Dendritic cellbased Cancer Immunotherapy November 2 2015 Shigetaka

Dendritic cell-based Cancer Immunotherapy November 2, 2015 Shigetaka Shimodaira, MD, Ph. D Center for Advanced Cellular Therapy Shinshu University Hospital

Disclosure of Interest Shigetaka Shimodaira I declare no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.

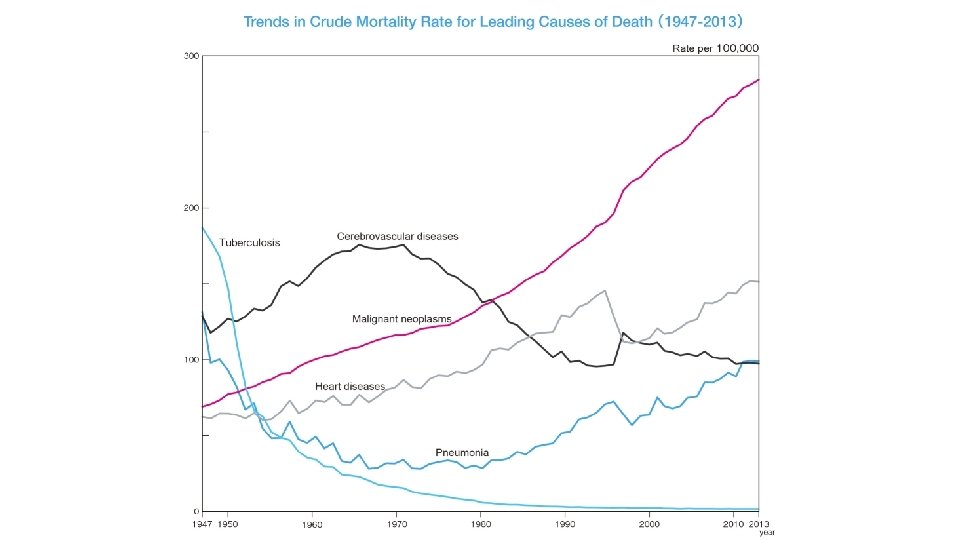
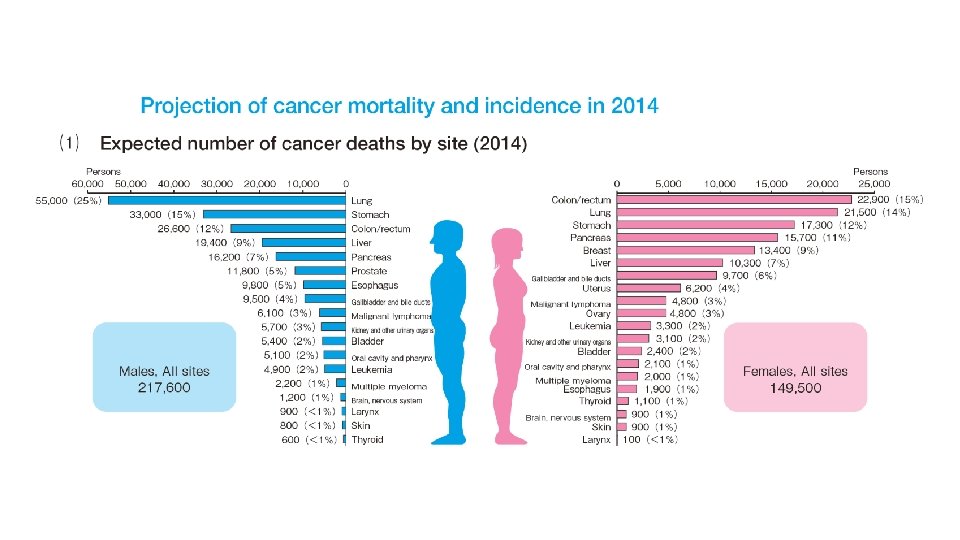
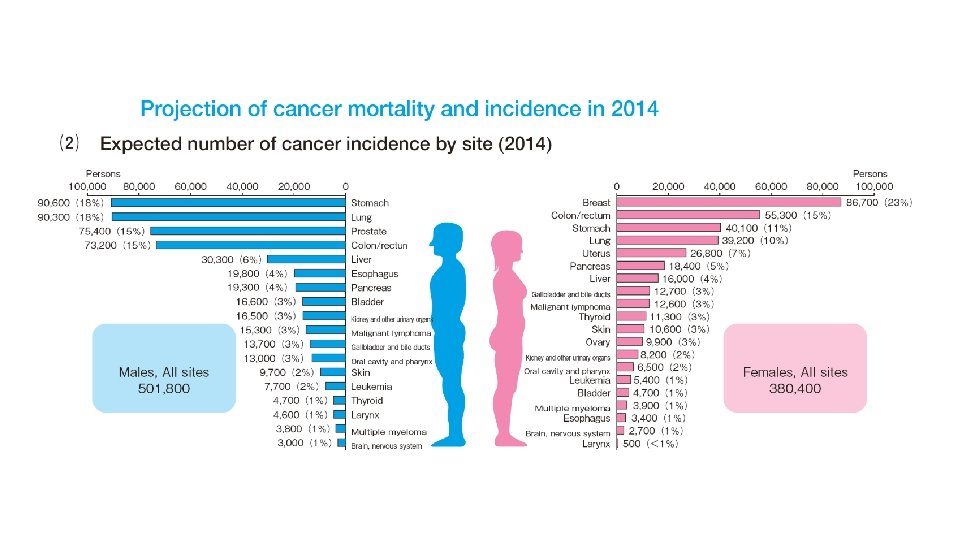
Contents • Introduction • Dendritic cell (DC) and immune cells under cancer environment • Cancer mortality and incidence in Japan • Manufacturing of a DC Vaccine and Wilms’ Tumor Gene 1 peptides-pulsed DC (WT 1 -DC) vaccination • Immune monitoring with tetramer assay and enzyme immunosorbent (ELISPOT) assay • DC vaccination technology • Granulocyte colony-stimulating factor (G-CSF) and DC vaccine and vaccination • Allogenic WT 1 -DC vaccination for acute leukemia

Nobel winner's last big experiment: Himself CHICAGO/NEW YORK (Reuters) By Julie Steenhuysen and Michelle Nichols Thu Oct 6, 2011 In the last few years of his life, Dr. Ralph Steinman made himself into an extraordinary human lab experiment, testing a series of unproven therapies including some he helped to create - as he waged a very personal battle with pancreatic cancer. http: //www. reuters. com/article/2011/10/06/us-nobel-medicine-experiment-id. USTRE 7956 CN 20111006
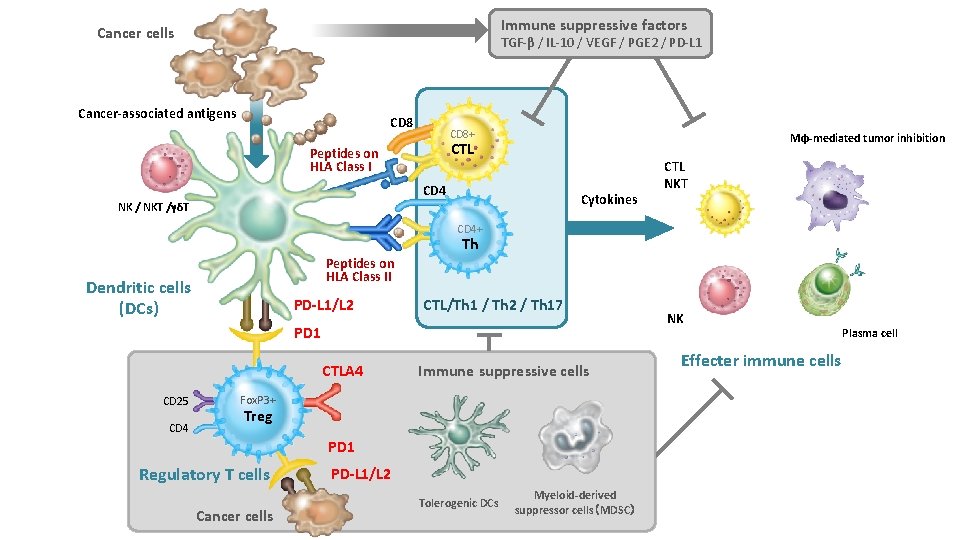
Immune suppressive factors Cancer cells TGF-β / IL-10 / VEGF / PGE 2 / PD-L 1 Cancer-associated antigens CD 8+ Mφ-mediated tumor inhibition CTL Peptides on HLA Class I CD 4 Cytokines NK / NKT /γδT CTL NKT CD 4+ Th Peptides on HLA Class II Dendritic cells (DCs) PD-L 1/L 2 CTL/Th 1 / Th 2 / Th 17 PD 1 CTLA 4 CD 25 CD 4 Immune suppressive cells Fox. P 3+ Treg PD 1 Regulatory T cells Cancer cells PD-L 1/L 2 Tolerogenic DCs Myeloid-derived suppressor cells(MDSC) NK Effecter immune cells Plasma cell
