DES Bioabsorbable and DCB Technologies CRT February 25

DES Bioabsorbable and DCB Technologies CRT February 25 th 2013, Washington DC Martin T Rothman MB, Ch. B, FACC FESC FRCP Vice President Medical Affairs Coronary & Renal Denervation Medtronic Inc. Professor of Interventional Cardiology Barts Health London, UK

Martin T. Rothman, MD, MB, Ch. B I/we have no real or apparent conflicts of interest to report.

Integrity build animation For distribution only in markets where the Integrity coronary stent is approved. Medtronic, Inc. All rights reserved. UC 201004771 EN 7/10 3

Continuous Sinusoid Technology The Next Revolution in Stent Technology Enabling designs that could not be attempted in the past Bare Metal Stents Integrity S 10 Core Wire New Alloys Drug-Eluting Stents Resolute Integrity Platforms for next gen DES coatings • Bio. Linx (Resolute) • PC Technology (Endeavor) • Bioabsorbable polymer • Nanoporous CAUTION: Some Design elements not approved for sale or clinical use
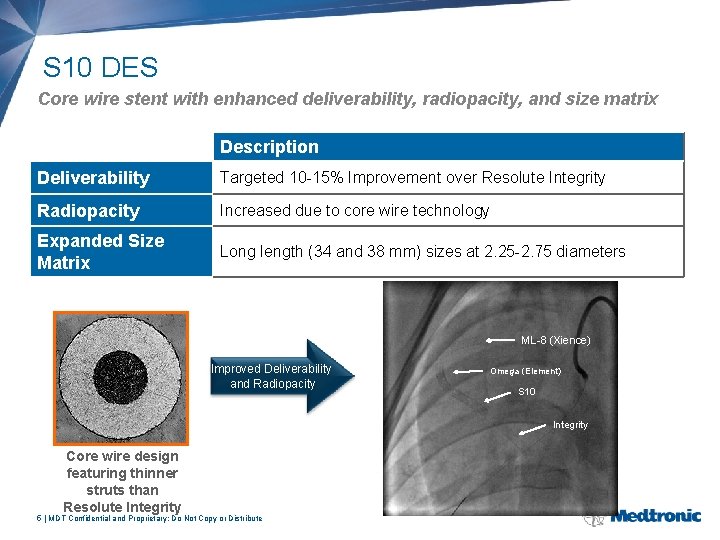
S 10 DES Core wire stent with enhanced deliverability, radiopacity, and size matrix Attribute Description Deliverability Targeted 10 -15% Improvement over Resolute Integrity Radiopacity Increased due to core wire technology Expanded Size Matrix Long length (34 and 38 mm) sizes at 2. 25 -2. 75 diameters ML-8 (Xience) Improved Deliverability and Radiopacity Omega (Element) S 10 Integrity Core wire design featuring thinner struts than Resolute Integrity 5 | MDT Confidential and Proprietary; Do Not Copy or Distribute
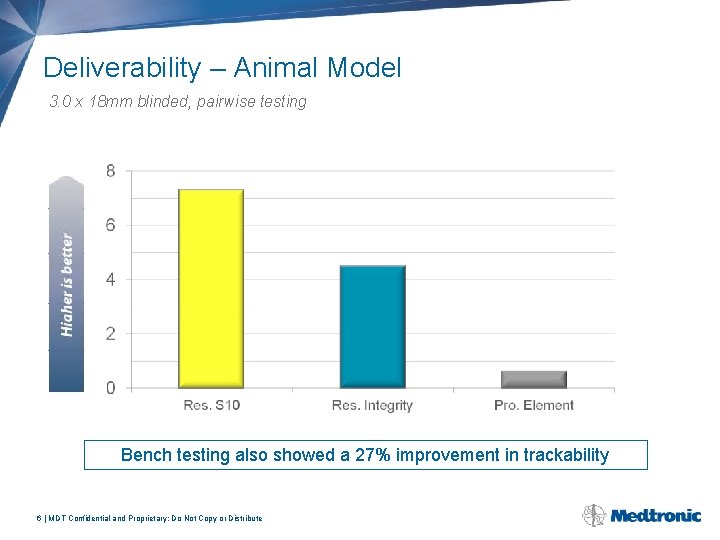
Deliverability – Animal Model 3. 0 x 18 mm blinded, pairwise testing Bench testing also showed a 27% improvement in trackability 6 | MDT Confidential and Proprietary; Do Not Copy or Distribute


If it catches, what happens? S 10 Platform Element Platform 8 | MDT Confidential and Proprietary; Do Not Copy or Distribute

Bioabsorbable Stent Technology Objective: DES-like performance with clinically relevant degradation time, workhorse performance without sacrificing critical design targets Design Targets Medtronic Goal Efficacy equivalent to DES In all lesions treated by DES Adequate radial strength 3 -6 months Appropriate degradation time 6 -12 months Anticipated DAPT duration Equal or better than current DES Adequate deliverability & conformability Similar to current DES Minimal change to implant procedure Similar to current DES Polymer Metal § Increased strut size & crossing profile § Modestly increased strut size & crossing profile § 2+ years degradation § 6 -12 m degradation § Extended DAPT? § Low inflammation § Complex deployment § Simple DES-like deployment -Slow inflation -Limited post-dilation -Potential for malapposition 9 | MDT Confidential

