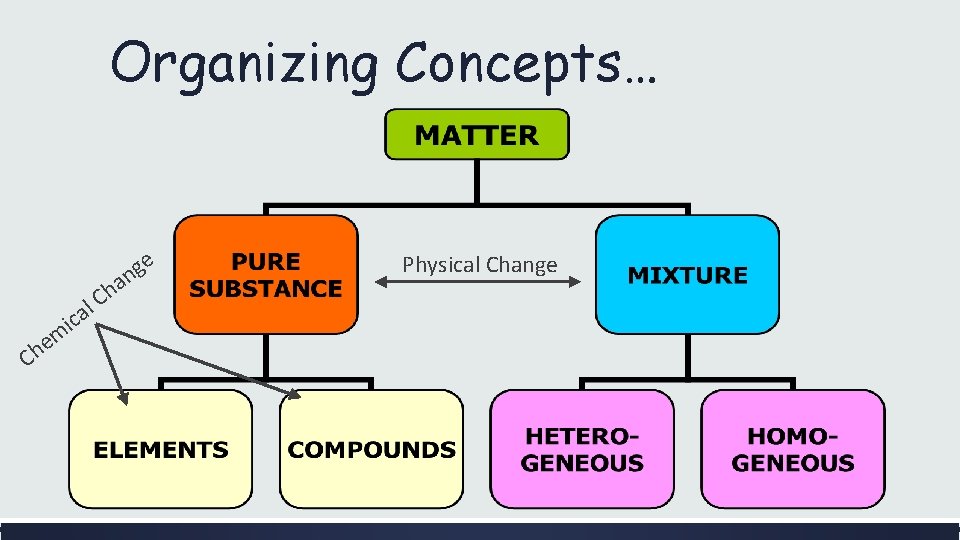
Classifying Matter Essential Questions: How is matter classified? How can physical and chemical changes change a substance’s classification? ■ ■ ■ Scientists like to classify things. One way that scientists classify matter is by its composition. Ultimately, all matter can be classified as mixtures, elements and compounds.

Why isn’t it a good idea to classify matter by its phases? Because one kind of substance can exist in more than one phase – such as H 20. And matter changes phases rather easily.
Organizing Concepts… e m e Ch l a ic g n a h C Physical Change

Pure Substances vs. Mixtures ■ Pure Substances ■ ■ ■ Made of either elements or compounds that are chemically bonded. Cannot be separated by physical means. Glucose: C 6 H 12 O 6 Table Salt: Na. Cl Oxygen Gas: O 2 Carbon Dioxide CO 2 ■ Mixtures: ■ Two or more substances mixed together but not chemically combined. ■ ■ ■ Tea = Crushed Leaf + H 2 O Rocks = minerals + sediments + organic matter Sugar Water = Sugar + H 2 O Each component retains its own identity; it does not change into something else. Can be separated by physical means

Pure Substances: Elements vs. Compounds ■ Elements ■ ■ Composed of one type of element. Atom – a single element. ■ ■ Compounds: ■ Ex: Ag, Li, C, Co, Diatomic Molecule – two of the same element. ■ ■ Ex: O 2, H 2, F 2 ■ Made up of 2 or more different kinds of atoms that are chemically combined. Ex: H 2 O, NH 4, CO 2, HCl

Mixtures ■ Heterogeneous ■ ■ ■ “Hetero-” refers to different Not evenly mixed Individual components can be easily seen Can be easily separated Ex: salt & pepper mix, rocks, cereal, bag of assorted candies. ■ Homogeneous ■ ■ ■ “Homo-” refers to same Evenly mixed Individual components can not be easily seen Not easily separated Ex: tea, salt & water solution, soda, milk, toothpaste, kool-aid.

Classify the following substances as: Pure substance, heterogeneous mixture, or homogeneous mixture ■ ■ ■ Gatorade Milk Shake Meat marinade Gasoline Dirt Sulfur ■ ■ ■ Helium gas Sugar Crystals Fruit Loops Mashed Potatoes Vinegar Air

Physical vs. Chemical Properties & Changes Essential Questions: ▪ How are physical and chemical properties different? ▪ How are physical and chemical changes different?

Physical Properties of Matter ■ Can be observed without changing a substance into another ■ Color ■ Odor ■ Luster– how shiny an object is ■ Mass – amount of matter that something has ■ Volume– amount of space that something takes ■ State of matter – physical forms in which a substance can exist ■ Melting point – temperature at which a substance changes from solid to liquid ■ Boiling point – temperature at which a substance changes from liquid to gas ■ Freezing point – temperature at which a substance changes from liquid to solid ■ Density– the mass of a substance compared to its volume ■ Solubility - whether it mixes (dissolvable) in water

Separating out Mixtures using Physical properties Example A) Sand salt are poured into a beaker. The result is a mixture b/c They are not chemically combined B) When water is added to the sandsalt mixture, Salt dissolves in water. Sand does not. C) When the liquid is poured through a filter, Salt water passes through the filter D) The remaining salt water is heated until Water is boiled off (evaporated) leaving salt behind

Physical Separation Techniques ▪ Filtration – separates an insoluble solid from a liquid. The solid remains in the filter paper while the liquid goes through the paper into the beaker. ▪ Distillation – Separates mixtures of liquids or purifies a liquid. The mixture is heated and the liquid with the lowest boiling pt. will boil and the vapor will be cooled and condensed into another flask. ▪ Evaporation – separates a liquid from a solid.