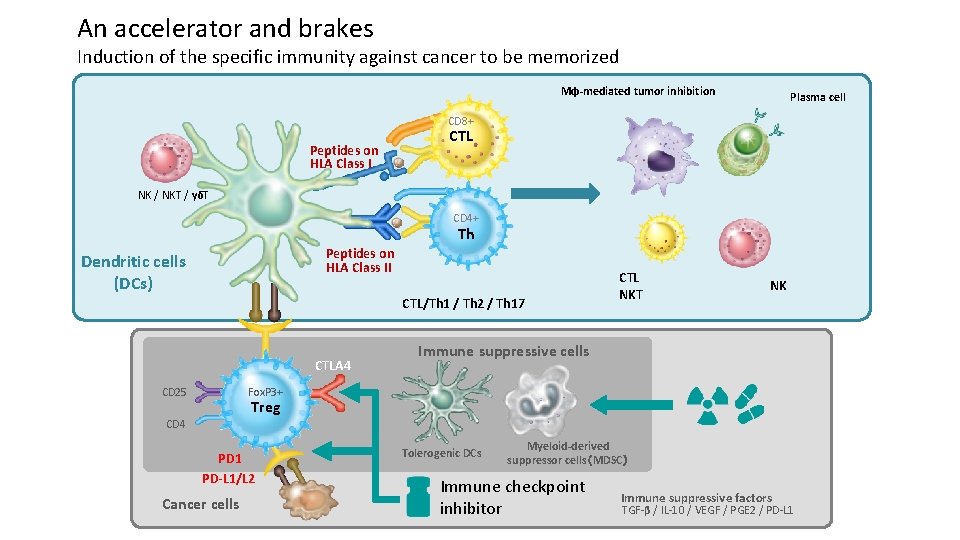
An accelerator and brakes Induction of the specific immunity against cancer to be memorized Mφ-mediated tumor inhibition Plasma cell CD 8+ Peptides on HLA Class I CTL NK / NKT / γδT CD 4+ Th Peptides on HLA Class II Dendritic cells (DCs) CTL/Th 1 / Th 2 / Th 17 CTLA 4 CD 25 CTL NKT NK Immune suppressive cells Fox. P 3+ Treg CD 4 PD 1 PD-L 1/L 2 Cancer cells Tolerogenic DCs Myeloid-derived suppressor cells(MDSC) Immune checkpoint inhibitor Immune suppressive factors TGF-β / IL-10 / VEGF / PGE 2 / PD-L 1


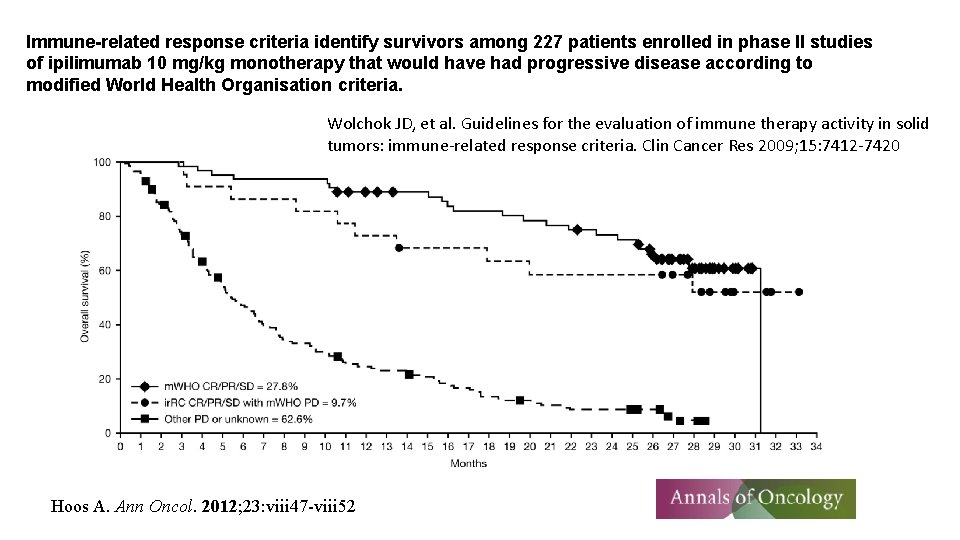
Immune-related response criteria identify survivors among 227 patients enrolled in phase II studies of ipilimumab 10 mg/kg monotherapy that would have had progressive disease according to modified World Health Organisation criteria. Wolchok JD, et al. Guidelines for the evaluation of immune therapy activity in solid tumors: immune-related response criteria. Clin Cancer Res 2009; 15: 7412 -7420 Hoos A. Ann Oncol. 2012; 23: viii 47 -viii 52
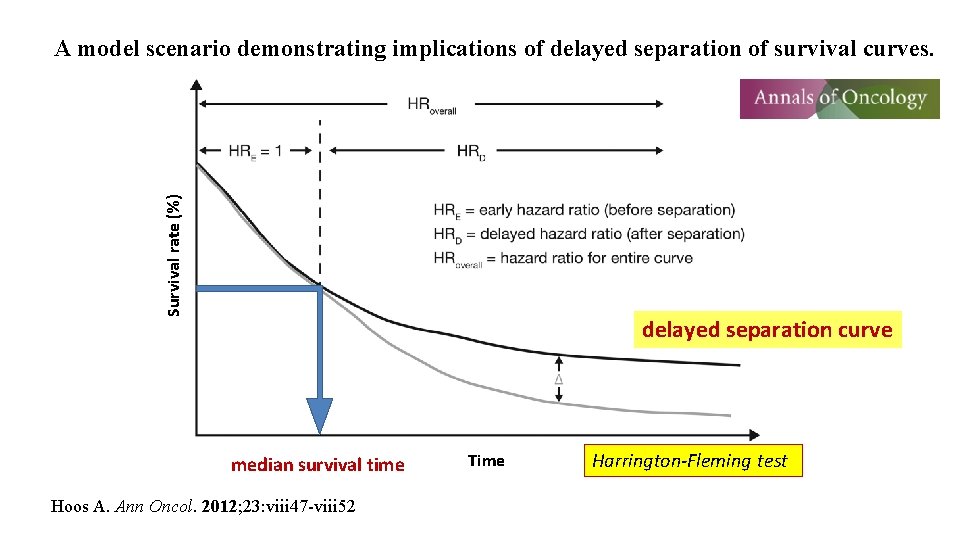
Survival rate (%) A model scenario demonstrating implications of delayed separation of survival curves. delayed separation curve median survival time Hoos A. Ann Oncol. 2012; 23: viii 47 -viii 52 Time Harrington-Fleming test
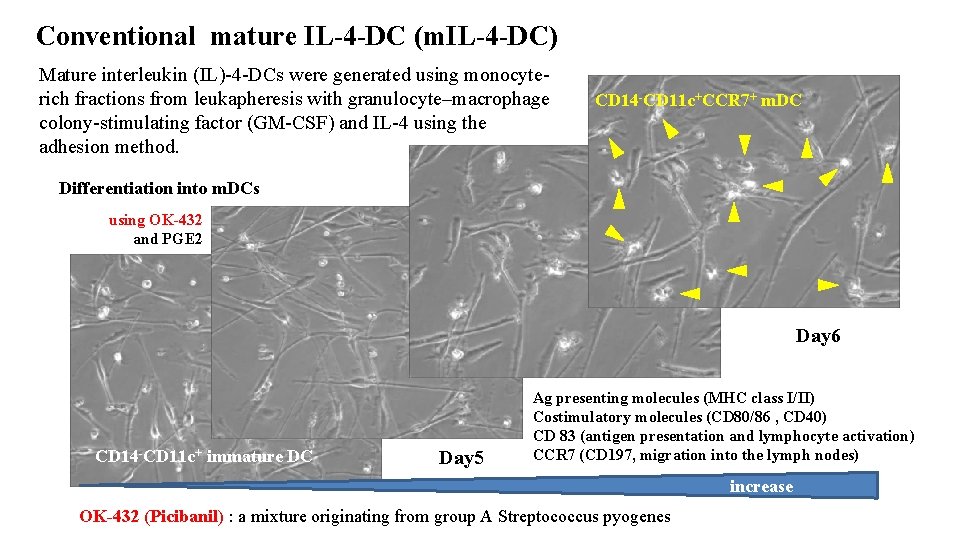
Conventional mature IL-4 -DC (m. IL-4 -DC) Mature interleukin (IL)-4 -DCs were generated using monocyterich fractions from leukapheresis with granulocyte–macrophage colony-stimulating factor (GM-CSF) and IL-4 using the adhesion method. CD 14 -CD 11 c+CCR 7+ m. DC Differentiation into m. DCs using OK-432 and PGE 2 Day 6 CD 14 -CD 11 c+ immature DC Day 5 Ag presenting molecules (MHC class I/II) Costimulatory molecules (CD 80/86 , CD 40) CD 83 (antigen presentation and lymphocyte activation) CCR 7 (CD 197, migration into the lymph nodes) increase OK-432 (Picibanil) : a mixture originating from group A Streptococcus pyogenes
