Definitions and Indicators 1 AcidBase Definitions Definition 1

Definitions and Indicators 1

Acid/Base Definitions • Definition #1: Arrhenius (traditional) • Acids – produce H+ ions (or hydronium ions H 3 O+) • Bases – produce OH- ions • Problem: Some bases don’t have hydroxide ions! 2
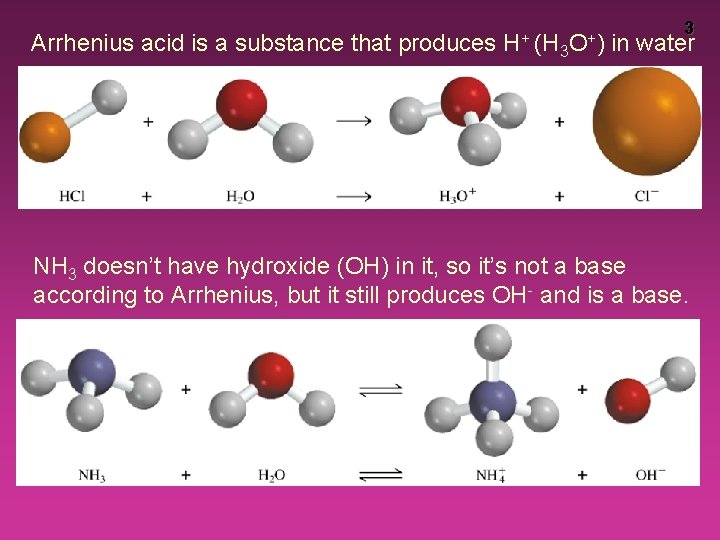
Arrhenius acid is a substance that produces H+ (H 3 O+) 3 in water NH 3 doesn’t have hydroxide (OH) in it, so it’s not a base according to Arrhenius, but it still produces OH- and is a base.
Acid/Base Definitions • Definition #2: Brønsted – Lowry 4 • Acids – proton donor • Bases – proton acceptor • A “proton” is the same as a hydrogen ion. It’s a hydrogen atom that lost its electron.
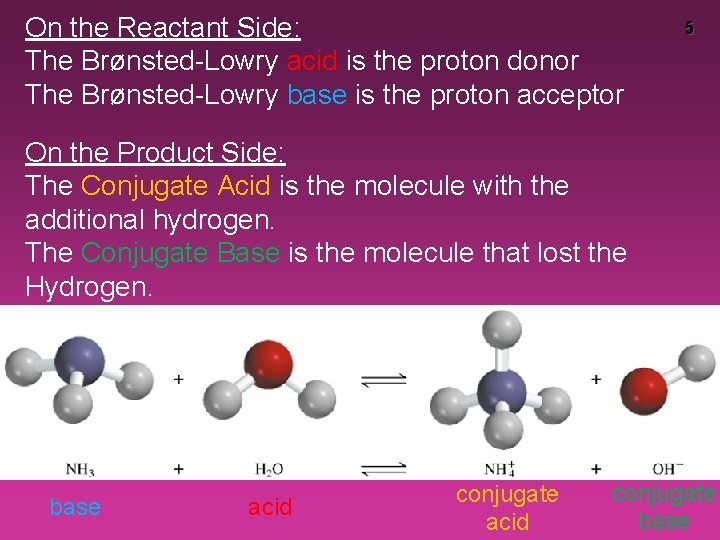
On the Reactant Side: The Brønsted-Lowry acid is the proton donor The Brønsted-Lowry base is the proton acceptor 5 On the Product Side: The Conjugate Acid is the molecule with the additional hydrogen. The Conjugate Base is the molecule that lost the Hydrogen. base acid conjugate base
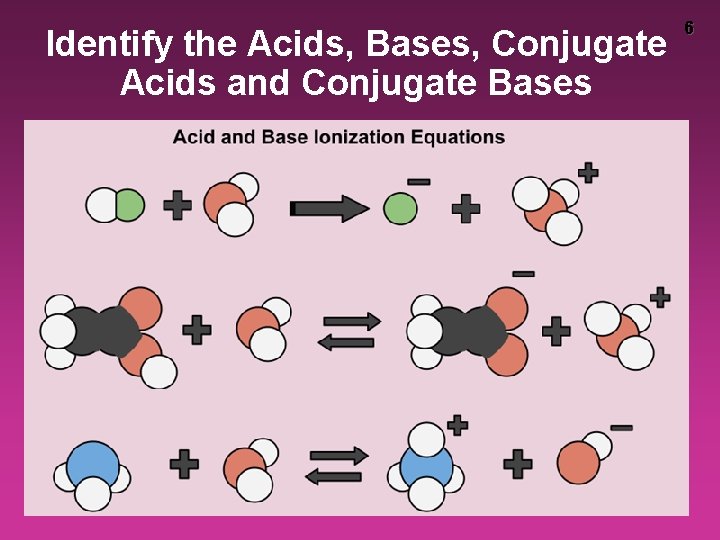
Identify the Acids, Bases, Conjugate Acids and Conjugate Bases 6
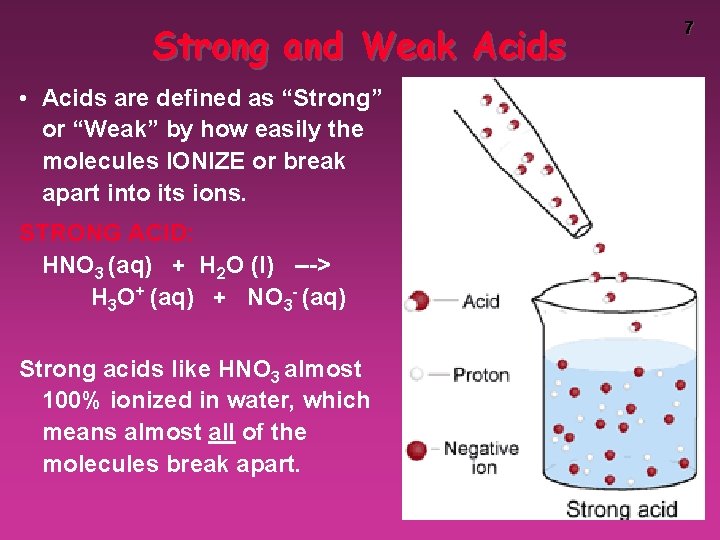
Strong and Weak Acids • Acids are defined as “Strong” or “Weak” by how easily the molecules IONIZE or break apart into its ions. STRONG ACID: HNO 3 (aq) + H 2 O (l) ---> H 3 O+ (aq) + NO 3 - (aq) Strong acids like HNO 3 almost 100% ionized in water, which means almost all of the molecules break apart. 7
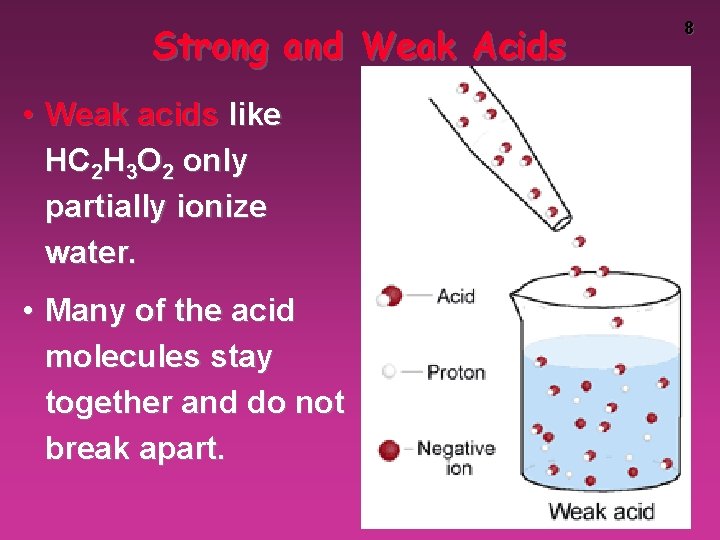
Strong and Weak Acids • Weak acids like HC 2 H 3 O 2 only partially ionize water. • Many of the acid molecules stay together and do not break apart. 8

