Demonstrate understanding of spectroscopic data in chemistry Chemistry






- Slides: 13

Demonstrate understanding of spectroscopic data in chemistry Chemistry A. S. 3. 2 91388 3 internal credits

Spectroscopy • Spectroscopy is the study of how EMR interacts with matter. • High wavelength = low energy • • We will look at 3 spectroscopic techniques: IR = infrared spectroscopy NMR = nuclear magnetic resonance spectroscopy Mass spectroscopy

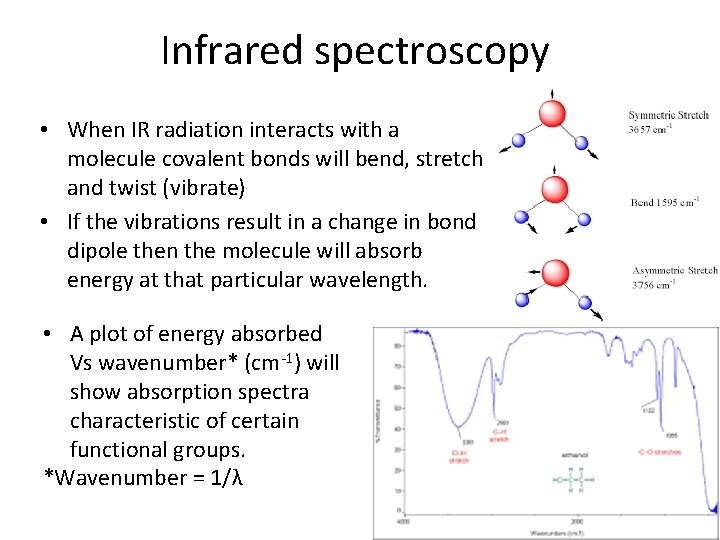
Infrared spectroscopy • When IR radiation interacts with a molecule covalent bonds will bend, stretch and twist (vibrate) • If the vibrations result in a change in bond dipole then the molecule will absorb energy at that particular wavelength. • A plot of energy absorbed Vs wavenumber* (cm-1) will show absorption spectra characteristic of certain functional groups. *Wavenumber = 1/λ

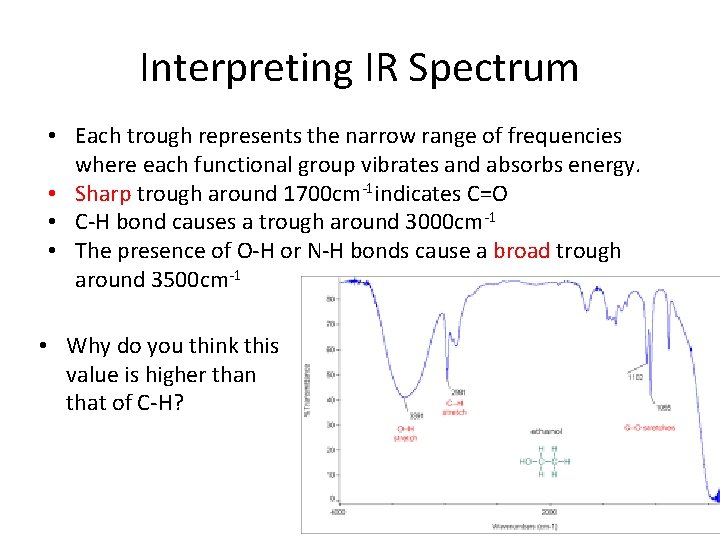
Interpreting IR Spectrum • Each trough represents the narrow range of frequencies where each functional group vibrates and absorbs energy. • Sharp trough around 1700 cm-1 indicates C=O • C-H bond causes a trough around 3000 cm-1 • The presence of O-H or N-H bonds cause a broad trough around 3500 cm-1 • Why do you think this value is higher than that of C-H?

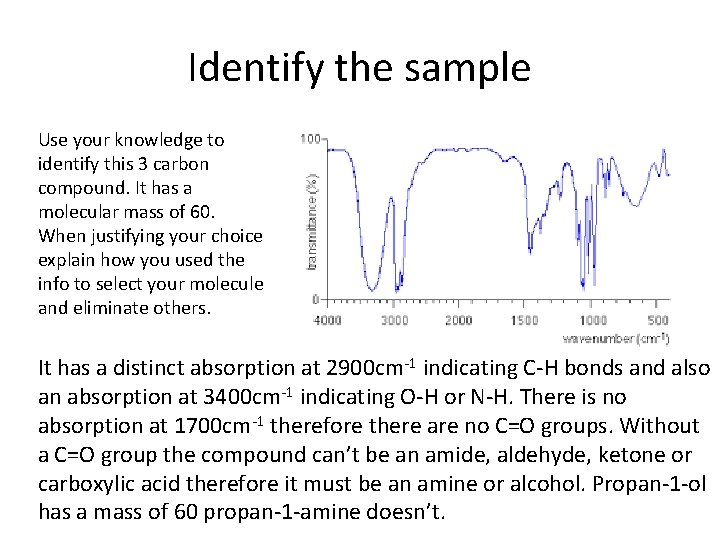
Identify the sample Use your knowledge to identify this 3 carbon compound. It has a molecular mass of 60. When justifying your choice explain how you used the info to select your molecule and eliminate others. It has a distinct absorption at 2900 cm-1 indicating C-H bonds and also an absorption at 3400 cm-1 indicating O-H or N-H. There is no absorption at 1700 cm-1 therefore there are no C=O groups. Without a C=O group the compound can’t be an amide, aldehyde, ketone or carboxylic acid therefore it must be an amine or alcohol. Propan-1 -ol has a mass of 60 propan-1 -amine doesn’t.

