Delivery of Gene Therapy 2020 MEDWEBUS00118 Viral Vector

Delivery of Gene Therapy 2020 MEDWEB-US-00118
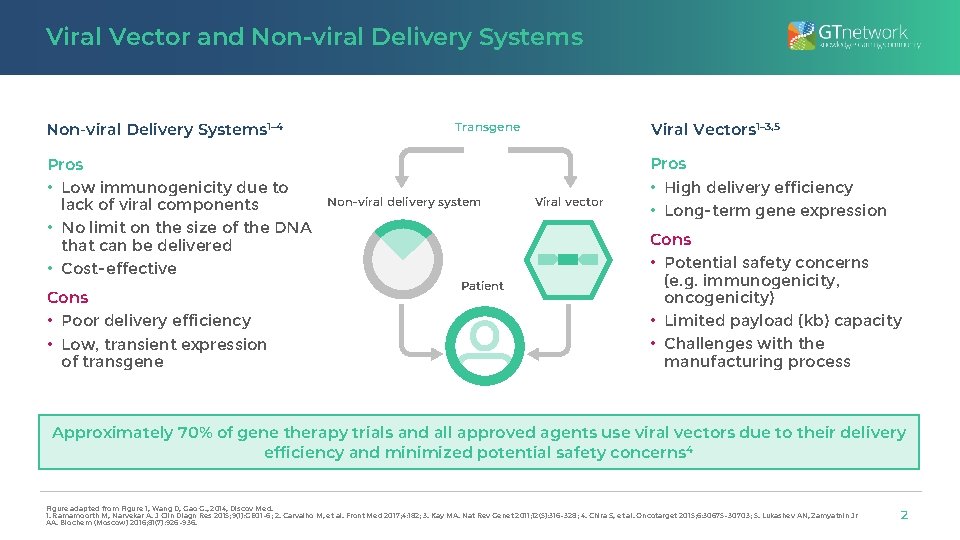
Viral Vector and Non-viral Delivery Systems 1– 4 Pros • Low immunogenicity due to lack of viral components • No limit on the size of the DNA that can be delivered • Cost-effective Cons • Poor delivery efficiency • Low, transient expression of transgene Viral Vectors 1– 3, 5 Transgene Non-viral delivery system Patient Viral vector Pros • High delivery efficiency • Long-term gene expression Cons • Potential safety concerns (e. g. immunogenicity, oncogenicity) • Limited payload (kb) capacity • Challenges with the manufacturing process Approximately 70% of gene therapy trials and all approved agents use viral vectors due to their delivery efficiency and minimized potential safety concerns 4 Figure adapted from Figure 1, Wang D, Gao G. , 2014, Discov Med. 1. Ramamoorth M, Narvekar A. J Clin Diagn Res 2015; 9(1): GE 01– 6; 2. Carvalho M, et al. Front Med 2017; 4: 182; 3. Kay MA. Nat Rev Genet 2011; 12(5): 316– 328; 4. Chira S, et al. Oncotarget 2015; 6: 30675– 30703; 5. Lukashev AN, Zamyatnin Jr AA. Biochem (Moscow) 2016; 81(7): 926– 936. 2
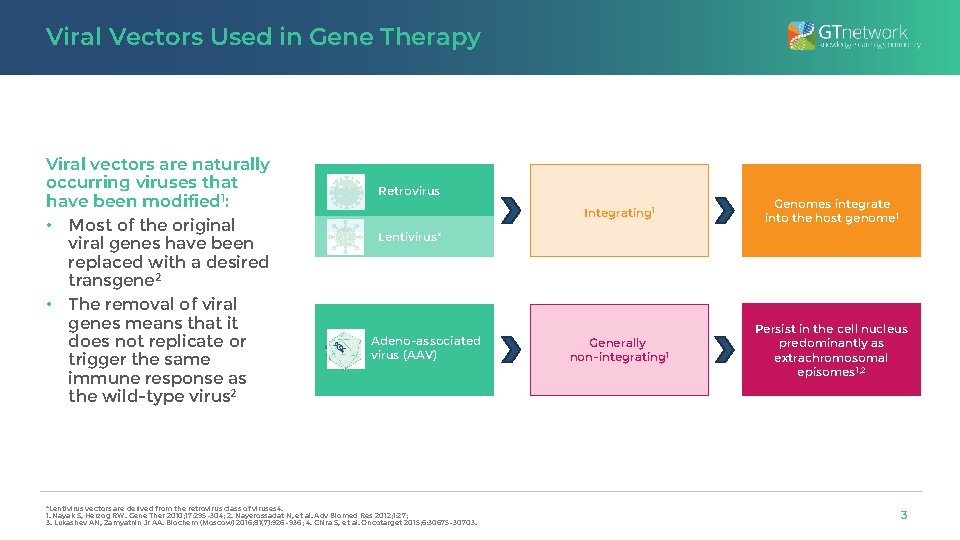
Viral Vectors Used in Gene Therapy Viral vectors are naturally occurring viruses that have been modified 1: • Most of the original viral genes have been replaced with a desired transgene 2 • The removal of viral genes means that it does not replicate or trigger the same immune response as the wild-type virus 2 Retrovirus