Definition of ACID Anything that produces hydrogen ions
















- Slides: 24

Definition of ACID: Anything that produces hydrogen ions in a water solution. HCl H+ + Cl- Proton donor – adds H+ to solvent

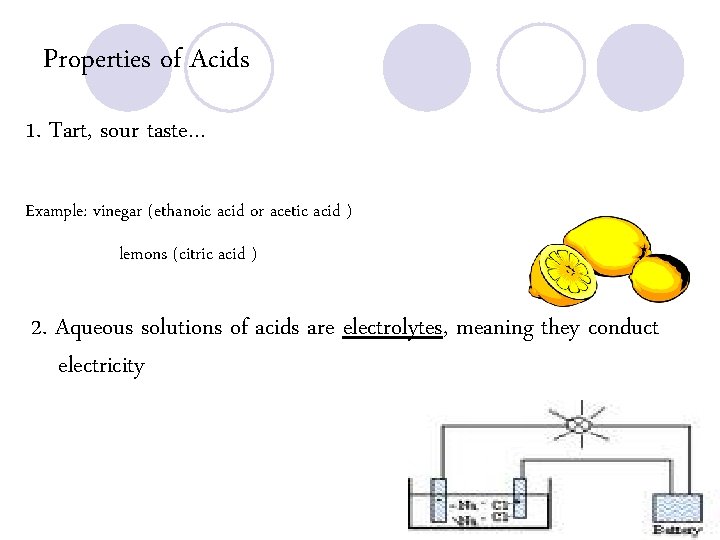
Properties of Acids 1. Tart, sour taste… Example: vinegar (ethanoic acid or acetic acid ) lemons (citric acid ) 2. Aqueous solutions of acids are electrolytes, meaning they conduct electricity

More Properties of Acids 3. They cause dyes called indicators to change color…turn blue litmus paper red 4. Metals react with acid solutions to form hydrogen… the acids corrode metals. 5. ACIDS react with BASES to form water and salt…neutralization rxn

Definition of BASE: Anything that produces hydroxide ions in a water solution. Na. OH Na+ + OH- Proton acceptor – removes H+ from solvent

Properties of Bases 1. React with acids to form water and salt 2. Taste bitter 3. Feel slippery 4. Can be strong or weak electrolytes 5. Change the colors of an indicator. . turn red litmus to blue Example: milk of magnesia (Mg. OH)

Common Bases all have OH at the end KOH Potassium Hydroxide Na. OH sodium Hydroxide Ca(OH) 2 calcium hydroxide Mg (OH) 2 magnesium hydroxide

Strong or Weak Acids and Bases The strength of an acid or base depends NOT on the amount of H+ ions or OHions but on the degree of ionization. What is ionization? It is the process of adding or removing electrons from an atom or molecule, which gives the atom or molecule a net charge.

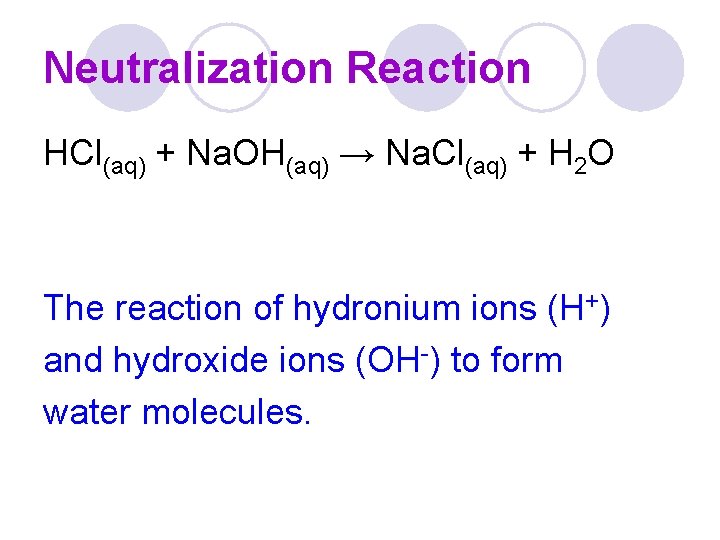
Neutralization Reaction HCl(aq) + Na. OH(aq) → Na. Cl(aq) + H 2 O The reaction of hydronium ions (H+) and hydroxide ions (OH-) to form water molecules.

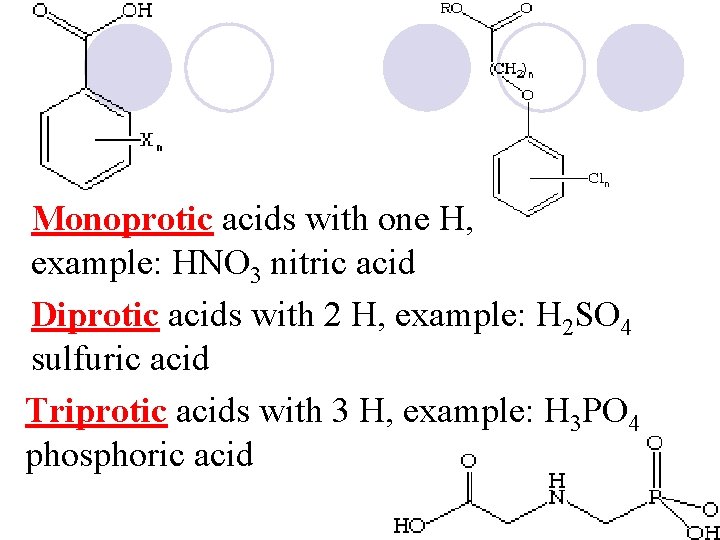
Monoprotic acids with one H, example: HNO 3 nitric acid Diprotic acids with 2 H, example: H 2 SO 4 sulfuric acid Triprotic acids with 3 H, example: H 3 PO 4 phosphoric acid