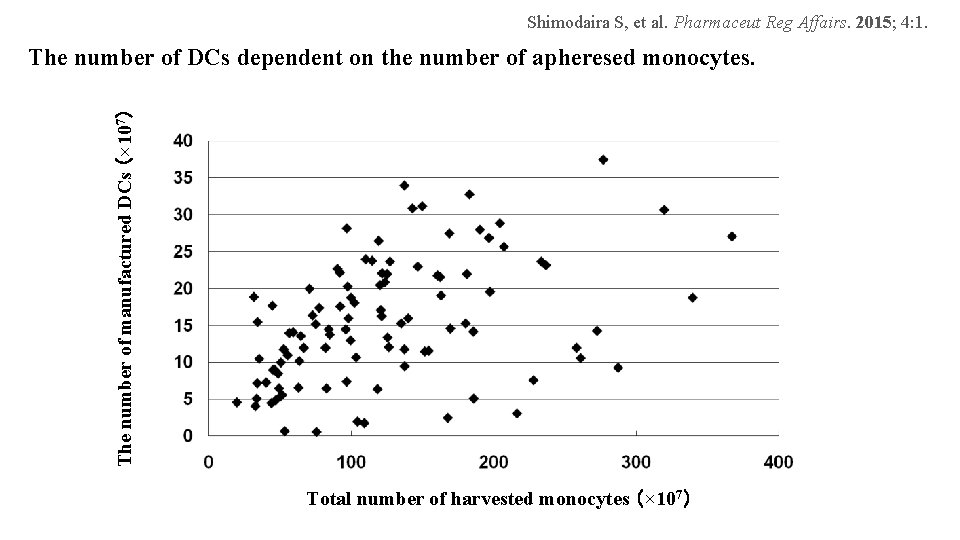
Shimodaira S, et al. Pharmaceut Reg Affairs. 2015; 4: 1. The number of manufactured DCs (× 107) The number of DCs dependent on the number of apheresed monocytes. Total number of harvested monocytes (× 107)
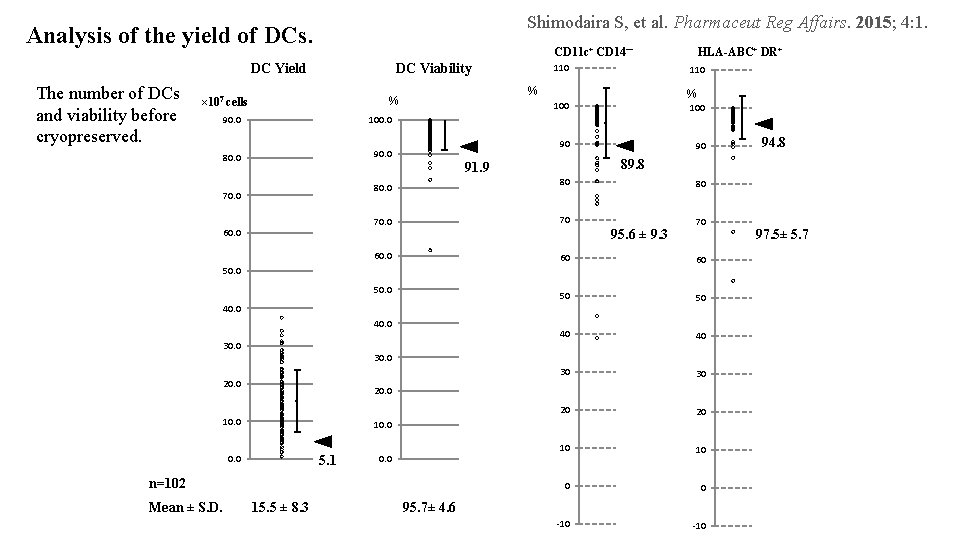
Shimodaira S, et al. Pharmaceut Reg Affairs. 2015; 4: 1. Analysis of the yield of DCs. CD 11 c+ CD 14- DC Viability DC Yield The number of DCs and viability before cryopreserved. 90. 0 100. 0 80. 0 90. 0 60. 0 90 80 80 70 60. 0 60 60 50 50 40 40 30 30 20 20 10 10 0 0 -10 95. 6 ± 9. 3 40. 0 70 30. 0 20. 0 10. 0 5. 1 0. 0 n=102 15. 5 ± 8. 3 94. 8 89. 8 91. 9 50. 0 Mean ± S. D. 100 90 50. 0 30. 0 % 100 80. 0 70. 0 110 % % × 107 cells 110 HLA-ABC+ DR+ 95. 7± 4. 6 97. 5± 5. 7
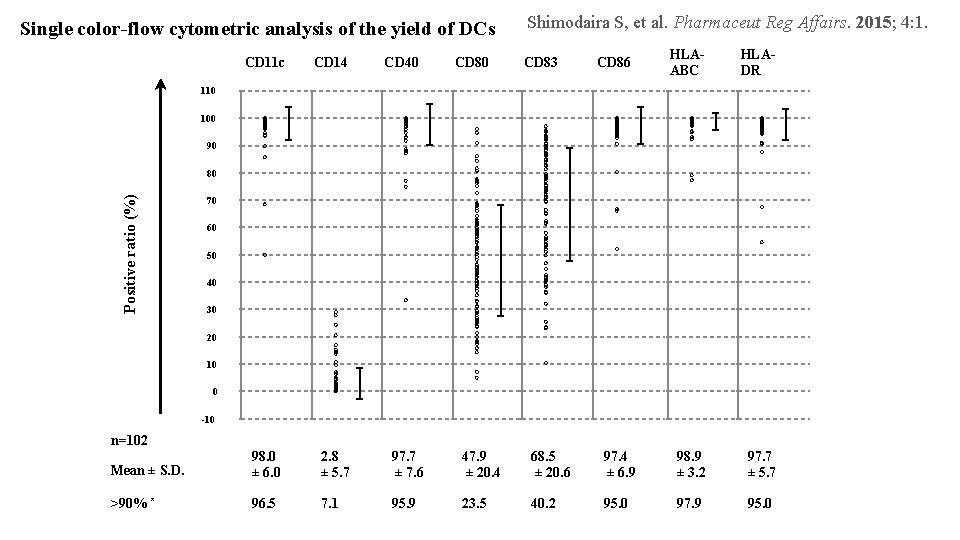
Single color-flow cytometric analysis of the yield of DCs CD 11 c CD 14 CD 40 CD 80 Shimodaira S, et al. Pharmaceut Reg Affairs. 2015; 4: 1. CD 83 CD 86 HLAABC HLADR 110 100 90 Positive ratio (%) 80 70 60 50 40 30 20 10 0 -10 n=102 Mean ± S. D. 98. 0 ± 6. 0 2. 8 ± 5. 7 97. 7 ± 7. 6 47. 9 ± 20. 4 68. 5 ± 20. 6 97. 4 ± 6. 9 98. 9 ± 3. 2 97. 7 ± 5. 7 >90% * 96. 5 7. 1 95. 9 23. 5 40. 2 95. 0 97. 9 95. 0
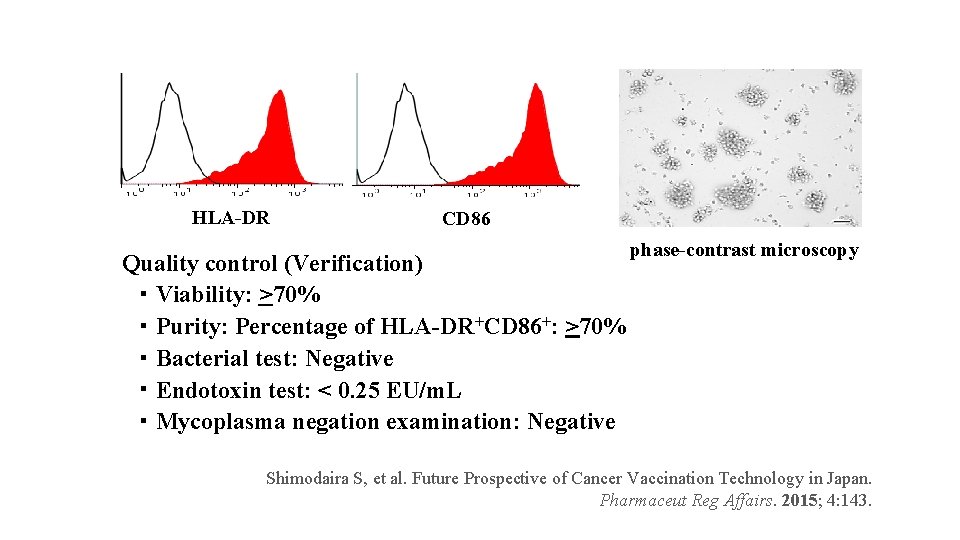
HLA-DR CD 86 phase-contrast microscopy Quality control (Verification) ・ Viability: >70% ・ Purity: Percentage of HLA-DR+CD 86+: >70% ・ Bacterial test: Negative ・ Endotoxin test: < 0. 25 EU/m. L ・ Mycoplasma negation examination: Negative Shimodaira S, et al. Future Prospective of Cancer Vaccination Technology in Japan. Pharmaceut Reg Affairs. 2015; 4: 143.