Chemical Properties of Matter ■ Describe a change that happens when two substances react with each other ■ Ex. Flammability, reactivity with other chemicals, corrosion with oxygen

Physical vs. Chemical Change ■ Physical Change Do not result in new substances: ■ ■ ■ Only physical properties change not chemical properties. Generally easy to reverse ■ Chemical Change Results in a new substance ■ ■ ■ Both physical and chemical properties change. Not generally reversible

Identifying a Chemical vs Physical Change ■ ■ ■ Change in state Change in shape Change in the amount of particles (mass) ■ Chemical Change ■ ■ ■ Production of odor Production of sound Release of heat or light Release of bubbles Color change

Warm-Up: Classify the following as a physical or a chemical change: ■ ■ Na. Cl (table salt) dissolves in water. An apple is cut Backing soda reacts with vinegar Rust forms on a nail ■ ■ ■ Water evaporates Bleaching your hair Mixing oil and water Fireworks exploding Crushing a can

December 3 rd Warm-Up ▪ Question 1: Starting with a mixture of a blue liquid and a clear liquid. If the blue liquid’s boiling point is 70ᵒC and the clear liquid’s boiling point is 120ᵒC. How can the two liquids be separated? ▪ Question 2: Is the following an element, compound, or mixture? ▪ Question 3: is it a liquid or a solid?

19

A few more… ▪ Q: Where does one put dirty dishes? ▪ A: In the “zinc” ▪ Q: What weapon can you make from the elements of potassium, nickel, and iron? ▪ A: KNi. Fe ▪ Q: What is the difference between cooking and chemistry? ▪ A: You should never lick the spoon 20

Element, Compound or Mixture?

Element, Compound or Mixture?

Element, Compound or Mixture?

Element, Compound or Mixture?
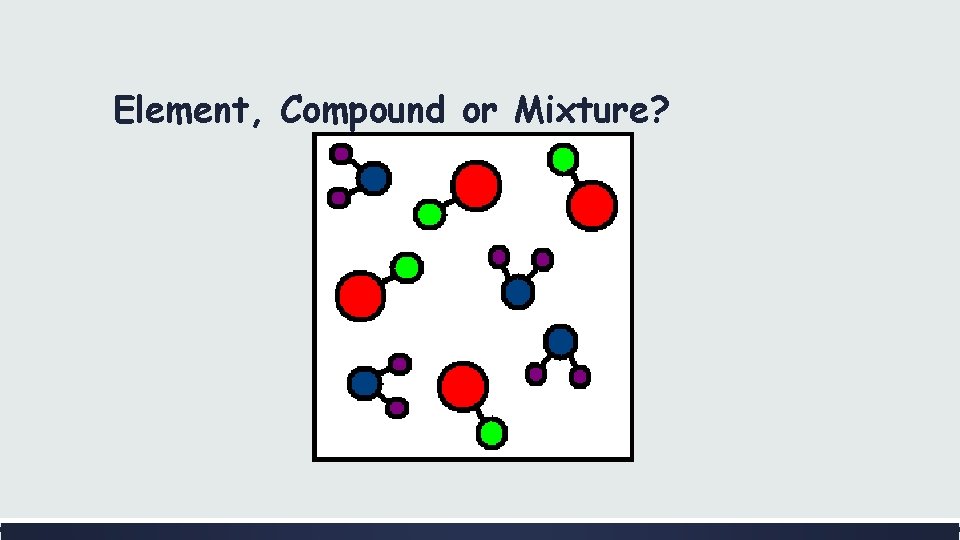
Element, Compound or Mixture?

Element, Compound or Mixture?

Classifying Matter Quick Quiz 1. This substance consists of 2 or more elements chemically combined. 2. This substance cannot be broken down by physical or chemical means. 3. The composition of this substance is variable and its physical properties depend on its composition. 4. This substance can be homogenous or heterogenous.
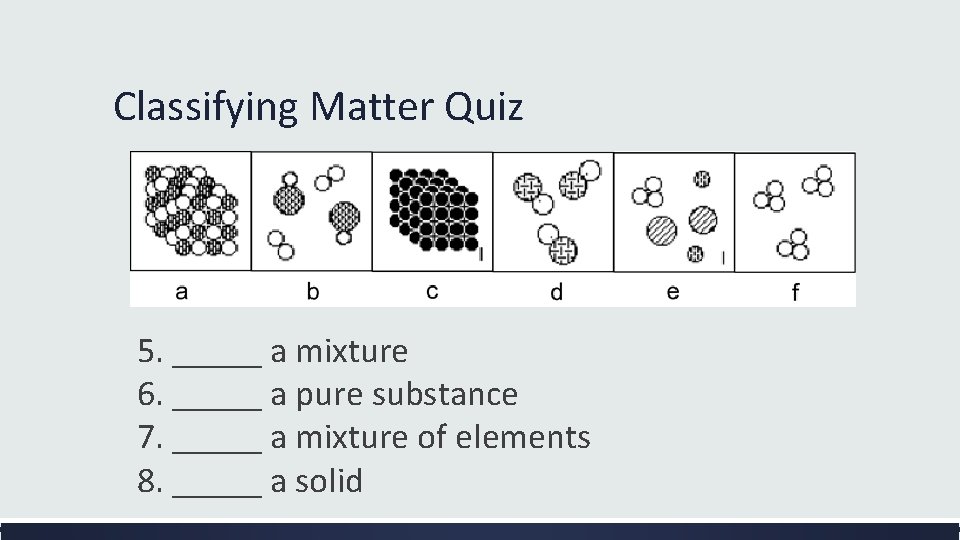
Classifying Matter Quiz 5. _____ a mixture 6. _____ a pure substance 7. _____ a mixture of elements 8. _____ a solid

December 4 th Warm-Up 1. The composition of this substance is variable and its physical properties depend on its composition. 2. What are the 2 subcategories under pure substances? 3. Which are considered pure substances?