Integrating 1 Genomes integrate into the host genome 1 Generally non-integrating 1 Persist in the cell nucleus predominantly as extrachromosomal episomes 1, 2 Lentivirus* Adeno-associated virus (AAV) *Lentivirus vectors are derived from the retrovirus class of viruses 4. 1. Nayak S, Herzog RW. Gene Ther 2010; 17: 295– 304; 2. Nayerossadat N, et al. Adv Biomed Res 2012; 1: 27; 3. Lukashev AN, Zamyatnin Jr AA. Biochem (Moscow) 2016; 81(7): 926– 936; 4. Chira S, et al. Oncotarget 2015; 6: 30675– 30703. 3
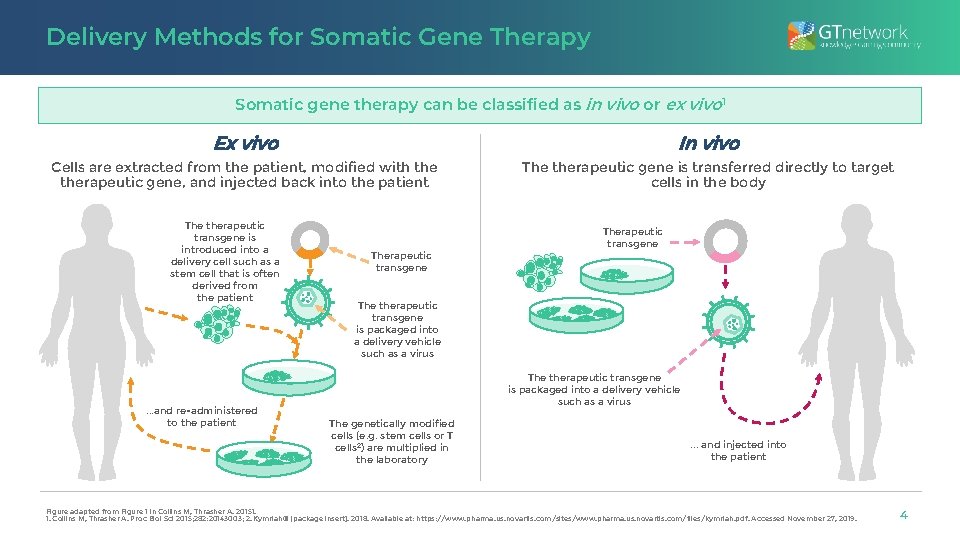
Delivery Methods for Somatic Gene Therapy Somatic gene therapy can be classified as in vivo or ex vivo 1 Ex vivo In vivo Cells are extracted from the patient, modified with therapeutic gene, and injected back into the patient The therapeutic gene is transferred directly to target cells in the body The therapeutic transgene is introduced into a delivery cell such as a stem cell that is often derived from the patient . . . and re-administered to the patient Therapeutic transgene The therapeutic transgene is packaged into a delivery vehicle such as a virus The genetically modified cells (e. g. stem cells or T cells 2) are multiplied in the laboratory . . . and injected into the patient Figure adapted from Figure 1 in Collins M, Thrasher A. 20151. 1. Collins M, Thrasher A. Proc Biol Sci 2015; 282: 20143003; 2. Kymriah® [package insert]. 2018. Available at: https: //www. pharma. us. novartis. com/sites/www. pharma. us. novartis. com/files/kymriah. pdf. Accessed November 27, 2019. 4
- Slides: 4