Describe what you can see happening when a

* ** *** Describe what you can see happening when a displacement reaction happens State which metal is more reactive in a pair of metals Use the reactivity series to predict if a pair of substances will undergo a displacement reaction
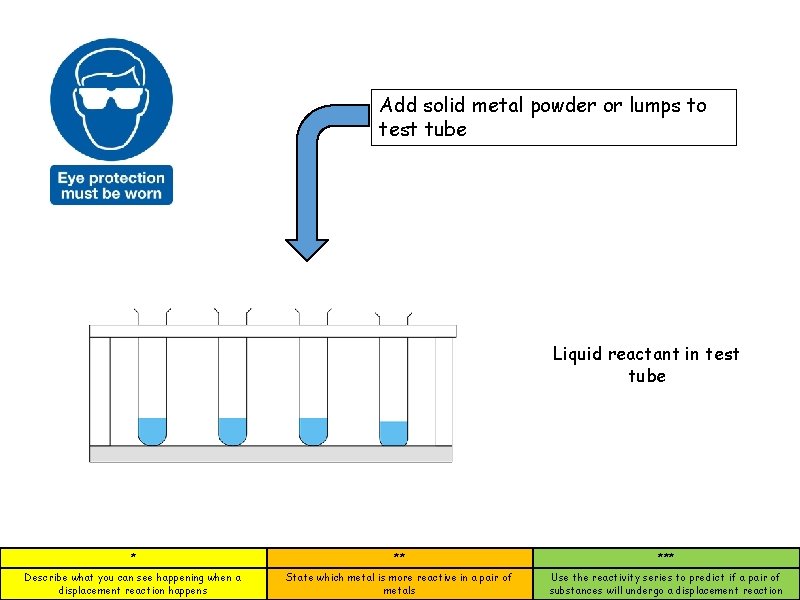
Add solid metal powder or lumps to test tube Liquid reactant in test tube * ** *** Describe what you can see happening when a displacement reaction happens State which metal is more reactive in a pair of metals Use the reactivity series to predict if a pair of substances will undergo a displacement reaction
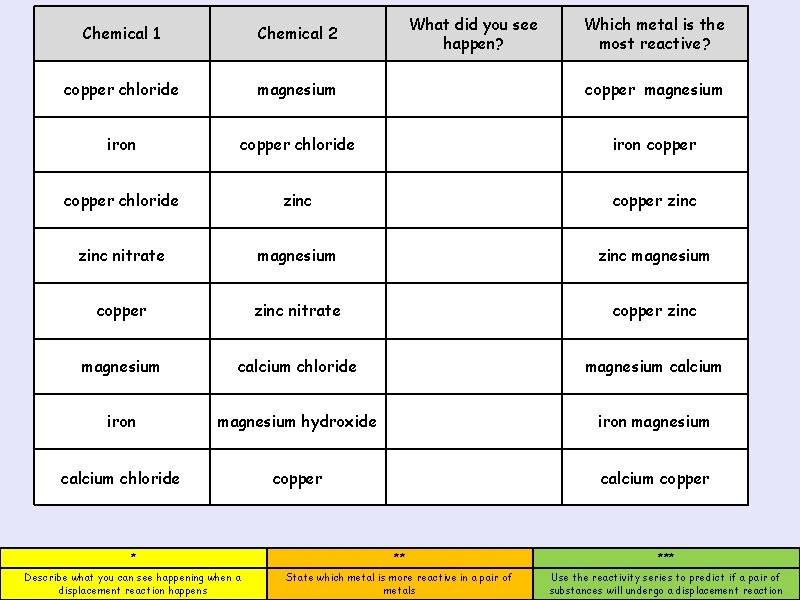
What did you see happen? Which metal is the most reactive? Chemical 1 Chemical 2 copper chloride magnesium copper magnesium iron copper chloride zinc copper zinc nitrate magnesium zinc magnesium * ** copper Describe what you can see happening when a displacement reaction happens (L 3) *** zinc nitrate State which metal is more reactive in a pair of metals (L 4) Use the reactivity series to predict if a pair of substances will undergo a displacement reaction (L 5) copper zinc magnesium calcium chloride magnesium calcium iron magnesium hydroxide iron magnesium calcium chloride copper calcium copper * ** *** Describe what you can see happening when a displacement reaction happens State which metal is more reactive in a pair of metals Use the reactivity series to predict if a pair of substances will undergo a displacement reaction