The Development of Atomic Theory Essential Questions: 1. Who are the 6 scientist that contributed to the model of the atom? 2. What were their respective atom models? 3. What experiment are they known for?

What do Dinosaurs and Atoms have in Common? (Do not write in notes) No one has seen an atom or a dinosaur directly. We know of their existence only by indirect evidence. Our theories of both dinosaurs and atoms has changed over time based on this indirect evidence

Who was Democritus? Democritus was an ancient Greek philosopher who lived from 460 - 370 B. C. What did Democritus conclude about cutting matter in half? There was a limit to how far you could divide matter. You would eventually end up with a piece of matter that could not be cut.

What does the Greek word atomos mean? The Greek word “atomos” means Not able to be divided or “indivisible. ”

What did Democritus propose about the atom? ▪ Atoms are small hard particles. ▪ Made of a single material that’s formed into different shapes and sizes. ▪ They are always moving ▪ They form different materials by joining together.

Why weren’t Democritus’s ideas accepted? (summarize in notes) ▪ Aristotle was a very famous Greek philosopher who believed that matter could be divided into smaller and smaller pieces forever. He held a very strong influence on popular belief and his views on this were accepted for two thousand years.

John Dalton 1776 -1844 ▪ Two thousand years later a British chemist and schoolteacher brings back Democritus’s idea of the atom ▪ He performed many experiments to study how elements join together to form new substances ▪ He found that atoms combine in specific ratios and he supposed it was because the elements are made of atoms.

What 3 new ideas did John Dalton propose about the atom? ▪ All substances are made up of atoms which are small particles that cannot be created, divided, or destroyed. ▪ Atoms of the same element are exactly alike and atoms of different elements are different. ▪ Atoms join with other atoms to form different substances
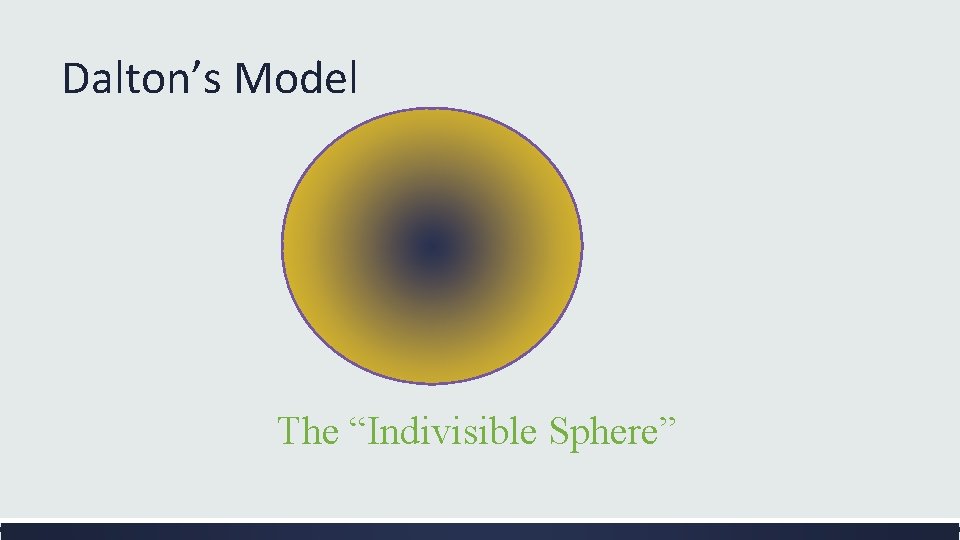
Dalton’s Model The “Indivisible Sphere”

J. J. Thomson 1856 -1940 ▪ What particle did Thomson discover? J. J. Thomson discovered that atoms are made of smaller negatively-charged particles called electrons. ▪ Thomson’s discovery was the result of doing experiments with “cathode ray tubes”

Thomson’s Cathode Ray Experiment Stream of electrons is attracted to positively charged plate here. "What are these particles? are they atoms, or molecules, or matter in a still finer state of subdivision? “ quote by Thomson