Rules for Formulas and Naming of Acids *General formula : Ha. Xb , where the “X” could stand for any element or polyatomic ion Formula: Hydrogen + another element Naming: Change the original endings of ions to the following: “ide”-----”ic”, with hydro at the beginning With polyatomics, just change “ite”-to-- “ous” & “ate”-to-”ic”

Formulas and Names of Acids Examples: HCl hydrogen chloride becomes HNO 3 hydrogen nitrate becomes H 2 SO 3 hydrogen sulfite becomes H 2 CO 3 hydrogen carbonate becomes HCl. O 3 hydrogen chlorate becomes HF hydrogen fluoride becomes

Naming Bases name bases normally: Na. OH = sodium hydroxide Ca(OH) 2 = calcium hydroxide Neutral - being neither acidic nor basic Example: pure water

Salts l Salts are ionic compouns that contain a positive ion that is not H+ and a negative ion that is not OHl Have high melting points l Good conductors of electric current

Aqueous solutions of Acids l The hydrogen ion in aqueous solution is best known as a hydronium ion: H 3 O+ l An example of an acid ionizing would be: l HNO 3(l) + H 2 O(l) H 3 O+(aq) + NO 3 -(aq)


p. H Scale l p. H of a solution is defined as the negative of the common logarithm of the hydronium atom concentration (H 3 O+) l p. OH negative of the common logarithm of hydroxide ion concentration (OH-) l p. H + p. OH = 14

p. H and p. OH l If p. H = -log (1 x 10 -6) p. H = 6 We know p. H + p. OH = 14 So 6 + p. OH = 14 p. OH = 8

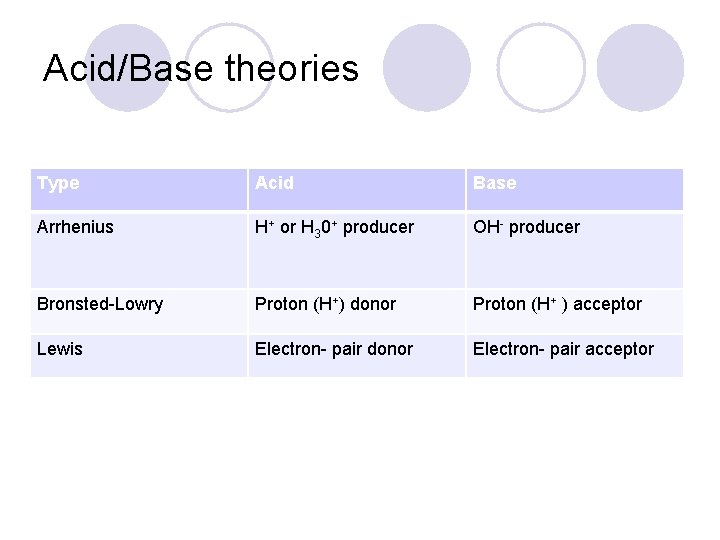
Acid/Base theories Type Acid Base Arrhenius H+ or H 30+ producer OH- producer Bronsted-Lowry Proton (H+) donor Proton (H+ ) acceptor Lewis Electron- pair donor Electron- pair acceptor

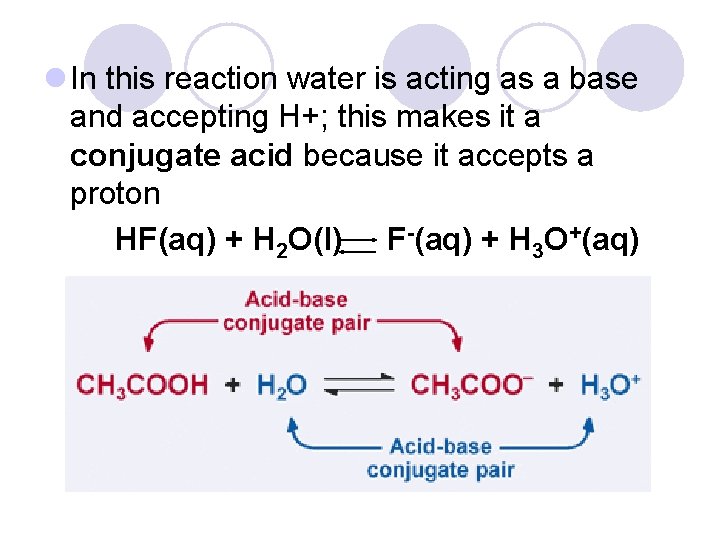
Conjugate Acids and Bases l When an acid gives up a proton (H+) it can re-accept that proton and act as a base this is known as a conjugate base HF(aq) + H 2 O(l) F-(aq) + H 3 O+(aq)

l In this reaction water is acting as a base and accepting H+; this makes it a conjugate acid because it accepts a proton HF(aq) + H 2 O(l) F-(aq) + H 3 O+(aq)