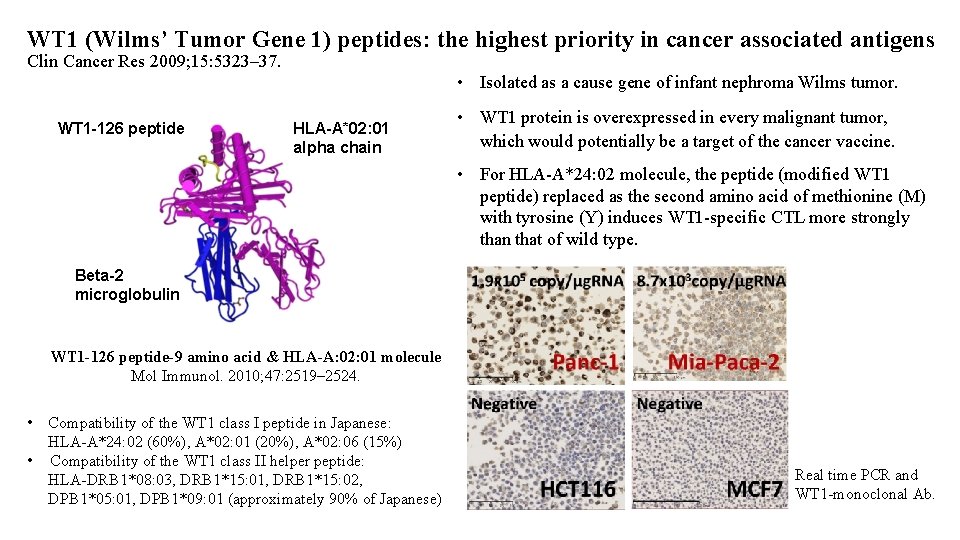
WT 1 (Wilms’ Tumor Gene 1) peptides: the highest priority in cancer associated antigens Clin Cancer Res 2009; 15: 5323– 37. • Isolated as a cause gene of infant nephroma Wilms tumor. WT 1 -126 peptide HLA-A*02: 01 alpha chain • WT 1 protein is overexpressed in every malignant tumor, which would potentially be a target of the cancer vaccine. • For HLA-A*24: 02 molecule, the peptide (modified WT 1 peptide) replaced as the second amino acid of methionine (M) with tyrosine (Y) induces WT 1 -specific CTL more strongly than that of wild type. Beta-2 microglobulin WT 1 -126 peptide-9 amino acid & HLA-A: 02: 01 molecule Mol Immunol. 2010; 47: 2519– 2524. • Compatibility of the WT 1 class I peptide in Japanese: HLA-A*24: 02 (60%), A*02: 01 (20%), A*02: 06 (15%) • Compatibility of the WT 1 class II helper peptide: HLA-DRB 1*08: 03, DRB 1*15: 01, DRB 1*15: 02, DPB 1*05: 01, DPB 1*09: 01 (approximately 90% of Japanese) Real time PCR and WT 1 -monoclonal Ab.
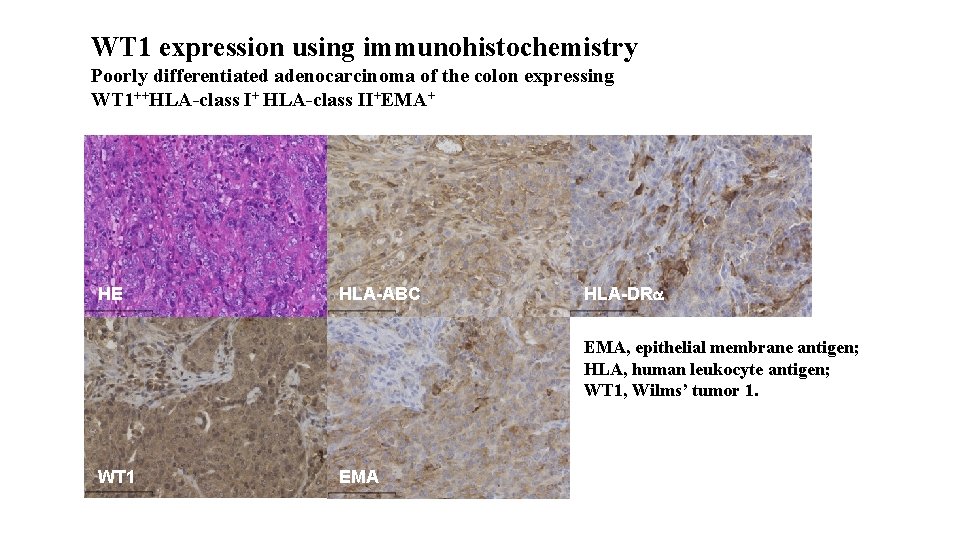
WT 1 expression using immunohistochemistry Poorly differentiated adenocarcinoma of the colon expressing WT 1++HLA-class I+ HLA-class II+EMA+ HE HLA-ABC HLA-DRa EMA, epithelial membrane antigen; HLA, human leukocyte antigen; WT 1, Wilms’ tumor 1. WT 1 EMA
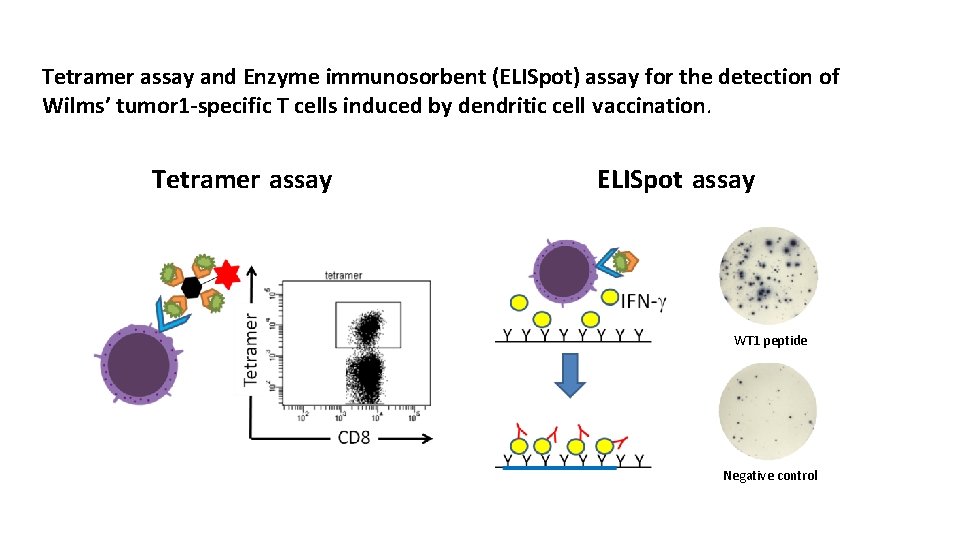
Tetramer assay and Enzyme immunosorbent (ELISpot) assay for the detection of Wilms’ tumor 1 -specific T cells induced by dendritic cell vaccination. Tetramer assay ELISpot assay WT 1 peptide Negative control
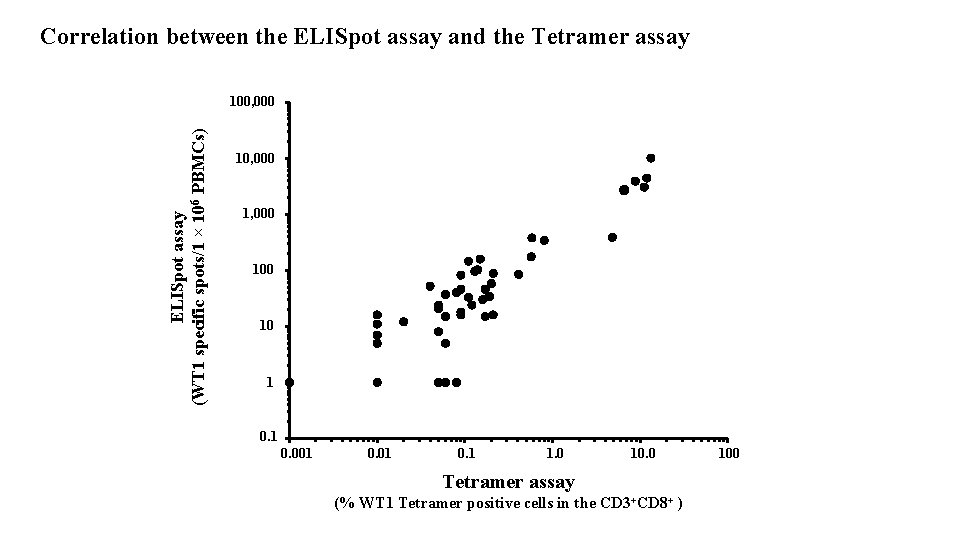
Correlation between the ELISpot assay and the Tetramer assay ELISpot assay (WT 1 specific spots/1 × 106 PBMCs) 100, 000 1, 000 10 1 0. 001 0. 1 1. 0 10. 0 Tetramer assay (% WT 1 Tetramer positive cells in the CD 3+CD 8+ ) 100