13 C NMR Spectroscopy • Powerful magnets are used to influence the spin of nuclei • Electrons around an atom will shield the nucleus from the magnetic field • Chemical shift ( δ ) is a measure, in parts per million (ppm), of how much the nucleus is affected. i. e. greater ppm = less shielded • The chemical shift is affected by: – The electronegativity of elements e. g. F will draw electrons away from C – The type of bonding e. g. double bonds draw electrons away from C – chemical environment e. g. the shielding of each C atom is dependent on the total of all the shielding effects acting on it.

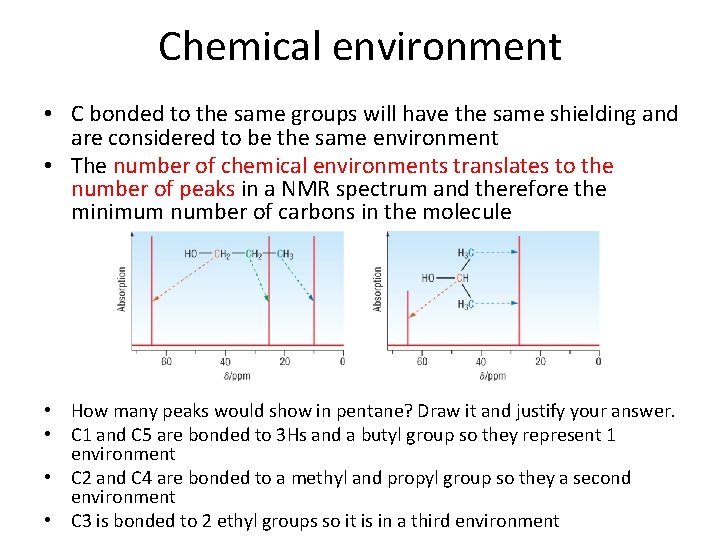
Chemical environment • C bonded to the same groups will have the same shielding and are considered to be the same environment • The number of chemical environments translates to the number of peaks in a NMR spectrum and therefore the minimum number of carbons in the molecule • How many peaks would show in pentane? Draw it and justify your answer. • C 1 and C 5 are bonded to 3 Hs and a butyl group so they represent 1 environment • C 2 and C 4 are bonded to a methyl and propyl group so they a second environment • C 3 is bonded to 2 ethyl groups so it is in a third environment

Interpreting NMR spectrum • The number of peaks = number of environments • C=O (aldehyde, ketone) appears at 180 -220 ppm • C=O (carboxylic acid, ester, acyl chloride, amide) appears at 160 -185 ppm • C=C appears at 100 -150 ppm (if only 1 line appears the molecule is symmetrical about the C=C bond) • C-O bond in Esters 60 -80 • C- high EN element or C-C=O appears at 30 -60 ppm • CH 3 appears at 0 - 30 ppm • Ignore any signal at 75 ppm this is the solvent

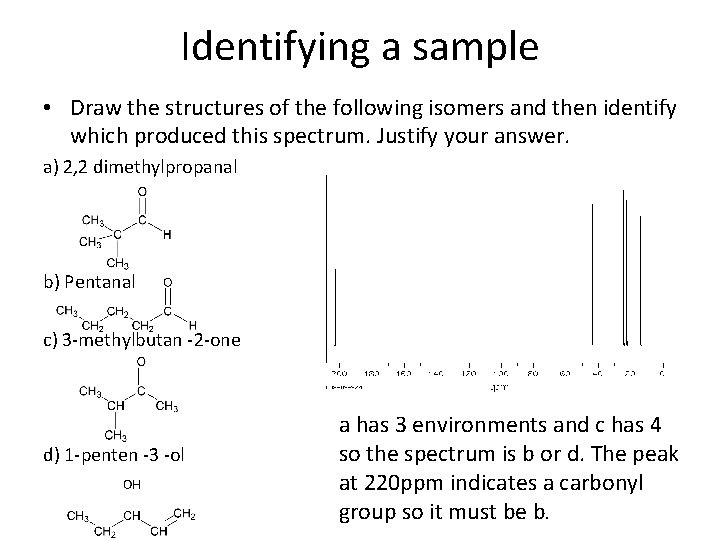
Identifying a sample • Draw the structures of the following isomers and then identify which produced this spectrum. Justify your answer. a) 2, 2 dimethylpropanal b) Pentanal c) 3 -methylbutan -2 -one d) 1 -penten -3 -ol a has 3 environments and c has 4 so the spectrum is b or d. The peak at 220 ppm indicates a carbonyl group so it must be b.

