Deferred Prosecution Agreements and Voluntary Disclosures By Darryl

Deferred Prosecution Agreements and Voluntary Disclosures By: Darryl S. Neier, CFE, MS Sobel & Co. , LLC 293 Eisenhower Parkway Livingston, NJ 07039 973 -994 -9494 Darryl. Neier@sobel-cpa. com

DPAs and NPAs • The difference between DPAs and NPAs: Ø DOJ files a criminal information in federal court Ø While NPAs generally are not filed in court • DOJs increasing reliance on DPAs and NPAs demonstrates its recognition that they are precision instruments to resolve allegations of corporate wrongdoing. • Since 2000, DOJ has entered into 230 reported agreements with corporate entities, extracting a total of $31. 6 billion in fines, penalties, forfeitures, and related civil settlements. • The U. S. Securities and Exchange Commission announced the adoption of DPAs and NPAs as part of its Cooperation Initiative in January 2010.
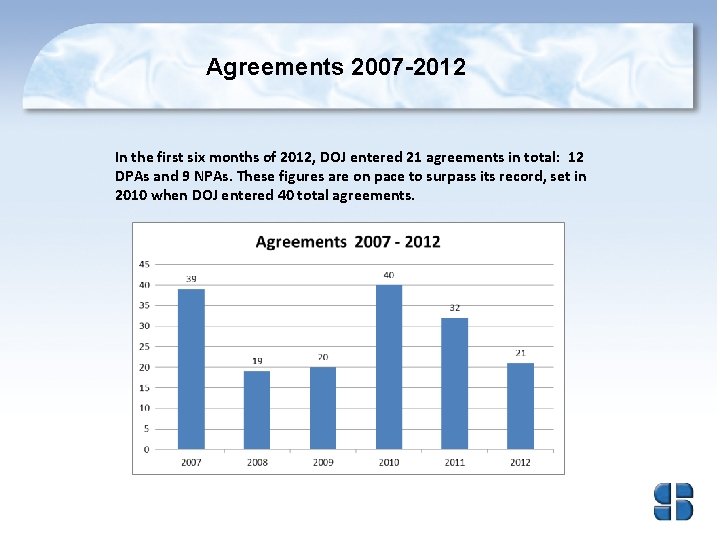
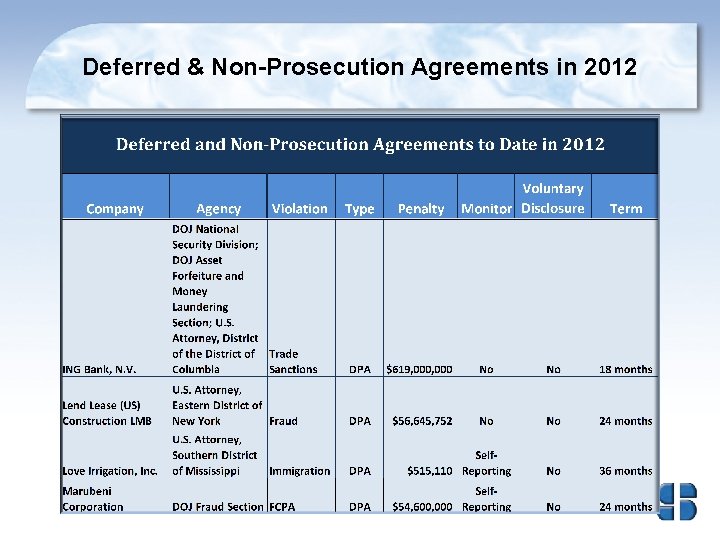
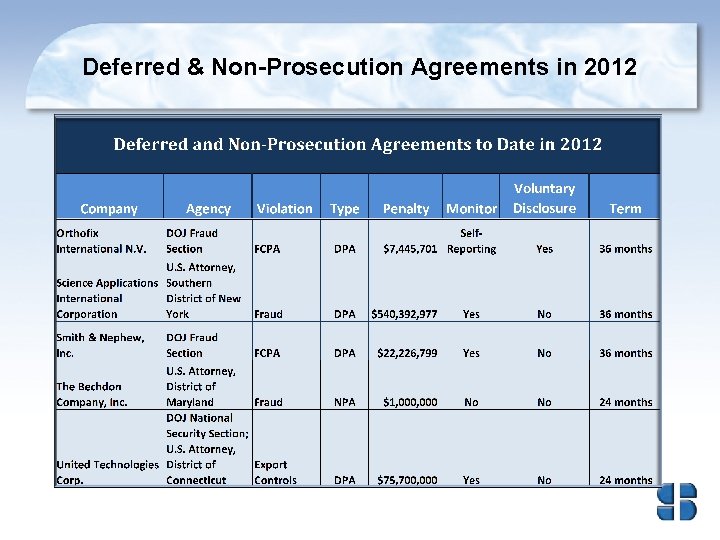
Agreements 2007 -2012 In the first six months of 2012, DOJ entered 21 agreements in total: 12 DPAs and 9 NPAs. These figures are on pace to surpass its record, set in 2010 when DOJ entered 40 total agreements.