WT 1 -DC vaccine and vaccination 1. DC vaccine therapy approved under “Advanced Medical Care in Japan” 2. DC manufacturing protocol: 2. 1. HLA-DNA typing to determine WT 1 compatibility 2. 2. Apheresis: 165 m. L mononuclear cells collected from 4 L of blood (COM-TEC; Fresenius Kabi, Germany) 2. 3. Adhesive monocytes cultured with IL-4 and GM-CSF 2. 4. Mature DCs induced by OK-432 (streptococcal preparation) stored in the gas layer of liquid N 2 until clinical use. 3. DC vaccination: 1– 3 × 107 DC injected into the axillary and inguinal areas with OK-432 (1– 2 KE/dose) at intervals of 2 weeks for 7 sessions (1 course) during individual chemotherapy and radiotherapy.
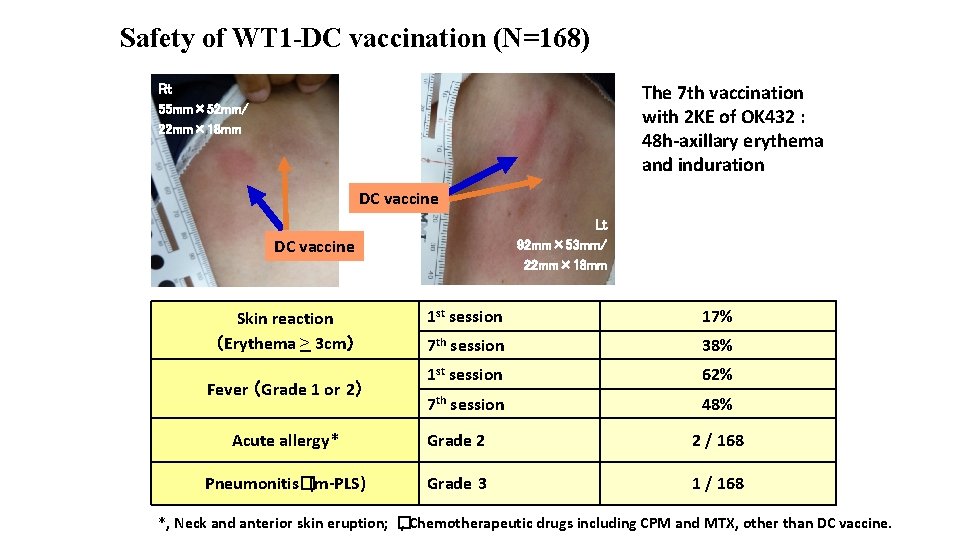
Safety of WT 1 -DC vaccination (N=168) The 7 th vaccination with 2 KE of OK 432 : 48 h-axillary erythema and induration Rt 55 mm× 52 mm/ 22 mm× 18 mm DC vaccine Lt 92 mm× 53 mm/ 22 mm× 18 mm DC vaccine Skin reaction (Erythema > 3 cm) Fever (Grade 1 or 2) 1 st session 17% 7 th session 38% 1 st session 62% 7 th session 48% Acute allergy* Grade 2 2 / 168 Pneumonitis�(m-PLS) Grade 3 1 / 168 *, Neck and anterior skin eruption; � , Chemotherapeutic drugs including CPM and MTX, other than DC vaccine.
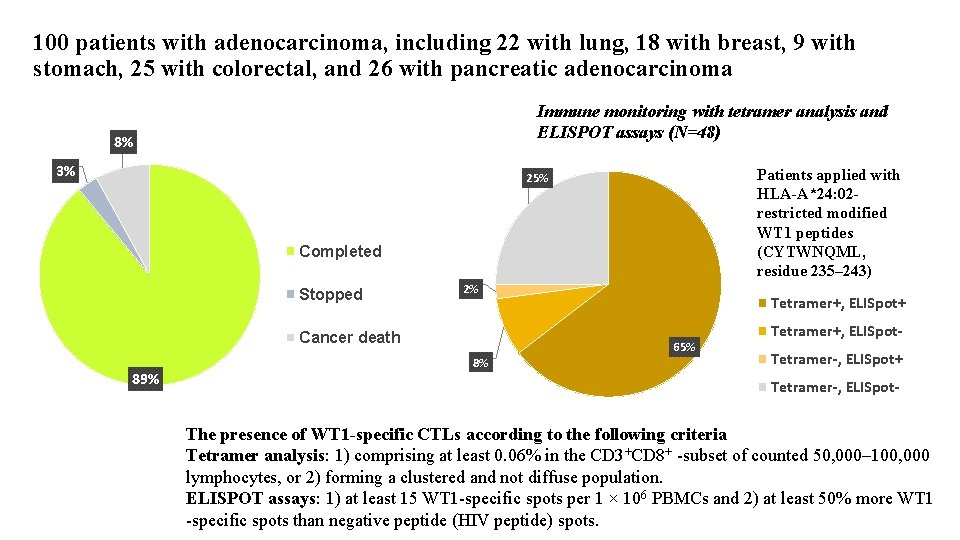
100 patients with adenocarcinoma, including 22 with lung, 18 with breast, 9 with stomach, 25 with colorectal, and 26 with pancreatic adenocarcinoma Immune monitoring with tetramer analysis and ELISPOT assays (N=48) 8% 3% Patients applied with HLA-A*24: 02 restricted modified WT 1 peptides (CYTWNQML, residue 235– 243) 25% Completed Stopped 2% Cancer death 89% 8% Tetramer+, ELISpot+ 65% Tetramer+, ELISpot. Tetramer-, ELISpot+ Tetramer-, ELISpot- The presence of WT 1 -specific CTLs according to the following criteria Tetramer analysis: 1) comprising at least 0. 06% in the CD 3+CD 8+ -subset of counted 50, 000– 100, 000 lymphocytes, or 2) forming a clustered and not diffuse population. ELISPOT assays: 1) at least 15 WT 1 -specific spots per 1 × 106 PBMCs and 2) at least 50% more WT 1 -specific spots than negative peptide (HIV peptide) spots.