Strength of conjugate acids and bases l HCl is a strong acid ¡This means it gives up H+ ions easily ¡Therefore Cl- must be a very weak base l The stronger an acid is the weaker its conjugate base l The stronger the base the weaker the conjugate acid

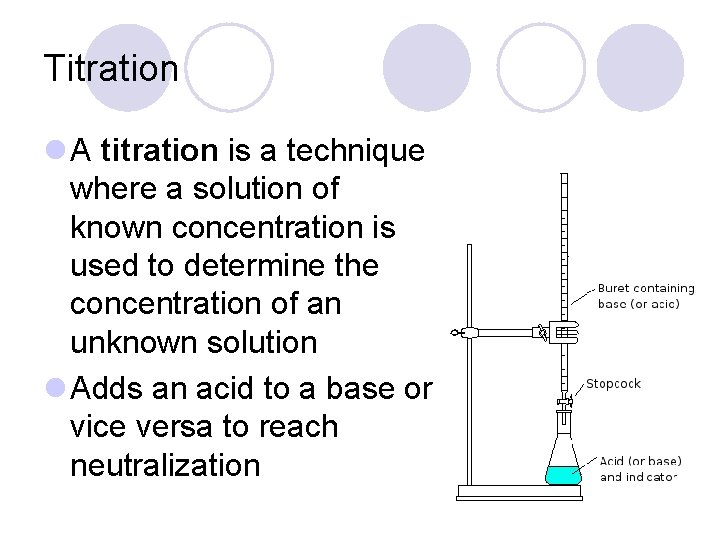
Titration l A titration is a technique where a solution of known concentration is used to determine the concentration of an unknown solution l Adds an acid to a base or vice versa to reach neutralization

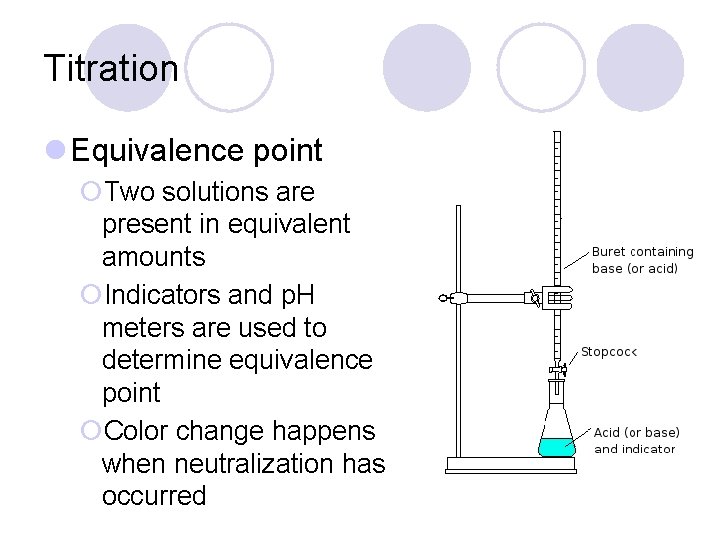
Titration l Equivalence point ¡Two solutions are present in equivalent amounts ¡Indicators and p. H meters are used to determine equivalence point ¡Color change happens when neutralization has occurred

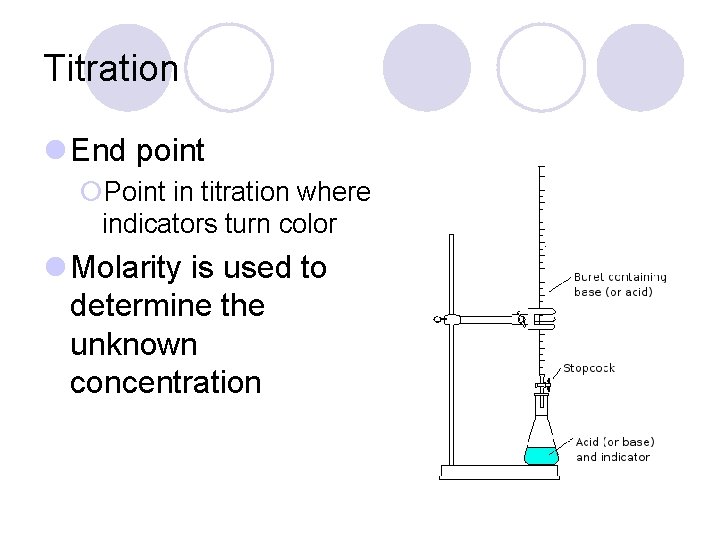
Titration l End point ¡Point in titration where indicators turn color l Molarity is used to determine the unknown concentration