Density Key vocabulary Density mass volume What is

Density Key vocabulary: Density, mass, volume

What is density? l Density is a measure of how compact the particles in a substance are. l Examples: A softball is less dense than a bocce ball. A balloon filled with water is more dense than a balloon filled with air.
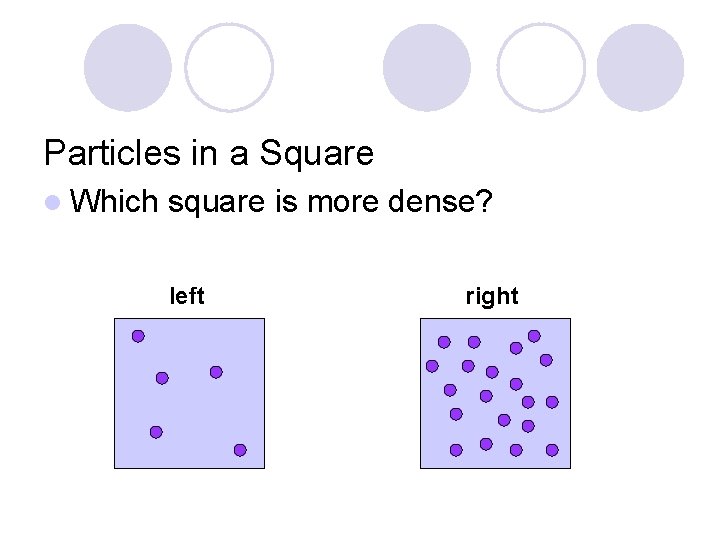
Particles in a Square l Which square is more dense? left right
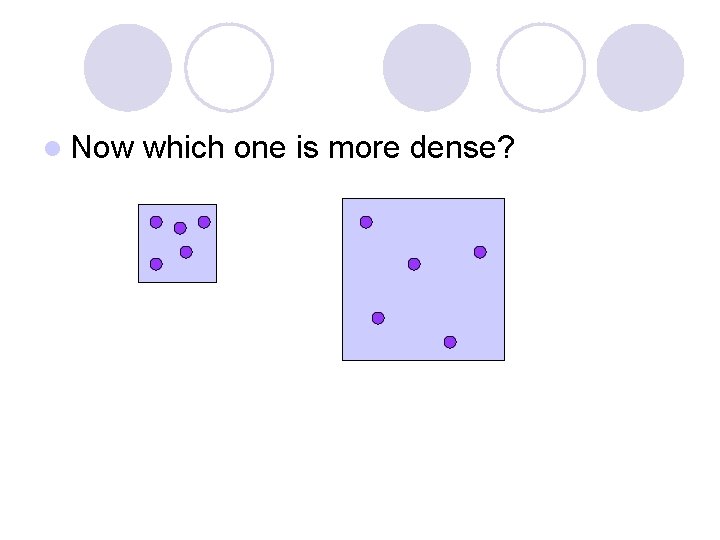
l Now which one is more dense?

Density depends on 2 things: l Mass - how much Matter is in an object - measured in grams with symbol g or kilograms with symbol kg l Volume - how much space the object takes up - measured in milliliters (m. L) or Liters (L) or cubic centimeters cm 3 or cc

Which is more dense? Clothes spread out all over your bedroom OR l The same clothes packed in your suitcase? l Why? l All your books and notebooks jammed in your back pack OR l One book by itself in your back pack l Why? l

Density can be calculated! l Density = D = mass/Volume l Formulas are written with symbols l So, the formula for density is: D = m/V l Units for density: kg/L g/cm 3 g/m. L l Why are these the units for density? Any mass unit over any volume unit can be a density unit.

Let’s try a density problem together l Frank has a paper clip. It has a mass of 9 g and a volume of 3 cm 3. What is its density? l Frank also has an eraser. It has a mass of 3 g, and a volume of 1 cm 3. What is its density?

Work on these problems with your neighbor. l Jack has a rock. The rock has a mass of 6 g and a volume of 3 cm 3. What is the density of the rock? l Jill has a pen. The pen has a mass of 8 g and a volume of 2 cm 3. What is the density of the pen?

Now, try these on your own. l Alicia has a watch. It has a mass of 4 g and a volume of 2 cm 3. What is the density of the watch? l Mia has a wallet. It has a mass of 15 g and a volume of 5 cm 3. What is the density of the wallet?

Find the density of a wood block: 1. Measure its mass _____ g l 2. Determine its volume. length x width x height = volume ___cm x ___cm = ____ l 3. Calculate its density. formula: l NPI: Answer with units:

Liquid Density l Review! l How What is the formula for Density? do we derive the density of a liquid, without getting the balance all wet?