Bioabsorbable Stent Technology Proprietary Alloy • Balance of consistent performance, mechanical strength/workability, and surface treatment compatibility Absorption control Demonstrating controlled dissolution of stent Surface treatments control degradation Target: 3 -6 mo. structural integrity 12 mo. full degradation Day 0 Strut cross-section after degradation begins to occur 10 | MDT Confidential

Bioabsorbable Stent Technology Promising in-vivo results demonstrated with multiple surface treatment / alloy combinations In vivo Corrosion Treatment 1 OCT at 30 d Treatment 1 Micro. CT at 30 d Good wall apposition and no visible corrosion Consistent diameter, good vessel support Biocompatibility Treatment 1 @ 30 d Treatment 2 @ 30 d Proven good biocompatibility of the alloy and the surface treatments Arm Description Stenosis % Neointimal Thickness Treatment 1 24. 20 0. 19 Treatment 2 22. 47 0. 17 Integrity BMS 19. 79 0. 16 11 | MDT Confidential

Continuous Sinusoid Technology The Next Revolution in Stent Technology Enabling designs that could not be attempted in the past Bare Metal Stents Integrity Drug-Eluting Stents Drug-Filled Stent Resolute Integrity CAUTION: Design concept not approved for sale or clinical use Resolute and Resolute Integrity are not approved for clinical use in the US

Drug Filled Stent (DFS) Technology Polymer Free Drug Delivery Innovative DES design • Essentially a BMS surface • Designed to address drug carrier issues such as: § Polymer biocompatibility § Inflammation upon polymer degradation § Surface coating durability CAUTION: Design concepts not approved for sale or clinical use
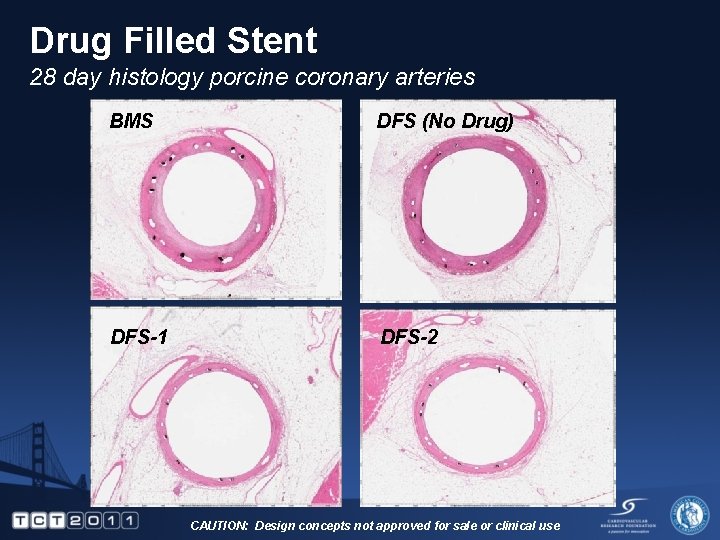
Drug Filled Stent 28 day histology porcine coronary arteries BMS DFS (No Drug) DFS-1 DFS-2 CAUTION: Design concepts not approved for sale or clinical use
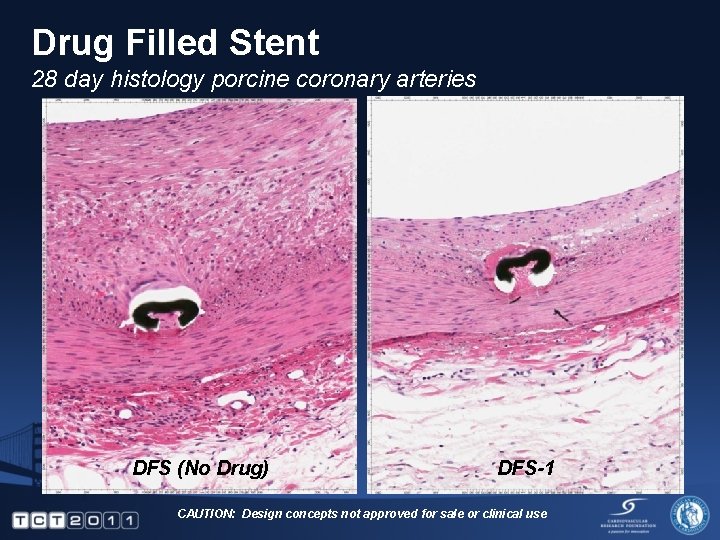
Drug Filled Stent 28 day histology porcine coronary arteries DFS (No Drug) DFS-1 CAUTION: Design concepts not approved for sale or clinical use

DES, Biodegradable, DFS • Program for next generation DES well advanced ¡ Enhanced resistance to longitudinal stent deformation • Biodegradable metal stent ¡ Early data very encouraging • Drug filled stent ¡ Advanced program with excellent data
- Slides: 16