Department of Glycobiology Major research areas a Identifying

• Department of Glycobiology • Major research areas: a) Identifying of putative N-, and O-glycan biomarkers related to disease pathogenesis: aplication of glyco-proteomics and glyco-genomics methods to gain understanding of aberrant glycosylation associated with various physiological and pathophysiological status (Inborn Metabolic Disorders and Cancer) * Inherrited metabolic disorders of glycoconjugate metabolism - Lysosomal storage disorders (LSD) glycogenosis and Pompe disorder: Caused by defects in degradation of glycoconjugates – >40 known subtypes – Characteristic by specific metabolite accumulated in tissues/body fluids - Congenital disorders of glycosylation (subtypes of CDGs): Caused by defects in glycan biosynthesis and processing – >70 known subtypes – Characteristic by specific aberrant N-, and O- glycan structures in tissues/body fluids * The structure of glycan alternation associated with colorectal cancer. Whole blood serum glycoprofiling and structure of O- glycans in pathogenesis.
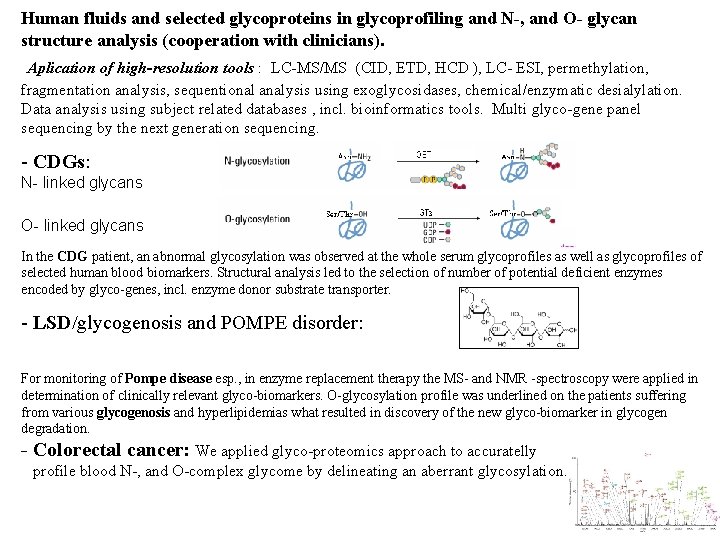
Human fluids and selected glycoproteins in glycoprofiling and N-, and O- glycan structure analysis (cooperation with clinicians). Aplication of high-resolution tools : LC-MS/MS (CID, ETD, HCD ), LC- ESI, permethylation, fragmentation analysis, sequentional analysis using exoglycosidases, chemical/enzymatic desialylation. Data analysis using subject related databases , incl. bioinformatics tools. Multi glyco-gene panel sequencing by the next generation sequencing. - CDGs: N- linked glycans O- linked glycans In the CDG patient, an abnormal glycosylation was observed at the whole serum glycoprofiles as well as glycoprofiles of selected human blood biomarkers. Structural analysis led to the selection of number of potential deficient enzymes encoded by glyco-genes, incl. enzyme donor substrate transporter. - LSD/glycogenosis and POMPE disorder: Oligosaccharide metabolite product For monitoring of Pompe disease esp. , in enzyme replacement therapy the MS- and NMR -spectroscopy were applied in determination of clinically relevant glyco-biomarkers. O-glycosylation profile was underlined on the patients suffering from various glycogenosis and hyperlipidemias what resulted in discovery of the new glyco-biomarker in glycogen degradation. - Colorectal cancer: We applied glyco-proteomics approach to accuratelly profile blood N-, and O-complex glycome by delineating an aberrant glycosylation.

b) Transglycosylases of yeasts and plants: enzymes enabling the restructuralization of energy poor cell walls during the cell development, growth, stressinduced respond / enzymes with expected role in cryptococcal capsule rearrangement and following pathogenesis / stress-induced proteins obtained as side-products of proteomic approach to plant and yeast resources: - Development of activity assay methods based on fluorescently labelled acceptor oligosaccharides (one-pot method for acceptor labelling and activity assays as dot-blot method, assay after IEF-PAGE, high-throughput methods based on glycochips or in-gel reactions, HPLC, etc. ). - Characterization of yeasts GH 16 and GH 72 transglycosylases - Crh group (Saccharomyces cerevisiae) with focus on their substrate specificities and Phr group (Candida albicans) on importance and cooperation of individual forms in pathogenesis → possible importance as targets of new generation drugs based on inhibition of these enzymes. - Screening for glycosidases on surface of capsular yeasts, purification and characterization of an alpha-galactosidase form from Cryptococcus flavescens → the ever first lactose-inducible yeast long-chain alpha-galactosidase of GH 36 family.
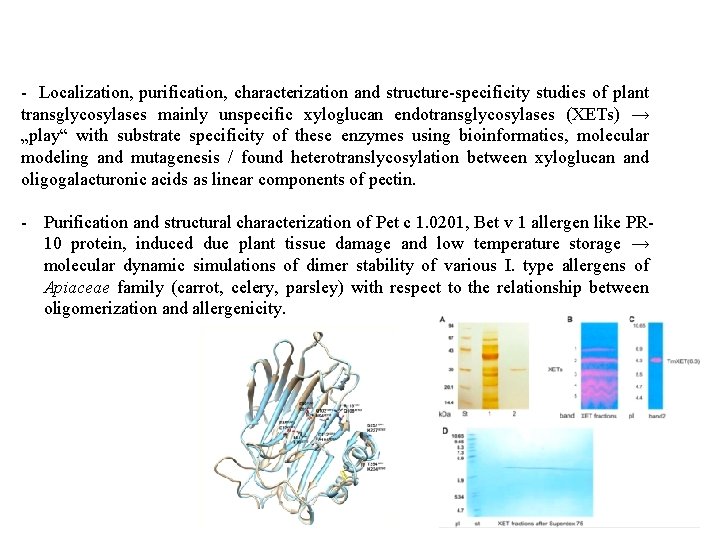
- Localization, purification, characterization and structure-specificity studies of plant transglycosylases mainly unspecific xyloglucan endotransglycosylases (XETs) → „play“ with substrate specificity of these enzymes using bioinformatics, molecular modeling and mutagenesis / found heterotranslycosylation between xyloglucan and oligogalacturonic acids as linear components of pectin. - Purification and structural characterization of Pet c 1. 0201, Bet v 1 allergen like PR 10 protein, induced due plant tissue damage and low temperature storage → molecular dynamic simulations of dimer stability of various I. type allergens of Apiaceae family (carrot, celery, parsley) with respect to the relationship between oligomerization and allergenicity.

c) Study of antimicrobial substances of royal jelly: investigation of the efficacies of major antimicrobial royal jelly substances - peptide defensin 1 and five 8 -10 carbon fatty acids, against several collection strains and field isolates of Paenibacillus larvae - bacterial pathogen causing serious honeybee brood disease American foulbrood - the disease causes economic losses to beekeepers and farmers dependent on pollination Disease-causing agent: Paenibacillus larvae - the pathogen occuring in contaminated food propagates first in midgut and then in bodies of infected larvae what causes their death - infection spreads in colony what leads to its collaps Royal jelly - food of young larvae produced by honeybees - antimicrobial substances present in it could in adequate concentrations hinder the propagation of pathogen bacteria in larval midgut Example of substances: 10 -hydroxy-2 -decenoic acid 3, 10 -dihydroxydecanoic acid Determination of minimal inhibitory concentrations of a substance by broth microdilution method
- Slides: 5