9 Can you rank these unknown acids in terms of their strength?
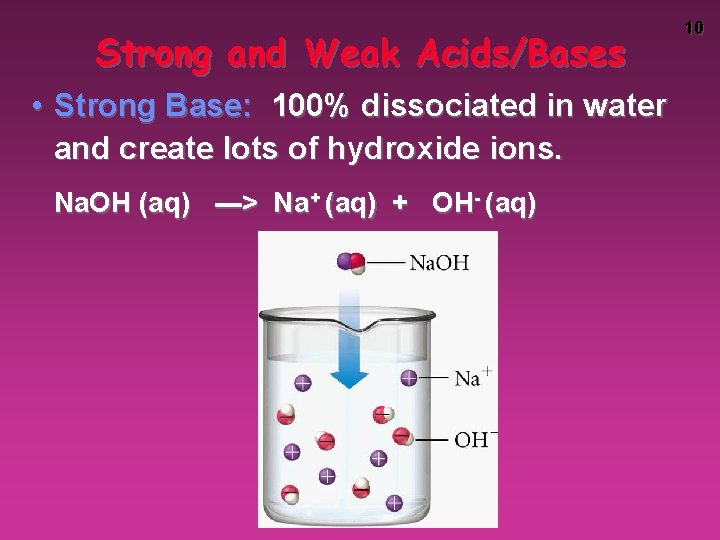
Strong and Weak Acids/Bases • Strong Base: 100% dissociated in water and create lots of hydroxide ions. Na. OH (aq) ---> Na+ (aq) + OH- (aq) 10

