Density Section 2 3 High population density large









- Slides: 13

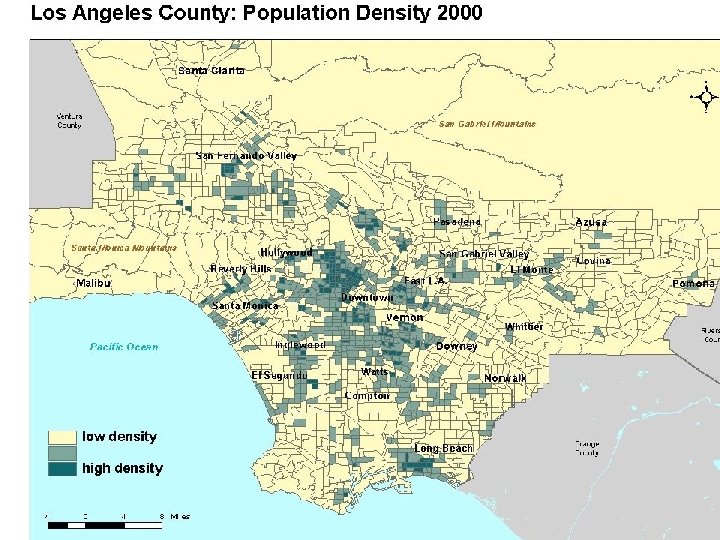
Density Section 2. 3


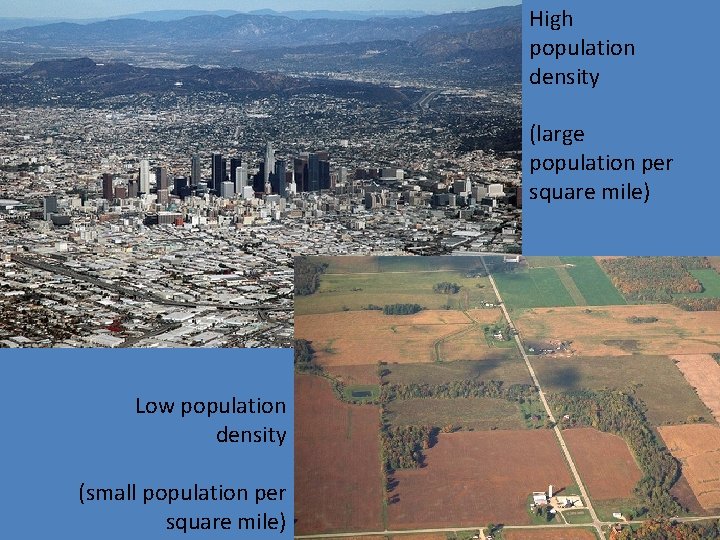
High population density (large population per square mile) Low population density (small population per square mile)

Density in Science • Definition: Mass per unit volume • Why do we need to know about density? – Density is a physical property of elements and compounds. – Density is easy to measure using a balance and a ruler (for large objects) and a graduated cylinder & water (for small objects) – Unknown elements and compounds can be identified by finding their density!

