Describing Chemical Reactions Chemical Reactions l A chemical

Describing Chemical Reactions

Chemical Reactions l A chemical reaction is the process by which one or more substances change into one or more new substances. l Reactants are the original substances in a chemical reaction. l Products are the substances that are created in a chemical reaction.

Evidence of a chemical change

Evidence of a chemical change

Chemical v. Physical l chemical change – new substance forms with properties that differ from original substance – – – l density boiling point melting point physical change - changes of state – – evaporation condensation melting freezing

Constructing a Chemical Rxn l A chemical equation shows the chemical formulas and relative amounts of all reactants and products. l A word equation contains the names of the reactants and products. l Equations must be balanced.
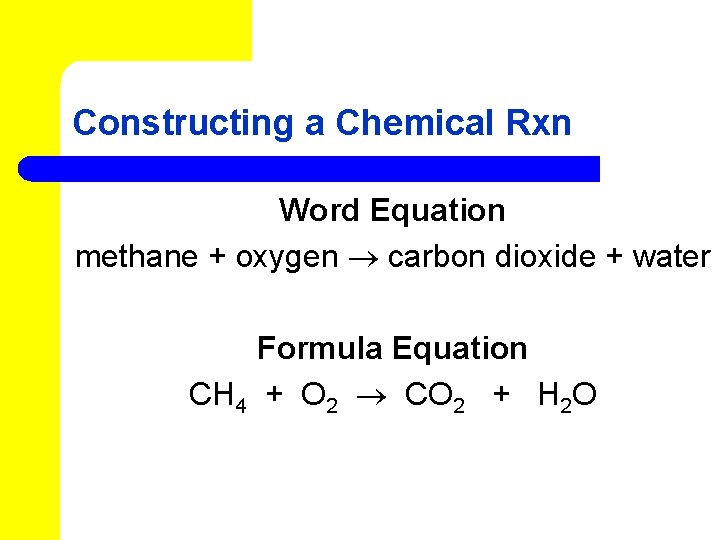
Constructing a Chemical Rxn Word Equation methane + oxygen carbon dioxide + water Formula Equation CH 4 + O 2 CO 2 + H 2 O

Constructing a Chemical Rxn Other symbols

The Great Balancing Act

Think… l Law of Conservation of Mass states that mass cannot be created or destroyed by a chemical or physical change l Therefore, ALL chemical equations must be balanced. That is, having the same number of atoms on both sides of the arrow

Balancing Equations l l The number of atoms for each element must be the same on the reactants’ side and on the products’ side. A coefficient multiplies the number of atoms of each element in the formula that follows. H 2 O: 2 hydrogen atoms, 1 oxygen atom 2 H 2 O: 4 hydrogen atoms, 2 oxygen atoms

Summarize the steps

Summarize the steps

The 5 Reaction Types

Synthesis Reactions In a synthesis reaction a single compound forms from two or more reactants. Two elements form a binary compound C + O 2 CO 2 2 C + O 2 2 CO Two compounds form a ternary compound Ca. O(s) + H 2 O(l) Ca(OH)2(s) CO 2(g) + H 2 O(l) H 2 CO 3(aq)

Decomposition Reactions In a decomposition reaction a single compound breaks down, often with the input of energy, into two or more elements or simpler compounds. Decomposition of water 2 H 2 O(l) electricity O 2(g) + 2 H 2(g) A metal carbonate decomposes to form a metal oxide and carbon dioxide. Ca. CO 3(s) heat Ca. O(s) + CO 2(g)

Sample Problem l Predict the product(s) and write a balanced equation for the reaction of potassium with chlorine.

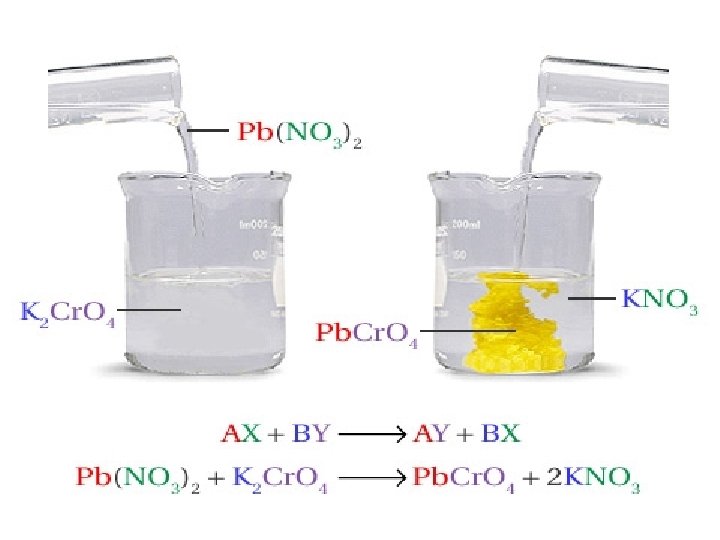
Double-Displacement Reactions • In a double-displacement reaction two compounds in aqueous solution appear to exchange ions and form two new compounds. • One of the products must be a solid precipitate, a gas, or a molecular compound, such as water. HCl(aq) + Na. OH(aq) HOH(l) + Na. Cl(aq)

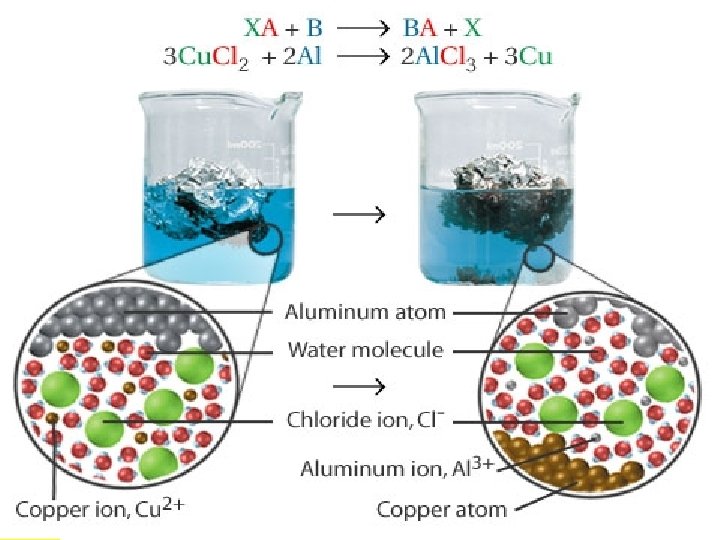
Displacement Reactions In a displacement reaction a single element reacts with a compound and displaces another element from the compound. 2 Al(s) + 3 Cu. Cl 2(aq) 2 Al. Cl 3(aq) + 3 Cu(s) Aluminum displaces copper.


Sample Problem • Determining Products by Using the Activity Series Magnesium is added to a solution of lead(II) nitrate. Will a reaction happen? If so, write the equation and balance it.

Combustion Reactions A combustion reaction is a reaction of a carbon based compound with oxygen. Combustion of propane C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O Combustion of ethanol CH 3 CH 2 OH + 3 O 2 2 CO 2 + 3 H 2 O
- Slides: 24