The Plum Pudding Model ▪ Thomson did not know how the electrons in an atom were arranged. He believed they were mixed throughout an atom. ▪ He proposed that the atom was a sphere of positively charged material. Spread throughout the atom were the negatively charged electrons similar to plums in a pudding or chocolate chips in ice cream.
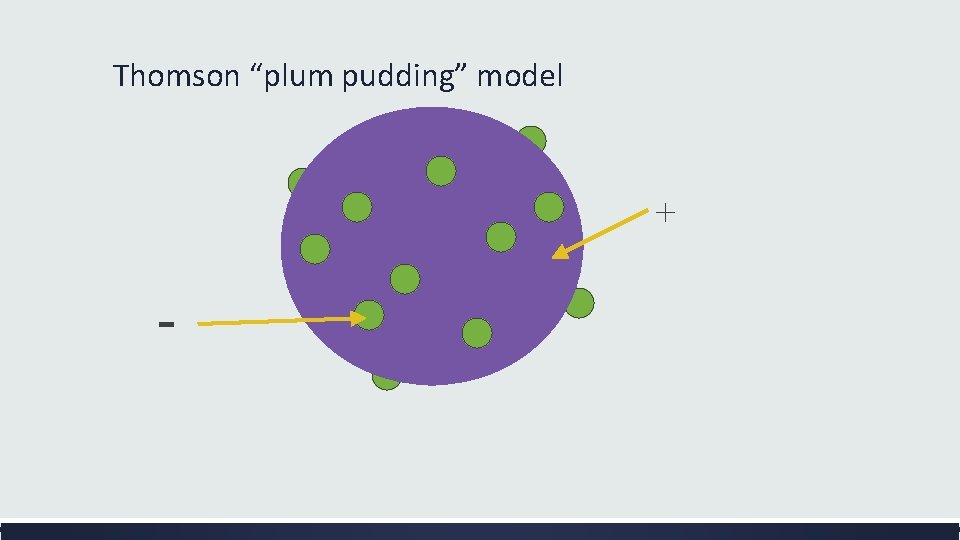
Thomson “plum pudding” model + -

Ernest Rutherford (1871 - 1937) ▪ Awarded the Nobel Prize in Chemistry for his discovery of alpha particles, positively charged particles emitted from radioactive elements ▪ Was a student of J. J. Thomson but disagreed with the “Plum Pudding Model” ▪ Devised an experiment to investigate the structure of positive and negative charges in the atom.
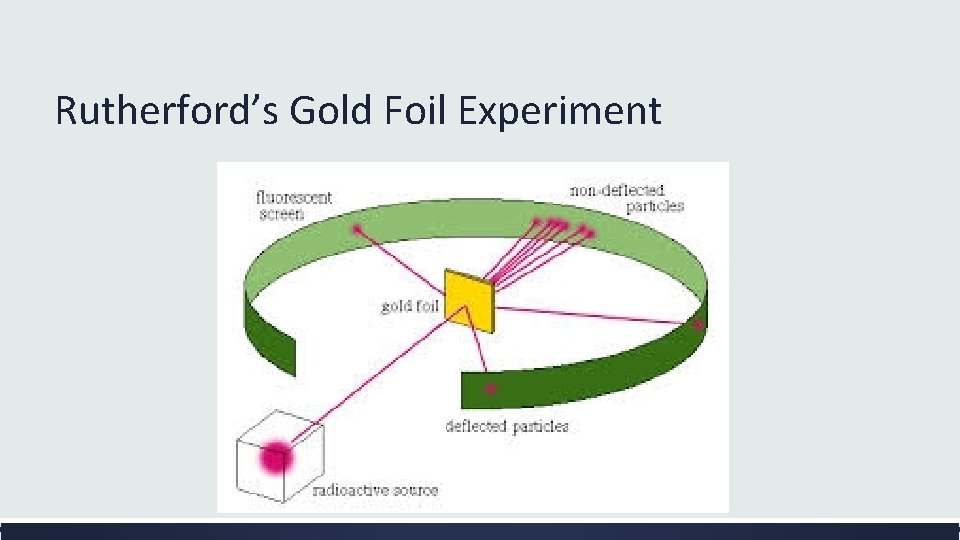
Rutherford’s Gold Foil Experiment

What did most of the particles shot at the gold foil do? ▪ Most of the particles traveled straight through the gold foil What was the surprising behavior of a few of the particles? ▪ A few of the particles were deflected and some even bounced back

Rutherford’s Revised Atomic Theory (1911) Result: Most of the positively charged particles went straight through the gold foil. Atomic Theory : Most of the matter of the atom is found in a very small part of the atom. This is called the nucleus of the atom. It is very tiny and extremely dense. Result: Some of the positively charged particles were deflected or even bounced back. Atomic Theory: Like charges repel so the nucleus must have a positive charge. If electrons have a negative charge they could not be in a positively charged nucleus. Electrons must surround the nucleus at a distance. Result: The diameter of the nucleus is 100, 000 times smaller than the diameter of the entire gold atom. Atomic Theory: Atoms are mostly empty space with a tiny, massive nucleus at the center.