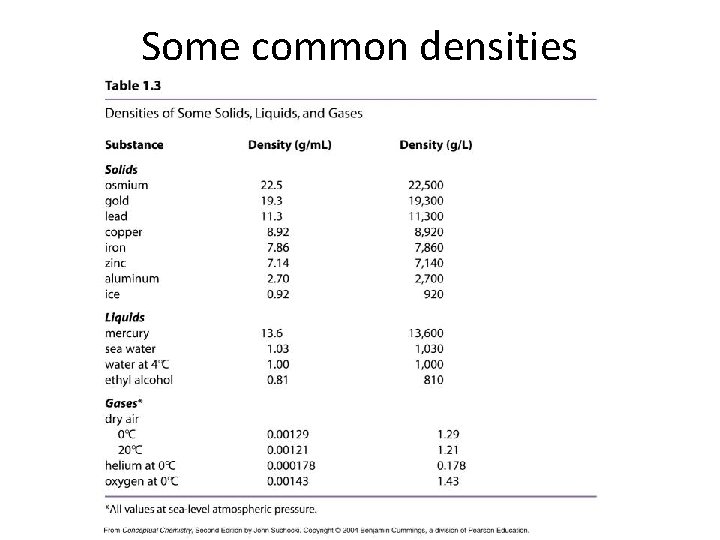
Some common densities

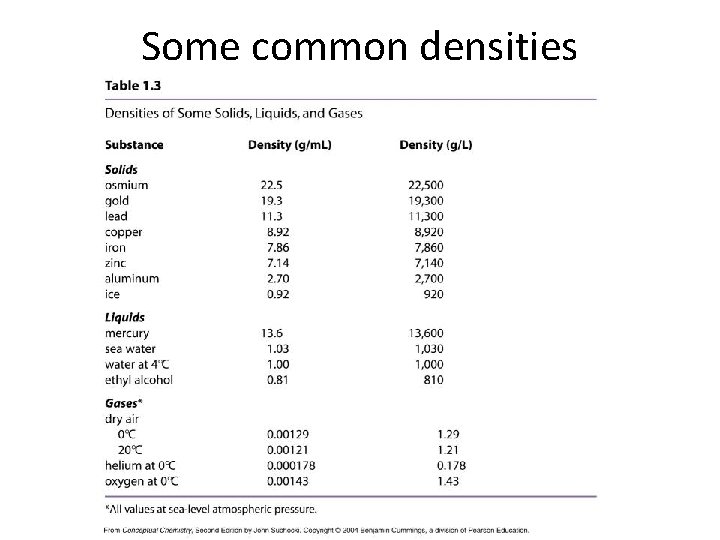
Density and Buoyancy • Water has a density of 1. 0 g/m. L – Any substance with a density greater than 1. 0 g/m. L will sink in water (all metals, for example) – Any substance with a density less than 1. 0 g/m. L will float in water (ice, alcohol, all gases) • Mercury has a density of 13. 6 g/m. L – Any substance with a density greater than 13. 6 g/m. L will sink in mercury (don’t try this with gold!) – Any substance with a density less than 13. 6 g/m. L will float in mercury, including lead!

Density Formulas • Density = mass ÷ volume D= VD= m V V m V 1 1 VD= m D D D

Units of Density g m Density has units of D= m. L V

Sample problem • An unknown liquid filled up to the 50. 1 m. L mark on a graduated cylinder has a mass of 40. 5 g. What is the density of the substance? What is the unknown liquid? • D = m/V • D = 40. 5 g / 50. 1 m. L • D = 0. 80838 g / m. L • D = 0. 808 g/m. L (only 3 significant digits allowed!)

Some common densities

More densities No density: vacuum (0 g/m. L) Water: 1. 0 g/m. L Rocks: 2 to 7 g/m. L Copper: 8. 92 g/m. L Highest density element: Osmium (22. 5 g/m. L) Neutron degenerate matter: 1 x 1015 g/m. L (only found in neutron stars) • Black hole: near-infinite density (5 solar masses compressed to a point in space) • • •


Group work 1. A 20. 0 g sample of lead causes the water level in a graduated cylinder to rise from 50. 0 m. L to 51. 8 m. L. Calculate the density of lead. 2. A platinum ring has a mass of 40. 0 g and a density of 21. 1 g/m. L. If placed into a graduated cylinder filled to the 42. 0 m. L mark with water, to what level will the water rise? 3. A sample of aluminum has a volume of 2. 0 L and a density of 2. 7 g/m. L. Calculate the mass of the sample. 4. An unknown material has a volume of 51, 000 m. L and a mass of 50, 000 g. What is its density? Will it float in water? 5. A stack of 15 nickels has a volume of 4. 0 m. L and a density of 5. 2 g/m. L. What is the mass of each nickel?