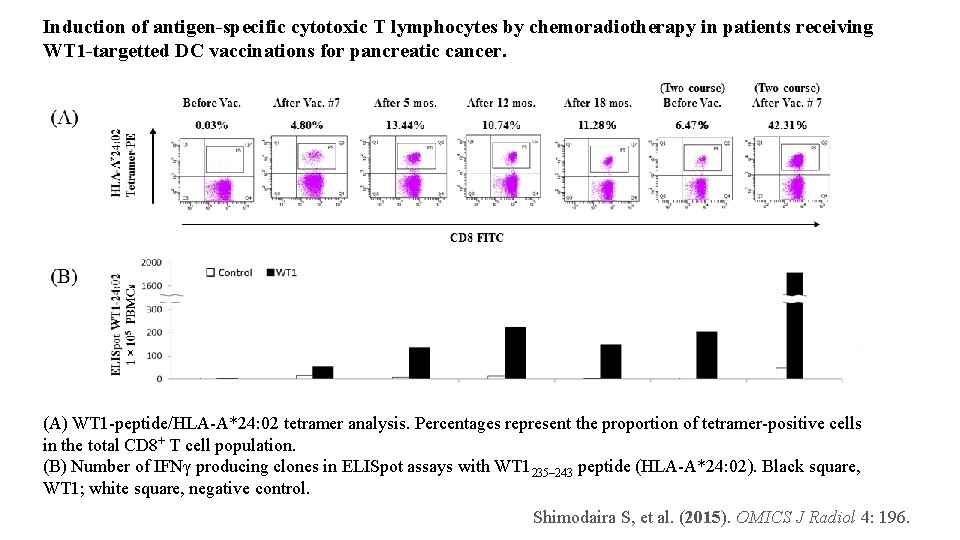
Induction of antigen-specific cytotoxic T lymphocytes by chemoradiotherapy in patients receiving WT 1 -targetted DC vaccinations for pancreatic cancer. (A) WT 1 -peptide/HLA-A*24: 02 tetramer analysis. Percentages represent the proportion of tetramer-positive cells in the total CD 8+ T cell population. (B) Number of IFNγ producing clones in ELISpot assays with WT 1235– 243 peptide (HLA-A*24: 02). Black square, WT 1; white square, negative control. Shimodaira S, et al. (2015). OMICS J Radiol 4: 196.
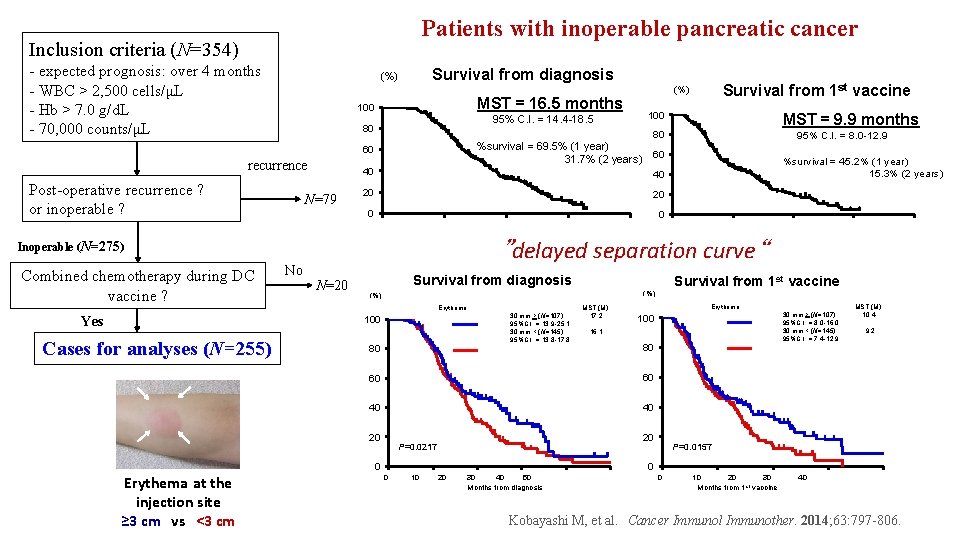
Patients with inoperable pancreatic cancer Inclusion criteria (N=354) - expected prognosis: over 4 months - WBC > 2, 500 cells/μL - Hb > 7. 0 g/d. L - 70, 000 counts/μL Survival from diagnosis (%) MST = 16. 5 months 100 recurrence Post-operative recurrence ? or inoperable ? N=79 60 %survival = 69. 5% (1 year) 31. 7% (2 years) 60 40 40 20 20 0 0 300 Yes N=20 900 300 600 900 1200 Survival from 1 st vaccine (%) Erythema 30 mm > (N=107) 95%C. I. = 13. 9 -25. 1 30 mm < (N=145) 95%C. I. = 13. 8 -17. 8 80 MST (M) 17. 2 Erythema 30 mm > (N=107) 95%C. I. = 8. 0 -16. 0 30 mm < (N=145) 95%C. I. = 7. 4 -12. 9 100 16. 1 MST (M) 10. 4 9. 2 80 60 60 40 40 20 20 P=0. 0217 0 Erythema at the injection site ≥ 3 cm vs <3 cm %survival = 45. 2% (1 year) 15. 3% (2 years) 0 1200 Survival from diagnosis 100 Cases for analyses (N=255) 600 95% C. I. = 8. 0 -12. 9 ”delayed separation curve“ Inoperable (N=275) No MST = 9. 9 months 80 0 Combined chemotherapy during DC vaccine ? 100 95% C. I. = 14. 4 -18. 5 80 Survival from 1 st vaccine (%) P=0. 0157 0 00 10 30020 30 50 600 40 900 Months from diagnosis 1200 1500 00 10 300 20 30 600 Months from 1 st vaccine 40 900 1200 Kobayashi M, et al. Cancer Immunol Immunother. 2014; 63: 797 -806.