Why is the head of a pin compared to the diameter of a stadium like an atom? The diameter of a pinhead is 100, 000 times smaller than the diameter of a stadium. Likewise the diameter of the nucleus of an atom is 100, 000 times smaller than the diameter of an atom
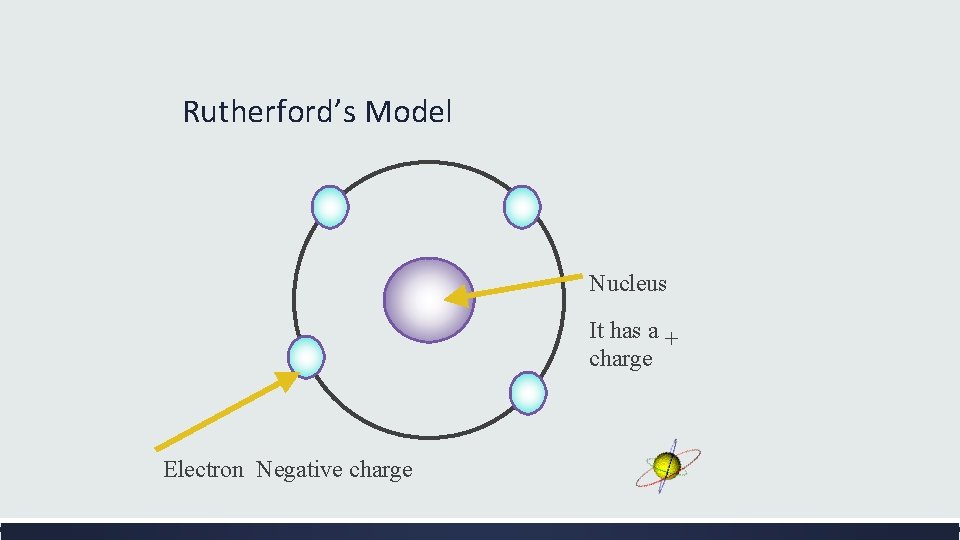
Rutherford’s Model Nucleus It has a + charge Electron Negative charge

The Bohr Model of the Atom (1913) What did Bohr learn about electron movement? ▪ Bohr proposed that electrons move in paths at certain distances around the nucleus. ▪ Electrons can jump from a path on one level to a path on another level. ▪ Click here http: //www. colorado. edu/ physics/2000/quantumzon e/bohr. html
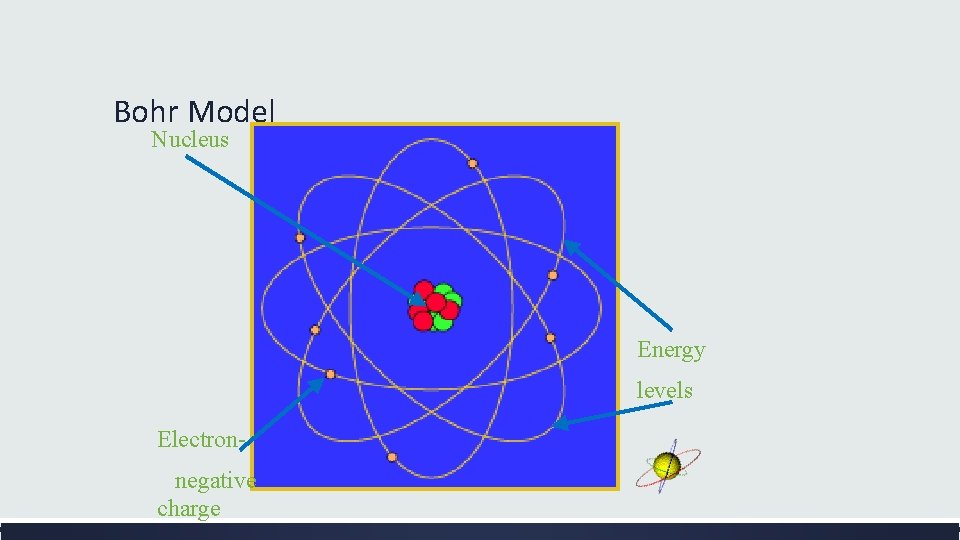
Bohr Model Nucleus Energy levels Electronnegative charge

The Modern Quantum Cloud Theory of the Atom ▪ Electrons travel in regions called “electron clouds” ▪ You cannot predict exactly where an electron will be found http: //www. fearofphysics. com/Atom/atom 3. html
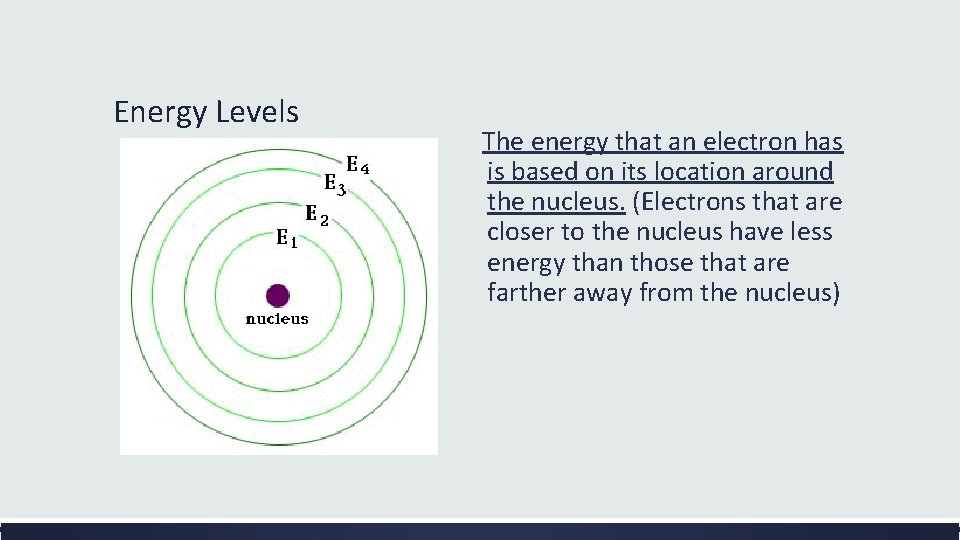
Energy Levels The energy that an electron has is based on its location around the nucleus. (Electrons that are closer to the nucleus have less energy than those that are farther away from the nucleus)

How can bookshelves help you understand the movement of electrons? • Each shelf represents an energy level • Each book represents an electron • You can move a book to a higher or lower shelf with the correct amount of energy. • A book cannot be between shelves (An electron can move by gaining or losing energy but can never be between energy levels)

How small are atoms? THERE ARE 2 X 1022 ATOMS IN A PENNY. If all the atoms in a penny were blown up to the size of a grain of sand they would cover the entire state of California

What can a scanning tunneling electron microscope show us? ▪ These images do not show an actual picture of an atom. They show a color-enhanced image of the surface of a material at the atomic level.