How to derive the density of a liquid? l 1. Measure the mass of an EMPTY graduated cylinder and record. l 2. Place some liquid in the graduated cylinder (carefully without making a mess!) l 3. Mass the graduated cylinder WITH the liquid and record.

Continued… l 4. Subtract! l 5. Read the graduated cylinder to measure the VOLUME of the liquid: l 6. Calculate the DENSITY using formula.

Let’s find the density of some liquids! l l l l l Pancake syrup Vegetable oil Mineral oil Motor oil Glycerine Dish soap Isopropyl alcohol Deionized water Salt water l l l l l Be sure to put on lab aprons before beginning your experiment!
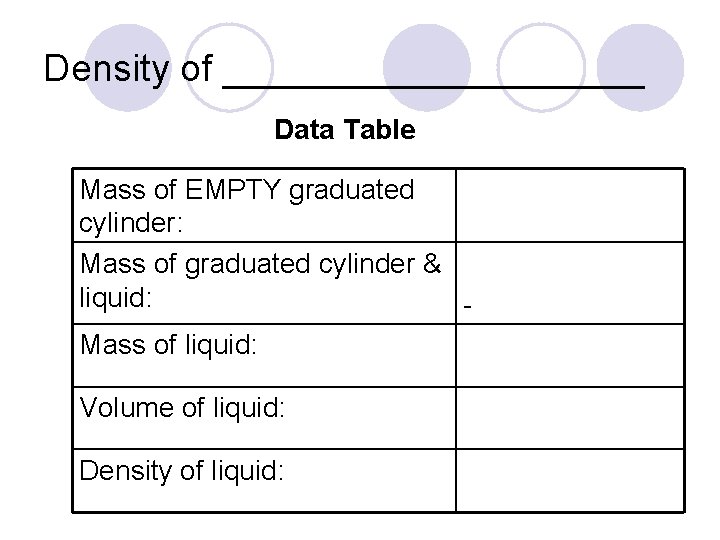
Density of __________ Data Table Mass of EMPTY graduated cylinder: Mass of graduated cylinder & liquid: Mass of liquid: Volume of liquid: Density of liquid:

Liquid Layers l If you pour together liquids that don’t mix and have different densities, they will form liquid layers. l The liquid with the lowest density will be on the top. l The liquid with the highest density will be on the bottom.

Liquid Layers l Check out this picture. Which layer has the highest density? l Which layer has the lowest density? Do solids have greater density than liquids? l Predict: How will various solids settle out in the liquid layers? l
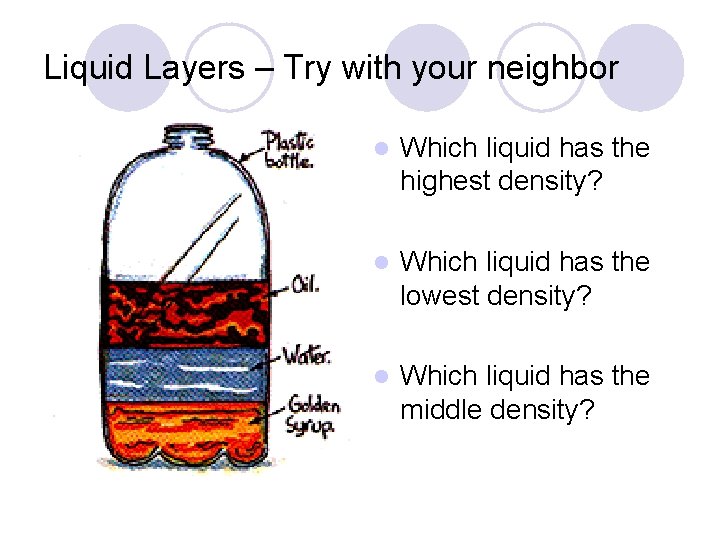
Liquid Layers – Try with your neighbor l Which liquid has the highest density? l Which liquid has the lowest density? l Which liquid has the middle density?

Liquid Layers – Try on your own! l Imagine that the liquids on the right have the following densities: ¡ ¡ ¡ l 15 g/cm 3 3 g/cm 3 7 g/cm 3 10 g/cm 3 9 g/cm 3 12 g/cm 3 Match the colors to the correct densities.

Review l What is the formula for density? l What happens if you pour together liquids that have different densities? l Do solids have greater density than liquids? l Will the substance on the top have the highest or lowest density? l Will the substance on the bottom have the highest or lowest density?

Super Scientist Question of the Day l Jake has an irregularly shaped rock, a ruler, graduated cylinder, water and a balance. l How can Jake find the density of the rock with the tools he has?
- Slides: 22