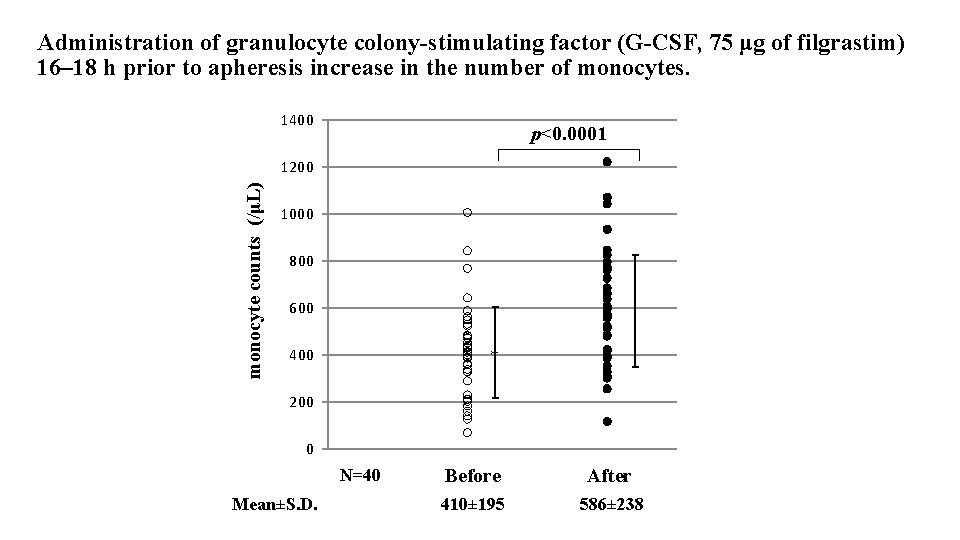
Administration of granulocyte colony-stimulating factor (G-CSF, 75 μg of filgrastim) 16– 18 h prior to apheresis increase in the number of monocytes. 1400 p<0. 0001 monocyte counts (/μL) 1200 1000 800 600 400 200 0 N=40 Mean±S. D. Before 410± 195 After 586± 238
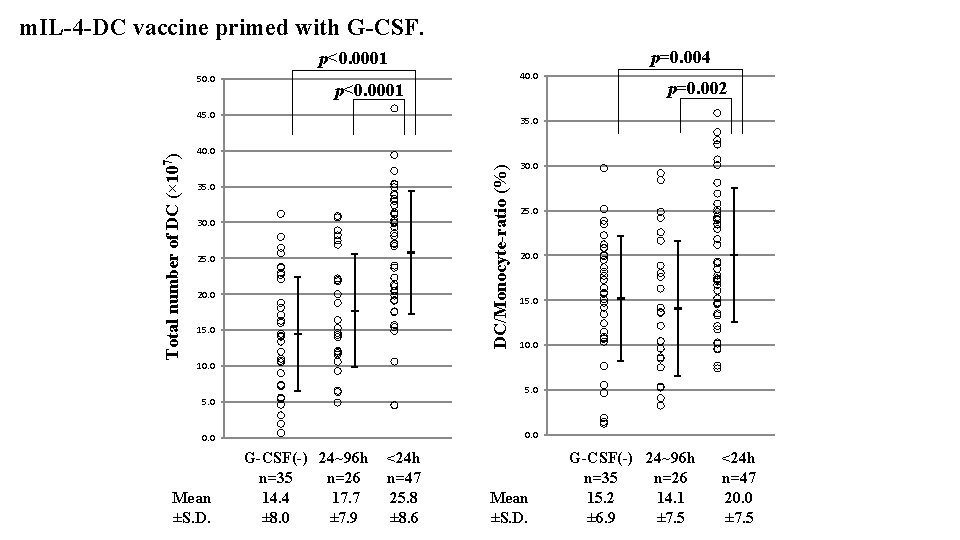
m. IL-4 -DC vaccine primed with G-CSF. p=0. 004 p<0. 0001 50. 0 40. 0 p<0. 0001 35. 0 40. 0 DC/Monocyte-ratio (%) Total number of DC (× 107) 45. 0 p=0. 002 35. 0 30. 0 25. 0 20. 0 15. 0 10. 0 5. 0 0. 0 Mean ±S. D. G-CSF(-) 24~96 h <24 h n=35 n=26 n=47 14. 4 25. 8 17. 7 ± 8. 0 ± 8. 6 ± 7. 9 Mean ±S. D. G-CSF(-) 24~96 h n=35 n=26 15. 2 14. 1 ± 6. 9 ± 7. 5 <24 h n=47 20. 0 ± 7. 5
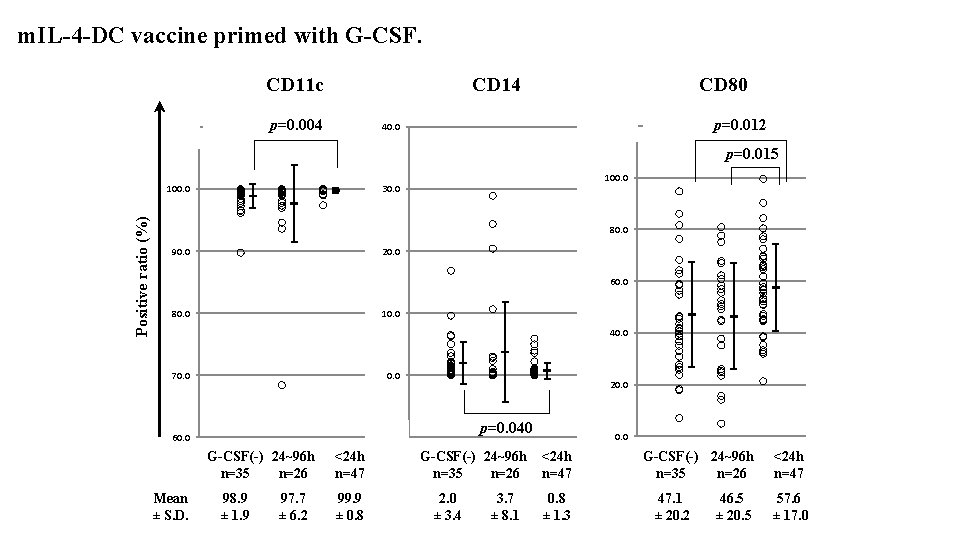
m. IL-4 -DC vaccine primed with G-CSF. CD 11 c CD 14 p=0. 004 110. 0 CD 80 p=0. 012 120. 0 40. 0 p=0. 015 Positive ratio (%) 100. 0 30. 0 80. 0 90. 0 20. 0 60. 0 80. 0 10. 0 40. 0 70. 0 60. 0 (10. 0) G-CSF(-) 24~96 h <24 h n=35 n=26 n=47 Mean ± S. D. 98. 9 ± 1. 9 97. 7 ± 6. 2 99. 9 ± 0. 8 20. 0 p=0. 040 0. 0 G-CSF(-) 24~96 h <24 h n=35 n=26 n=47 2. 0 ± 3. 4 3. 7 ± 8. 1 0. 8 ± 1. 3 G-CSF(-) 24~96 h <24 h n=35 n=26 n=47 47. 1 ± 20. 2 46. 5 ± 20. 5 57. 6 ± 17. 0
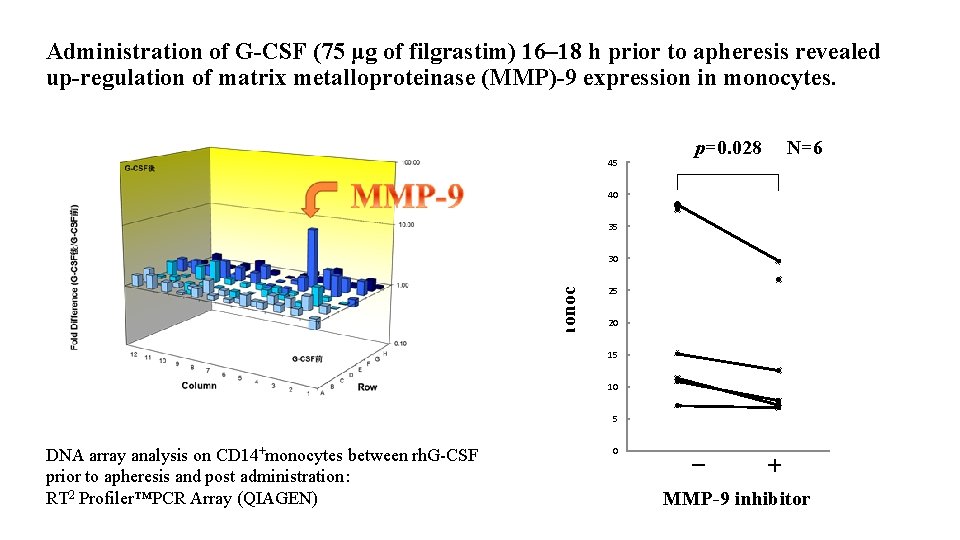
Administration of G-CSF (75 μg of filgrastim) 16– 18 h prior to apheresis revealed up-regulation of matrix metalloproteinase (MMP)-9 expression in monocytes. p=0. 028 N=6 45 m. DC / monocytes (%) 40 35 30 25 20 15 10 5 DNA array analysis on CD 14+monocytes between rh. G-CSF prior to apheresis and post administration: RT 2 Profiler™PCR Array (QIAGEN) 0 0 0. 5 1 1. 5 - + MMP-9 inhibitor 2
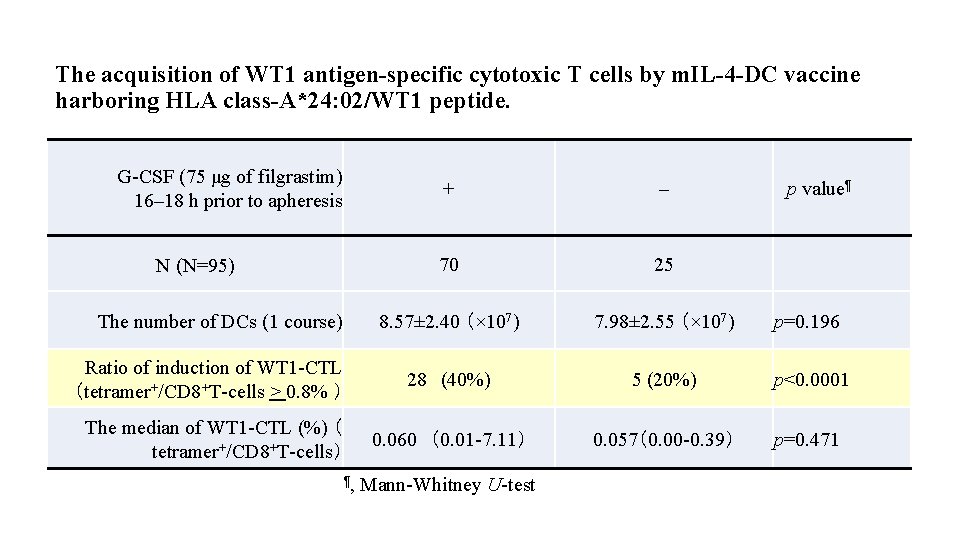
The acquisition of WT 1 antigen-specific cytotoxic T cells by m. IL-4 -DC vaccine harboring HLA class-A*24: 02/WT 1 peptide. G-CSF (75 μg of filgrastim) 16– 18 h prior to apheresis + – 70 25 8. 57± 2. 40 (× 107) 7. 98± 2. 55 (× 107) p=0. 196 Ratio of induction of WT 1 -CTL (tetramer+/CD 8+T-cells > 0. 8% ) 28 (40%) 5 (20%) p<0. 0001 The median of WT 1 -CTL (%) ( tetramer+/CD 8+T-cells) 0. 060 (0. 01 -7. 11) 0. 057(0. 00 -0. 39) p=0. 471 N (N=95) The number of DCs (1 course) ¶, Mann-Whitney U-test p value¶

Patient: 12 year-old-girl Disease onset: March, 2005 Laboratory data: <Peripheral Blood> WBC 191, 170/μl (blast 82%) <Bone marrow> NCC 720. 0 x 103/μl (blast 85. 8%) CD 9 78%, CD 10 95%, CD 19 95%, CD 20 35%, CD 22 67%, CD 24 89%, CD 33 0%, CD 34 95%, HLA-DR 97%, Cy-μ 1%, Karyotype: 46, XX [ 20] WT 1 m. RNA: 1. 1 x 104 copies/μg. RNA Diagnosis : B-cell precursor acute lymphoblastic leukemia Saito S, et al: Safety and tolerability of allogeneic dendritic cell vaccination with induction of WT 1 -specific T cells in a pediatric donor and pediatric patient with relapsed leukemia: A case report and review of the literature. Cytotherapy. 2015; 17: 330 -5.