December th 7 Warm-Up ▪ Question 1: What experiment did Rutherford perform? ▪ Question 2: How did Chadwick expand on the atomic model? ▪ Question 3: What did J. J. Thomson discover?

Gold Foil Experiment Reading Activity ▪ We will use our reading to gain an understanding of the work that Rutherford did to discover the nucleus. ▪ Number Paragraphs ▪ Underline Key Parts as you silently read. ▪ Compare underlined key parts with a neighbor. ▪ Answer Questions 1&2. ▪ Fill out the Outline of the Lab Report.

December th 8 Warm-Up ▪ Question 1: Pure substances can be further classified as _____ and ______. ▪ Question 2: A physical change can turn a _____ into a ______. ▪ Question 3: A chemical change can turn a _____ into a ______. ▪ Question 4: What is sub-atomic particles are found inside an atom’s nucleus?
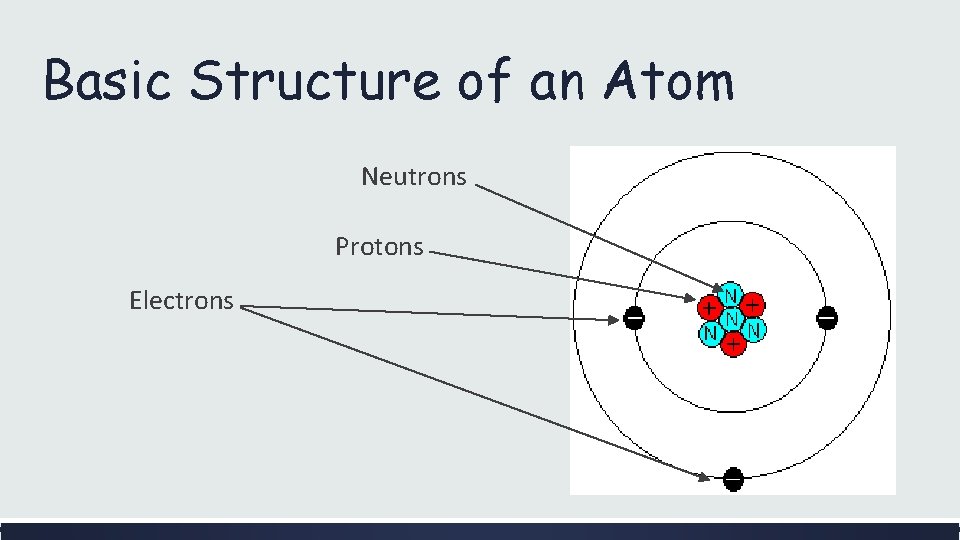
Basic Structure of an Atom Neutrons Protons Electrons
60

Introduction to the Periodic Table Essential Questions: 1. What information can we gain by one square on the periodic table? 2. What are isotopes? 3. What information can be gained by electron configuration notations?

I am Dmitri Mendeleev! I made the PERIODIC TABLE !

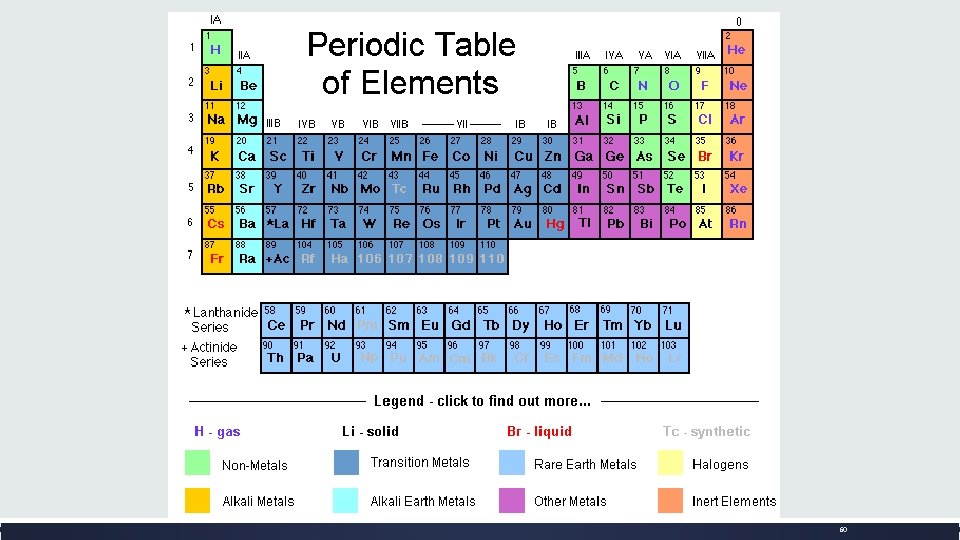
What is the PERIODIC TABLE? o. Shows all known elements in the universe. o. Organizes the elements by chemical properties.