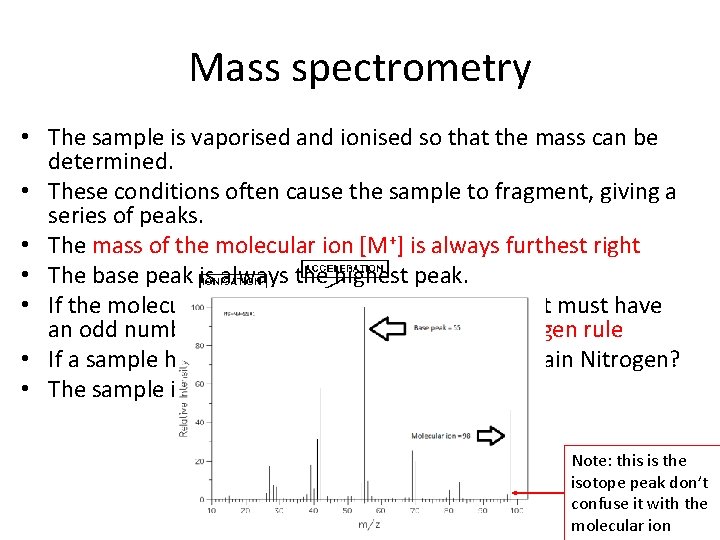
Mass spectrometry • The sample is vaporised and ionised so that the mass can be determined. • These conditions often cause the sample to fragment, giving a series of peaks. • The mass of the molecular ion [M+] is always furthest right • The base peak is always the highest peak. • If the molecular ion has an odd numbered mass it must have an odd number of N atoms in the sample = Nitrogen rule • If a sample has a molecular ion of 31 does it contain Nitrogen? • The sample is an amine what is it called? Note: this is the isotope peak don’t confuse it with the molecular ion

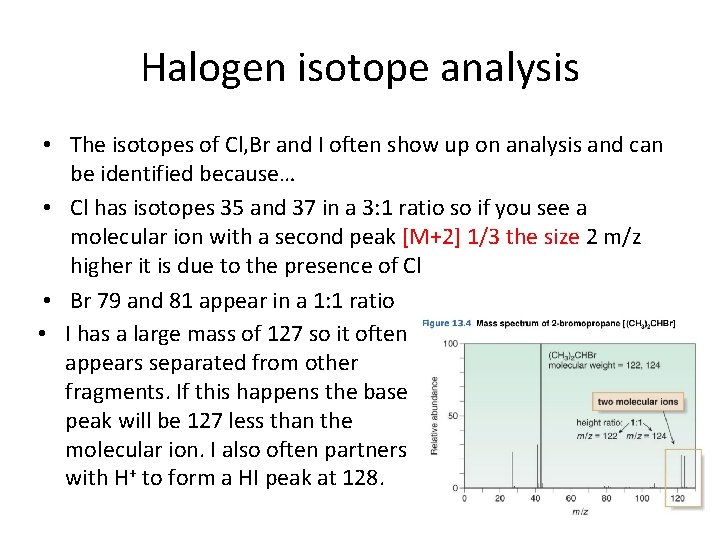
Halogen isotope analysis • The isotopes of Cl, Br and I often show up on analysis and can be identified because… • Cl has isotopes 35 and 37 in a 3: 1 ratio so if you see a molecular ion with a second peak [M+2] 1/3 the size 2 m/z higher it is due to the presence of Cl • Br 79 and 81 appear in a 1: 1 ratio • I has a large mass of 127 so it often appears separated from other fragments. If this happens the base peak will be 127 less than the molecular ion. I also often partners with H+ to form a HI peak at 128.

Fragmentation analysis • When energy is supplied to the sample it can break into fragments which give an indication of functional groups. • If there is a peak 1, 15, 17 or 18 less than the molecular ion it indicates… Fragment mass Fragment lost Indicates the presence of 1 Hydrogen COOH, CHO or NH 15 Methyl CH 3 17 Hydroxyl COOH or OH 18 water OH usually alcohol

Comparing data • Use the different spectroscopic data to identify the unknown sample. • Mass spectroscopy will give you info on the mass and the presence of N, Cl, Br, I • NMR will give you info about the structure and the presence of carbonyl or C=C • IR will give you info about the functional groups C=O, OH/NH