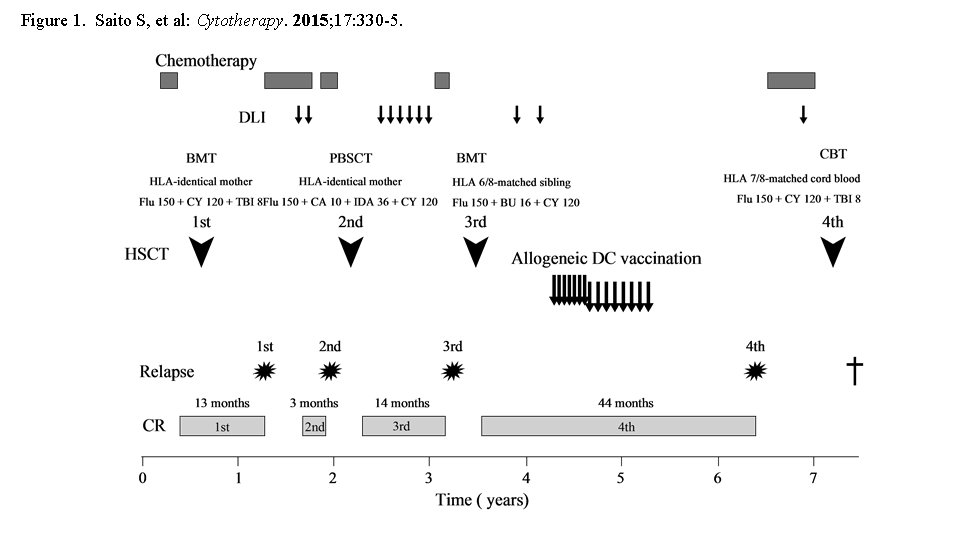
Figure 1. Saito S, et al: Cytotherapy. 2015; 17: 330 -5.
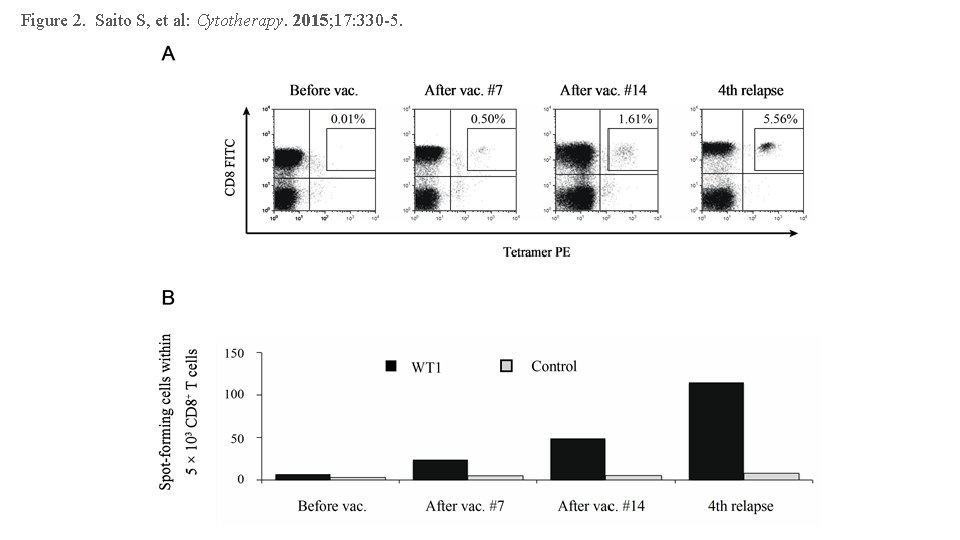
Figure 2. Saito S, et al: Cytotherapy. 2015; 17: 330 -5.
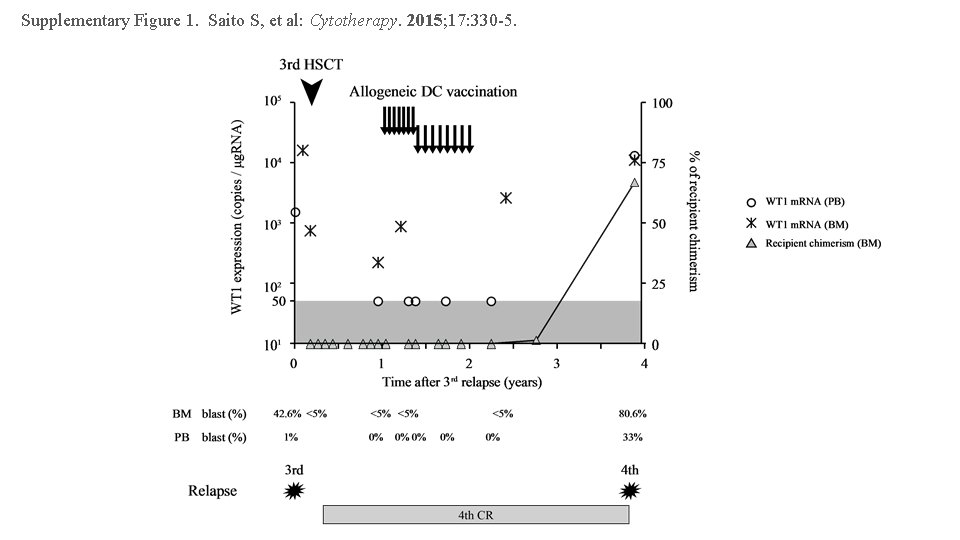
Supplementary Figure 1. Saito S, et al: Cytotherapy. 2015; 17: 330 -5.

Conclusion • DC-based immunotherapy targeting WT 1 was indicated to be safe and feasible for the management of advanced cancers exhibiting ‘delayed separation’ curve in some patients. • DC vaccine primed with G-CSF harboring HLA class-A*24: 02/WT 1 peptide exhibited a significant increase in the acquisition of WT 1 antigen-specific cytotoxic T cells. • Allogenic WT 1 -DC vaccination may be safe, tolerable, and even feasible for pediatric donors and patients with relapsed leukemia after hematopoietic stem cell transplantation (HSCT). This strategy would be relevant to the scope of the development of personalized therapy in HSCT. • In future, the blockade of immune checkpoints in combination with DCvaccination would be promising therapeutic strategies to activate therapeutic antitumor immunity for advanced cancers and hematological malignancies.
- Slides: 37