How do you read the PERIODIC TABLE?

What is the ATOMIC NUMBER? o. The number of protons found in the nucleus of an atom Or o. The number of electrons surrounding the nucleus of an atom.
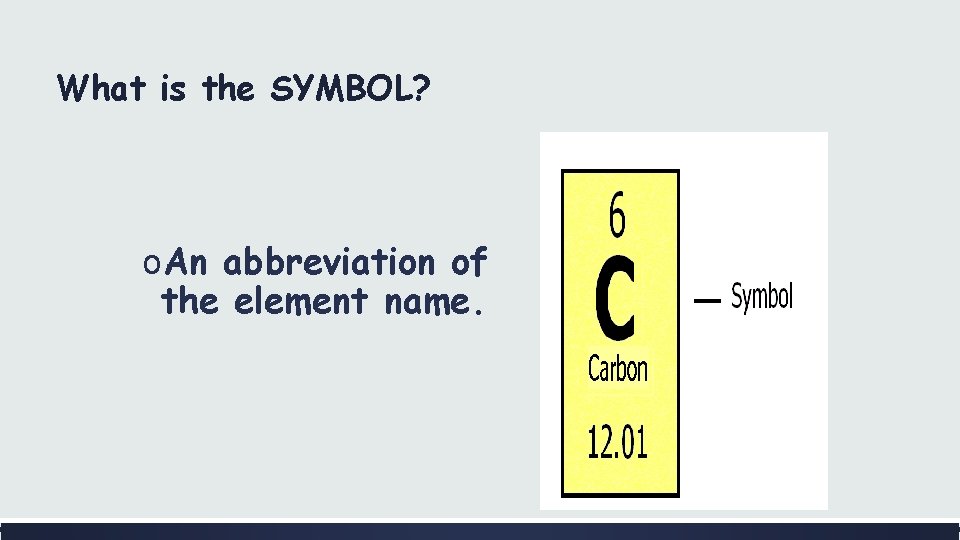
What is the SYMBOL? o. An abbreviation of the element name.
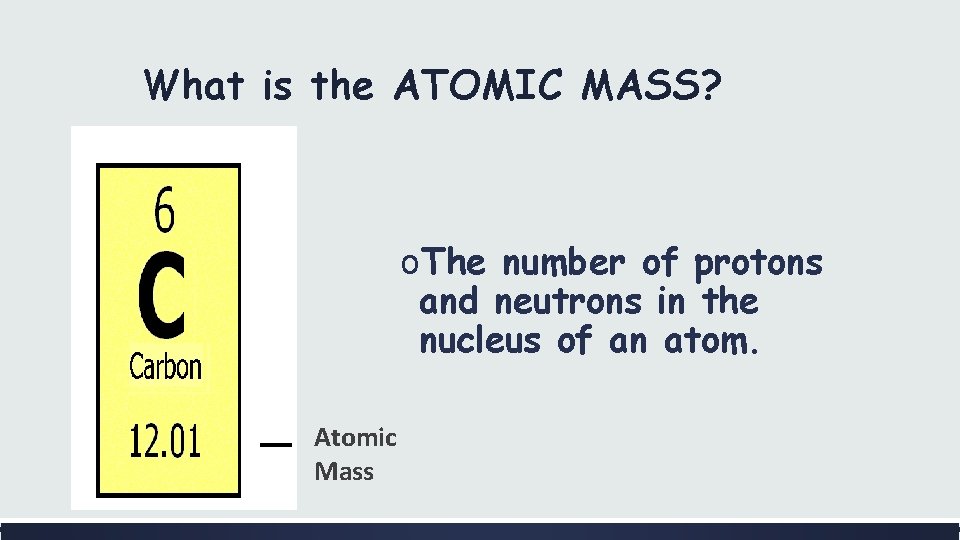
What is the ATOMIC MASS? o. The number of protons and neutrons in the nucleus of an atom. Atomic Mass

