Density Functional Theory of Iron Carbide and Steel

Density Functional Theory of Iron Carbide and Steel Surface Erosion Chemistry Wun C. Chiou, Jr Emily A. Carter University of California, Los Angeles Dept. of Chemistry & Biochemistry Thanks: Funding: U. S. Army Research Office Resources: Maui High Performance Computing Facility, Army Research Laboratory MSRC

- Steel Erosion Issues Problem: In a harsh operating environment, an exposed steel surface can erode, leading to decreased performance, safety concerns, and repair/replacement costs. Environment: T ~ 1700 K High Pressures Chemicals (H 2, O 2, CO 2, NO 2, and etc) Mechanical Forces Impact: Petrochemical Industry Processes Steam Reformers Gun Tubes Industrial Furnaces CO 2 -cooled Nuclear Reactors Coal Gasification Processes

- Carburization Definition: A high-temperature corrosion phenomenon caused by carbon ingress from the environment into metal components, leading to internal carbide precipitation and changes to the mechanical properties of the materials. Grabke, H. J. Carburization: A High Temperature Corrosion Phenomenon. MTI, 1998. Mechanism: C/CO transport to the steel surface C diffuses into the surface Solid state reaction with Fe in steel Fem + Cn Þ Fem. Cn Consequences: Phase-change stresses Altered ductile properties, grain cohesion MP-lowering: TM~1800 K Þ TM<1500 K Metal Dusting: disintegration of the steel in a dust of metal particles and C
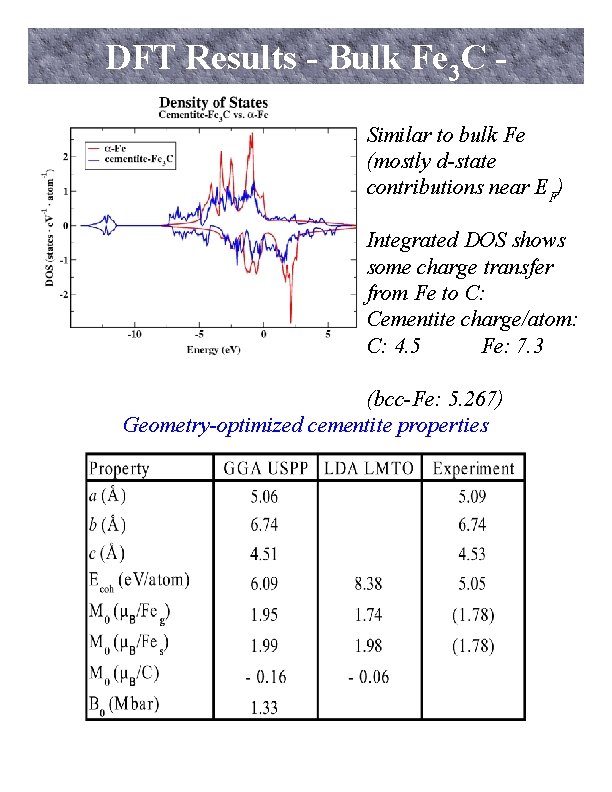