Strong and Weak Acids/Bases • Weak base: Only a few molecules dissociate and very few hydroxide ions are created. NH 3 (aq) + H 2 O (l) NH 4+ (aq) + OH- (aq) 11
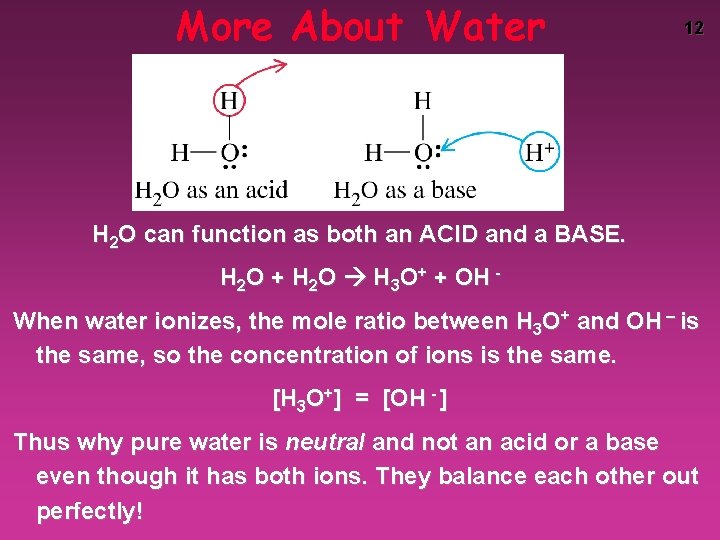
More About Water 12 H 2 O can function as both an ACID and a BASE. H 2 O + H 2 O H 3 O+ + OH When water ionizes, the mole ratio between H 3 O+ and OH – is the same, so the concentration of ions is the same. [H 3 O+] = [OH - ] Thus why pure water is neutral and not an acid or a base even though it has both ions. They balance each other out perfectly!

p. H testing • There are several ways to test p. H • Indicators: Indicators contain chemicals that are receptive and change colors in the presence of acids and bases. – Blue litmus paper (red = acid) – Red litmus paper (blue = basic) – p. H paper (multi-colored) – Universal indicator (multi-colored) – Indicators like phenolphthalein – Natural indicators like red cabbage, radishes 13

Paper testing • Paper tests like litmus paper and p. H paper – Put a stirring rod into the solution and stir. – Take the stirring rod out, and place a drop of the solution from the end of the stirring rod onto a piece of the paper – Read and record the color change. Note what the color indicates. – You should only use a small portion of the paper. You can use one piece of paper for several tests. 14

p. H meter • • Tests the voltage of the electrolyte Converts the voltage to p. H Very cheap, accurate The probe must be calibrated before using. 15
- Slides: 15