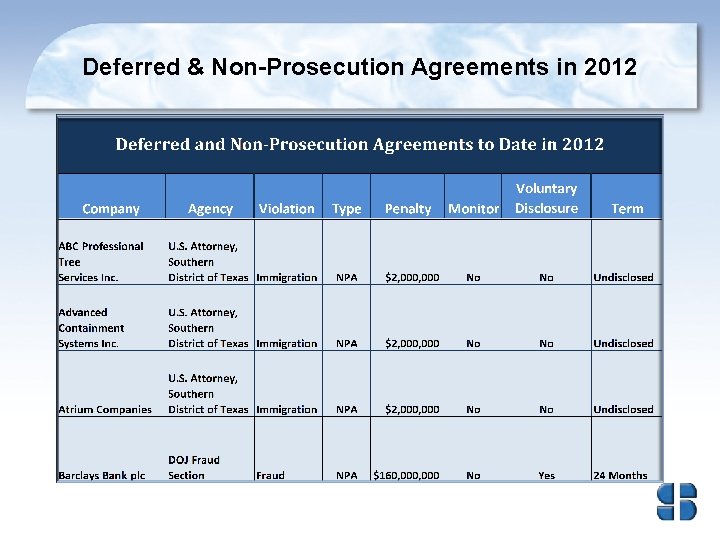
Deferred & Non-Prosecution Agreements in 2012
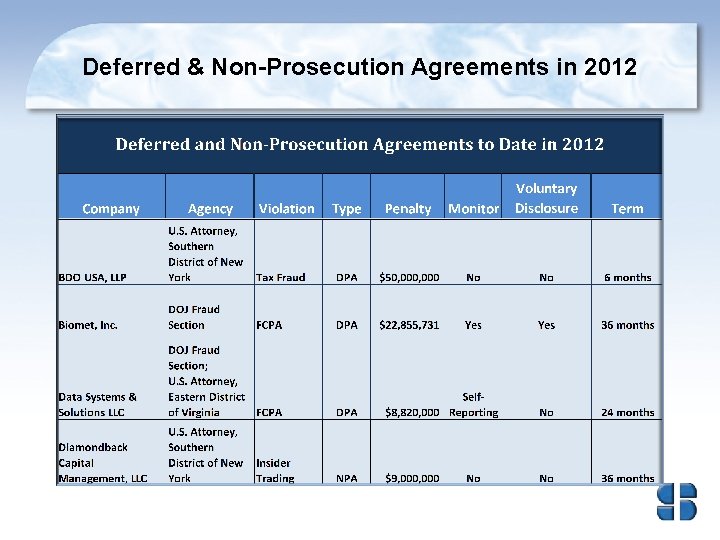
Deferred & Non-Prosecution Agreements in 2012
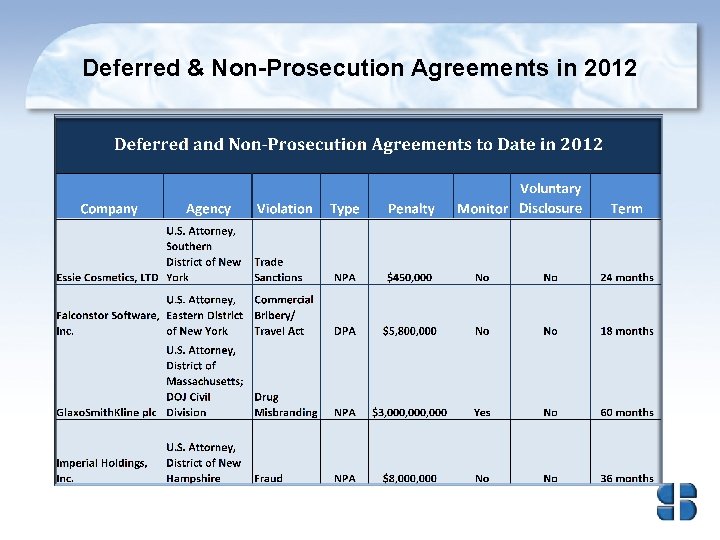
Deferred & Non-Prosecution Agreements in 2012
Deferred & Non-Prosecution Agreements in 2012
Deferred & Non-Prosecution Agreements in 2012