DFT Results - Bulk Fe 3 C Similar to bulk Fe (mostly d-state contributions near EF) Integrated DOS shows some charge transfer from Fe to C: Cementite charge/atom: C: 4. 5 Fe: 7. 3 (bcc-Fe: 5. 267) Geometry-optimized cementite properties
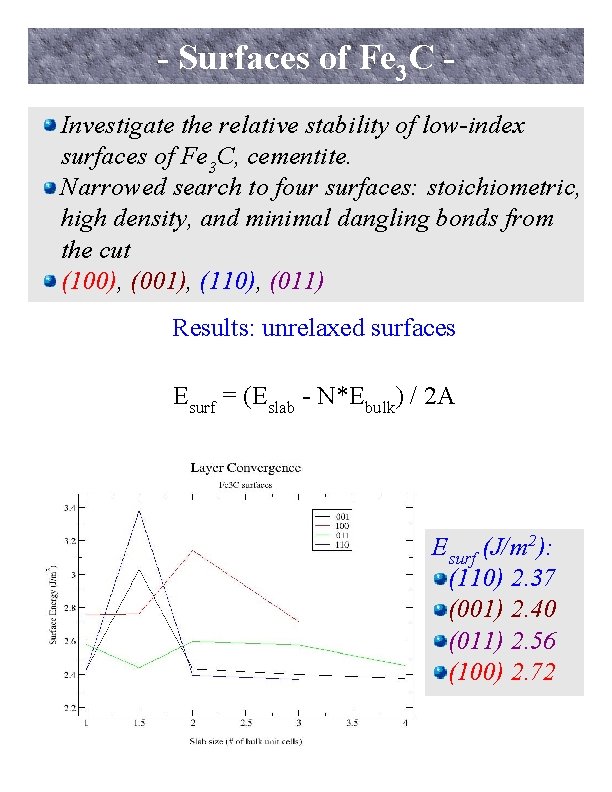
- Surfaces of Fe 3 C Investigate the relative stability of low-index surfaces of Fe 3 C, cementite. Narrowed search to four surfaces: stoichiometric, high density, and minimal dangling bonds from the cut (100), (001), (110), (011) Results: unrelaxed surfaces Esurf = (Eslab - N*Ebulk) / 2 A Esurf (J/m 2): (110) 2. 37 (001) 2. 40 (011) 2. 56 (100) 2. 72
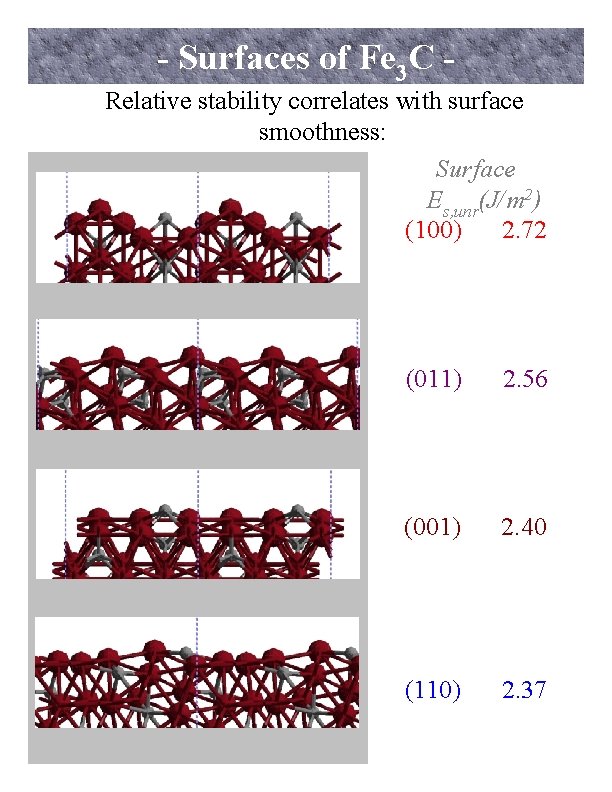
- Surfaces of Fe 3 C - Relative stability correlates with surface smoothness: Surface Es, unr(J/m 2) (100) 2. 72 (011) 2. 56 (001) 2. 40 (110) 2. 37
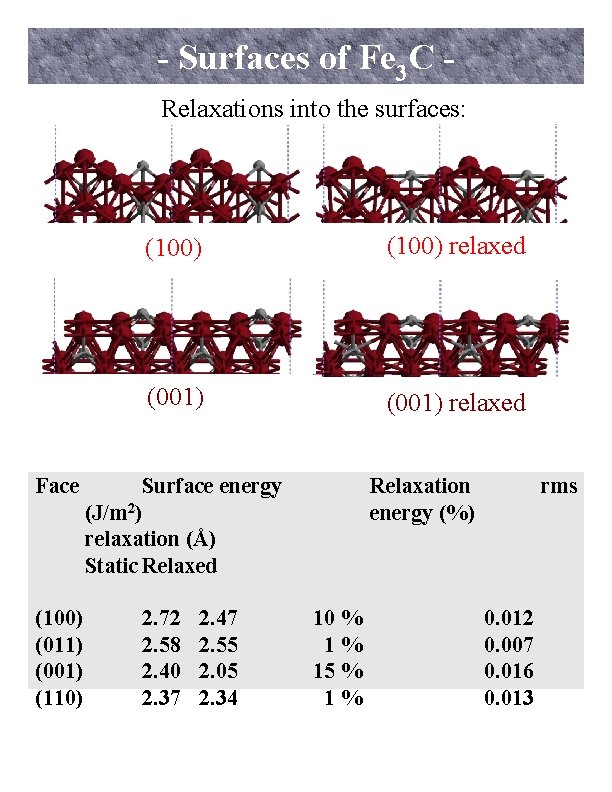
- Surfaces of Fe 3 C Relaxations into the surfaces: Face (100) (011) (001) (110) (100) relaxed (001) relaxed Surface energy (J/m 2) relaxation (Å) Static Relaxed 2. 72 2. 58 2. 40 2. 37 2. 47 2. 55 2. 05 2. 34 Relaxation energy (%) 10 % 1% 15 % 1% rms 0. 012 0. 007 0. 016 0. 013

- Conclusion Iron carbide and carburization are important aspects of steel erosion Bulk Ecoh improves on earlier predictions Cementite DOS is similar to pure Fe, but with some charge transfer to C Surface stability: (001) > (110) > (011) > (100) Fe 3 C/Fe Interface calculations Carbide formation
- Slides: 8