Density Review Background Info Phase Diagrams Phase Diagrams

Density Review
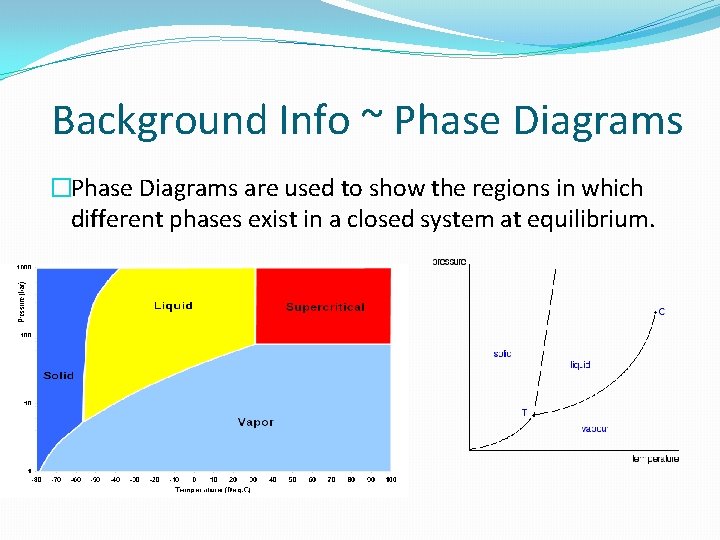
Background Info ~ Phase Diagrams �Phase Diagrams are used to show the regions in which different phases exist in a closed system at equilibrium.

GAS-LIQUID-SOLID �GASES: �Gases are made up of large numbers of very small particles (atoms and molecules). �Most of the volume in a gas is empty space �Constantly in motion, and move in straight lines at high speeds until they bump into something, when they ricochet off in another direction �The greater the temperature the faster the gas particles move. �Temperature cools: �As the particles cool and slow down, they condense into liquids. �The attractive forces in liquids are even stronger in solids. The attractive forces affect the motion of the particles.

PLASMA, BOSE-EINSTEIN CONDENSATES �Phases of matter include: solid, liquid, gas, plasma, and Bose. Einstein condensates. �Plasmas � are much like gases but they are composed of electrically charged particles like ions and electrons. � They’re the most common phase of matter in the visible universe. � Plasmas are found in stars, solar wind, the ionosphere, auroras, and lightening. � Plasmas are used in arc-welders and fluorescent tubes. The flame that you would see in a fire is a plasma. �Bose-Einstein condensates � are atoms at such low temperatures that they slow down and lose their individual identity. They form a cloud that behaves like a single object. �Phases are determined by temperature and pressure!!!!!!
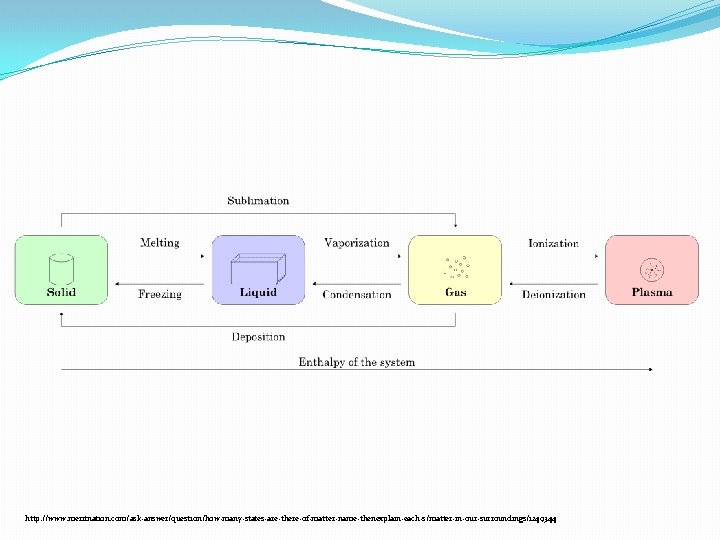
http: //www. meritnation. com/ask-answer/question/how-many-states-are-there-of-matter-name-thenexplain-each-s/matter-in-our-surroundings/1249344

Background Info � In the investigation we observed TWO chemical reactions: an antacid tablet in water, and a candle burning. � New molecules are made from existing atoms. � New materials have different properties than the original materials � � In each reaction, some material appeared to be destroyed and other matter will be created. What’s wrong with this idea? � Mass is not lost or gained, but merely rearranged at the atomic level! – the greatest indicator of chemical change. � For instance, shiny aluminum exposed to oxygen turns dull – it gets coated with aluminum oxide. � Liquids USUALLY have lower density (about 10%) than the solid forms but not always (think of water and ice…)

Physical Change �A change where the composition of the substance remains the same, even if the appearance does not. �No new substance is formed � Examples: changes of state (solid – liquid – gas) �Physical property changes could include a change in: texture, shape, size, color, odor, volume, mass, weight, and density. �A physical change is reversible

Chemical Change • Substances are changed into new substances with their own unique properties. • A new substance is formed • Examples: charcoal burns, iron rusts • Chemical change is not reversible

Physical Change �A change where the composition of the substance remains the same, even if the appearance does not. �Examples: changes of state (solid – liquid – gas) �What’s happening at the atomic level?