How do I find the number of protons, electrons, and neutrons in an element using the periodic table? o # of PROTONS (+)= ATOMIC NUMBER o # of ELECTRONS (-)= ATOMIC NUMBER IN A NEUTRAL ATOM o# of NEUTRONS (no charge) = ATOMIC _ ATOMIC MASS NUMBER
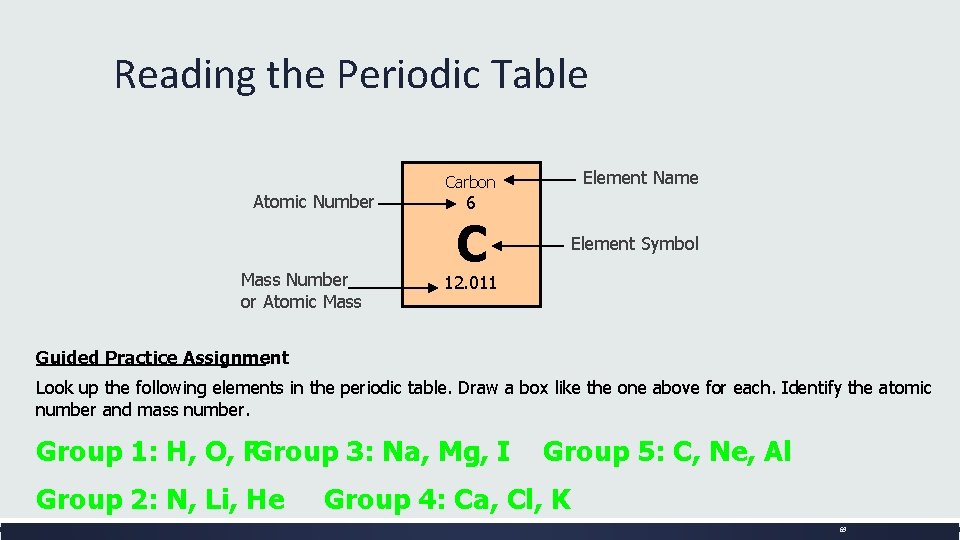
Reading the Periodic Table Atomic Number Mass Number or Atomic Mass Element Name Carbon 6 C Element Symbol 12. 011 Guided Practice Assignment : Look up the following elements in the periodic table. Draw a box like the one above for each. Identify the atomic number and mass number. Group 1: H, O, FGroup 3: Na, Mg, I Group 2: N, Li, He Group 5: C, Ne, Al Group 4: Ca, Cl, K 69
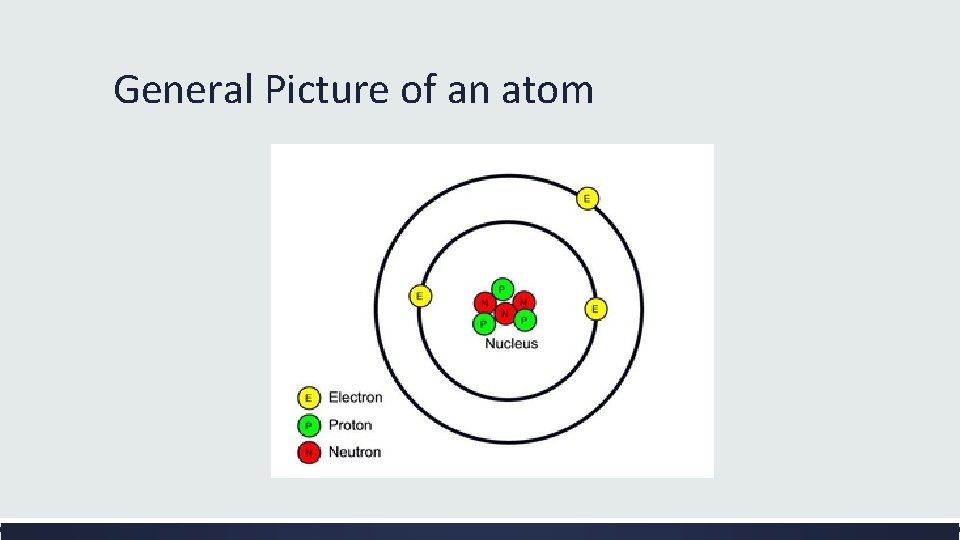
General Picture of an atom

Isotopes ▪ An ISOTOPE is a form of an element that has a different number of NEUTRONS than “normal” ▪ Carbon has three isotopes Notice how the # of protons does NOT change!

Isotopes of Oxygen Naturally occurring oxygen consists of three isotopes, 16 O, 17 O, and 18 O. State the number of protons, neutrons, and electrons in each of these carbon atoms. 16 O 8 17 O 8 18 O 8 #P _______ #N _______ #E _______

Solution 16 O 8 17 O 8 18 O 8 #P _8_ 8_ _8_ #N _8_ _9_ _10_ #E _8_ _8 _

Now you are almost as smart as I am! But not as handsome! Man, I look GOOD!

Electron Configuration ▪ Electrons are found energy levels. They are numbered 17 with 1 being the closest to the nucleus and 7 being the furthest. ▪ Within each energy level there are sub-levels (shells). These are lettered as s, p, d, f. ▪ The pattern goes: 1 s 2 s 2 p 3 s 3 p 4 s 3 d… ▪ The exponents are the number of electrons in each energy level. ▪ The total of the exponents tells you the number of electrons in the atom. ▪ Ex: Carbon has 6 electrons and it’s electron configuration is : 2 2 2 1 s 2 s 2 p

Electron Configuration Notation for Atoms ▪ Electron configuration notation specifies how many electrons an atom has and how many electrons are in each energy level and sub-level. ▪ EX: ▪ Nitrogen’s (N) electron configuration: 1 s 22 p 3 ▪ ▪ Total # of electrons = # of electrons in 1 st energy level = # of electrons in 2 nd energy level= What is the element with the following configuration: (Hint: count the exponents) ▪ 1 s 22 p 63 s 2?

December 9 th Warm-Up ▪ Question 1: How many electrons are in the following electron configuration? 1 s 22 p 63 s 2 ▪ Question 2: What element is represented in question 1? ▪ Question 3: How many neurons does Cl have? 35 Cl 7 ▪ Question 4: Who developed the periodic table?

Bohr’s Model Activity ▪ Discover how Bohr developed his model of the atom ▪ Fill out hand-out while we perform the activity


▪ Get out Review Guide ▪ Work on vocabulary ▪ Play Kahoot Game – Ch 1 ▪ What to study: Review ▪ Classifying matter ▪ History of atomic theory. . ▪ Atomic structure ▪ Reading the Periodic table

Test
- Slides: 81