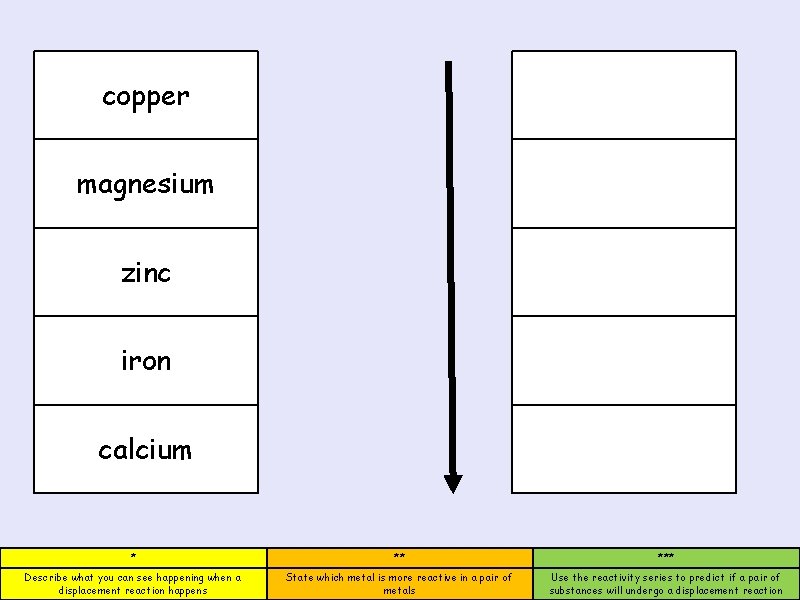
copper magnesium zinc iron calcium * ** *** Describe what you can see happening when a displacement reaction happens State which metal is more reactive in a pair of metals Use the reactivity series to predict if a pair of substances will undergo a displacement reaction
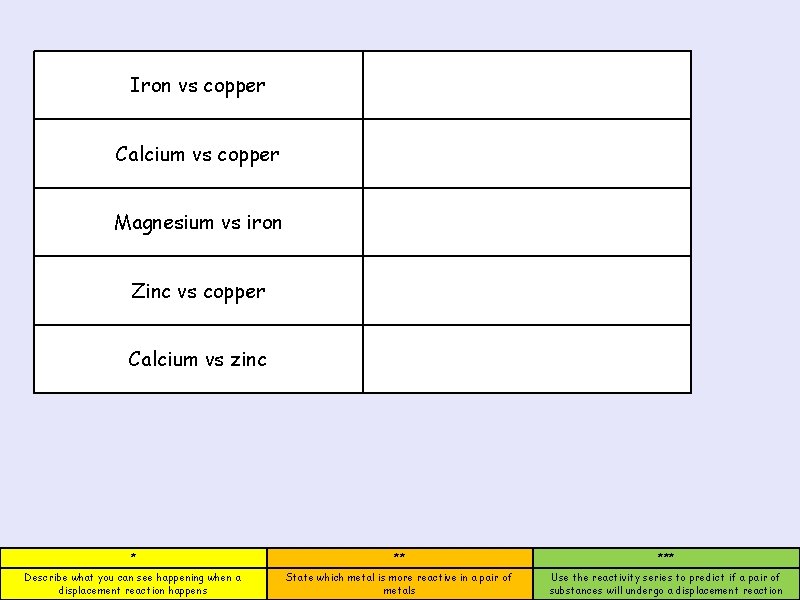
Iron vs copper Calcium vs copper Magnesium vs iron Zinc vs copper Calcium vs zinc * ** *** Describe what you can see happening when a displacement reaction happens State which metal is more reactive in a pair of metals Use the reactivity series to predict if a pair of substances will undergo a displacement reaction

Predict if there would be a displacement reaction: 1. Copper sulfate and magnesium 2. Magnesium nitrate and calcium 3. Zinc chloride and copper 4. Copper and calcium chloride 5. Copper sulfate and iron * ** *** Describe what you can see happening when a displacement reaction happens State which metal is more reactive in a pair of metals Use the reactivity series to predict if a pair of substances will undergo a displacement reaction
- Slides: 6