Physical and Chemical Change Physical Chemical �Take ice, and melt it. You can re-freeze it and get back ice. � If you pass an electric current through the water, and you evolve hydrogen and oxygen. And unless you collect it, and ignite it, you cannot get back water, so electrolysis of water is a chemical change. �Take a stick. Whittle it into tiny pieces. It is, in theory, possible to reassemble it and get your stick back, so that is a physical change. � But burn the wood, and you cannot get back your stick, so burning wood is a chemical change.

Element �A substance made from only one type of atom. �An element cannot be broken down (chemically) into a more simple substance. �Examples: Carbon is made entirely from Carbon atoms. �Sodium is made entirely from Sodium atoms. �But water is H 2 O… it has TWO elements!

Molecule �The simplest unit of a compound that retains all of the substance’s chemical properties.

Molecule �A molecule is a neutral group of atoms that are held together by covalent bonds

Diatomic Molecule �Molecules made up of two atoms, which can be the same element. �N 2, F 2, Cl 2, O 2.

Compound �A substance that is made of two or more elements that are joined by chemical bonds. �The compound’s properties differ from those of the elements that compose it.

Chemical Formulas How about CO 2? �A combination of symbols and numbers that show which elements, and the number of atoms of each element, that make up a molecule of a compound. The numbers here are called subscripts.

Balanced Equations �The number of atoms of each type must be equal to each other on either side of the arrow. WHY? ? �You cannot change the chemical formulas. �We put numbers (called coefficients) in front of the reactants or products to make it balance.

Homogenous Mixtures �Means “having the same composition and properties throughout. ” �Also called a solution, in which two or more substances are uniformly dispersed. �Can be solid, liquid, or gas. Smog build-up during the day

Heterogeneous Mixtures �Where two or more substances are not uniformly distributed. Granite, mostly a mixture of orthoclase and quartz.

Vocabulary Words �Density �Mass �Volume �Intensive Property �Extensive Property �Chemical Reaction �Physical Reaction �Sublimation �Evaporation �Atom a) Compactness; closely set or crowded condition measured in g/ml b) Smallest unit of an element that maintains the properties of that element c) Change of state from a liquid to a gas d) Substances are changed into new substances with their own unique properties e) Depends on the amount of matter present. (EX: Mass) f) A change where the composition of the substance remains the same, even if the appearance does not. Examples: changes of state (solid – liquid –gas) g) Does not depend on the amount of matter present. (EX Density) h) A measure of how much matter is in an object and measured in g i) A measure of how much space an object takes up and measured in m. L j) Change of state from a solid to a gas

Vocabulary Words �Molecule �Combustion �Heterogeneous �Homogeneous �Macroscopic �Pure Substance �Microscopic �Mixture �Solubility �Element a) b) c) d) e) f) g) h) i) j) Substance that can dissolve in a particular solvent A combination of two or more substances that are not chemically combined. Can be physically separated into its individual parts. Visible with the naked eye Substances that have a fixed composition and has exactly the same characteristic properties and composition A neutral group of atoms that are held together by covalent bonds The process of burning Where two or more substances are not uniformly distributed. Not visible without assistance A pure substance made from only one type of atom Means “having the same composition and properties throughout. ”Can be solid, liquid, or gas.

Do these equations follow the law of conservation? Na 1 Na 2 H 1 H 2 C 1 C 2 O 3 O 5 NO this equation does not follow the law of conservation

Do these equations follow the law of conservation? Zn 1 S 1 YES this equation does follow the law of conservation
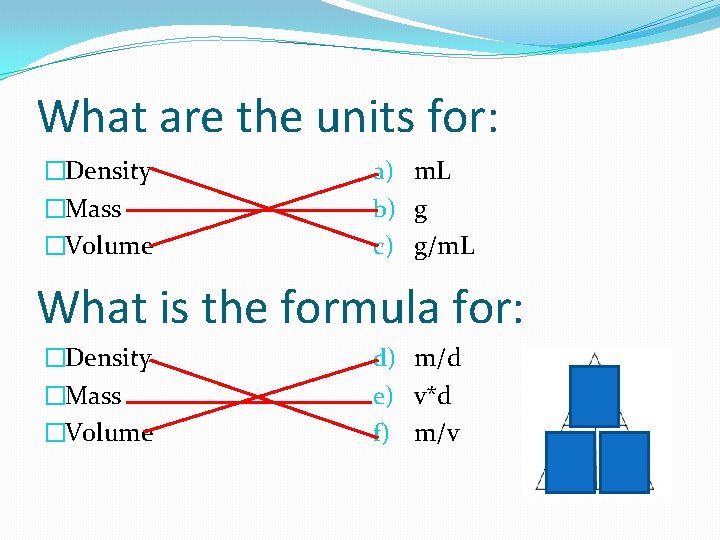
What are the units for: �Density �Mass �Volume a) m. L b) g c) g/m. L What is the formula for: �Density �Mass �Volume d) m/d e) v*d f) m/v
- Slides: 24