DENSITY DENSITY By definition How heavy something is

DENSITY

DENSITY: • By definition: How heavy something is compared to its size • In math: d=m/V • m=mass • V=volume • Example: The density of gold is 19. 3 g/m. L this means that every 1 m. L of gold weighs 19. 3 grams.

WHAT IS VOLUME? • Volume: the amount of space an object occupies. • Formula: • Cube = l·w·h • Sphere = 4/3πr 3 • Cylinder = πr 2 * h • How could you determine the volume of an irregular object? • Water displacement

CALCULATING VOLUME • Try this one… • Q: What is the volume of the cube? • V = 288 in 3 • Make sure to correctly label your answer!

THINGS YOU SHOULD KNOW: • Density of water: 1 g/m. L • An object will float in water if the density is less than 1 g/m. L (ie d=. 97 g/m. L) • An object will sink in water if the density is greater than 1 g/m. L (ie d=1. 1 g/m. L)
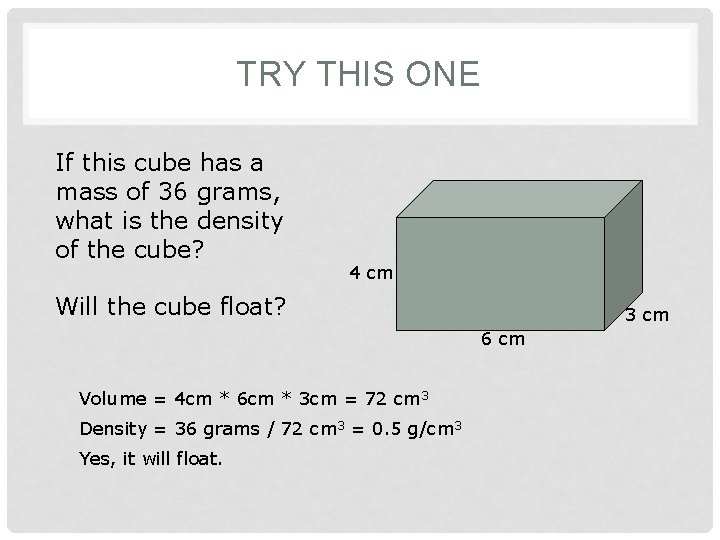
TRY THIS ONE If this cube has a mass of 36 grams, what is the density of the cube? 4 cm Will the cube float? 3 cm 6 cm Volume = 4 cm * 6 cm * 3 cm = 72 cm 3 Density = 36 grams / 72 cm 3 = 0. 5 g/cm 3 Yes, it will float.

A baseball has a mass of 146 grams. If it has a radius of 3. 5 cm, what is density of a baseball? Will a baseball float? First calculate the volume. V = 4/3π(3. 5 cm)3 = 180 cm 3 Density = 0. 81 g/cm 3 IT FLOATS

WHAT ABOUT THE DENSITY OF A CYLINDER? • If the cylinder shown has a mass of 18 grams, what is its density? 8 cm • Volume = 100. 5 cm 3 • Density = 0. 18 g/cm 3 2 cm

WHAT ABOUT WITH AN IRREGULAR OBJECT? • A rock has a mass of 260 grams. In order to determine its volume it is dropped into a graduated cylinder. The water level changes from 40 m. L to 120 m. L. What is the density of the rock? • Density = 260 g / 80 m. L = 3. 25 g/m. L • Will it float?

Q: What is the density of the Earth in g/m. L? Mass: 5. 98× 1024 kg Volume: 1. 08× 1024 L 5. 98× 1024 kg 1000 g = 5. 98× 1027 g 1 kg 1. 08× 1024 L 1000 m. L =1. 08× 1027 m. L 1 L A: 5. 98× 1027 g/ 1. 08× 1027 m. L ≈ 5. 5 g/m. L

TRY THIS ONE… • Q: A block of aluminum occupies a volume of 15. 0 m. L and weighs 40. 5 g. What is its density? d= 40. 5 g/15 m. L • A: d= 2. 7 g/m. L

MORE PRACTICE • Q: What is the weight of the ethyl alcohol that exactly fills a 200. 0 m. L container? The density of ethyl alcohol is 0. 789 g/m. L. Use density as a conversion factor in dimensional analysis 200. 0 m. L 0. 789 g 1 m. L • A: g=158 grams

USE DIMENSIONAL ANALYSIS A chunk of aluminum displaces 4 Liters of water. If the density of aluminum is 2. 70 grams/m. L, what is the mass in kilograms of the sample? 4 L 1000 m. L 2. 70 g 1 L 1 m. L 1 kg 1000 g = 10. 8 kg

ONE MORE • Q: A piece of metal has a density of 12. 6 kg/L. If you have 25 m. L of this metal, how many grams is it? 25 m. L 1 L 1000 m. L u A: 315 grams 12. 6 kg 1000 g 1 L 1 kg

THE END YAY FOR DENSITY…
- Slides: 15