Types of Allegations for 2012 DPA/NPAs Fraud & FCPA have Historically been the Leading Allegations of DPAs and NPAs ALLEGATIONS Fraud 6 FCPA 5 Immigration 4 Trade 3 Bribery 1 Food/Drug Act 1 Insider Trading 1 It should be noted that the number of corporate plea agreements it enters annually far exceed the number of DPAs and NPAs. In 2011, 96 corporate plea agreements were entered into.

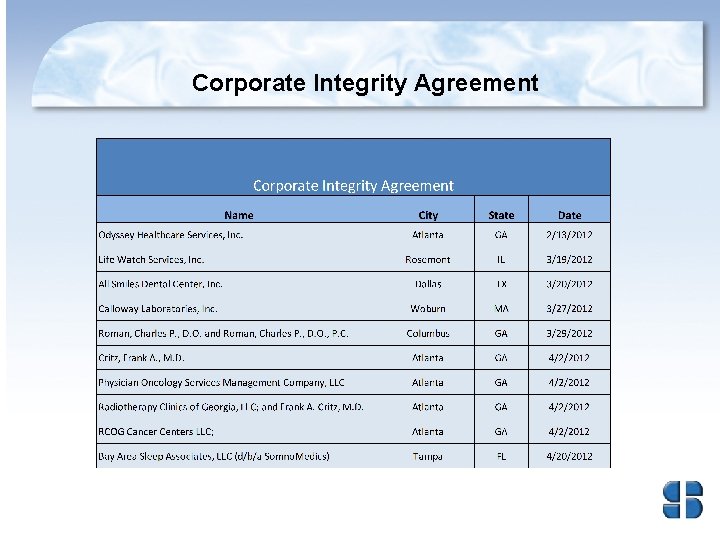
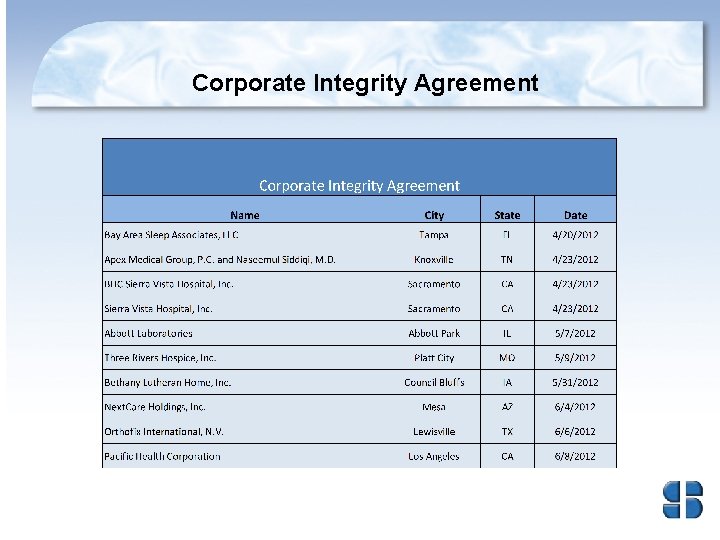
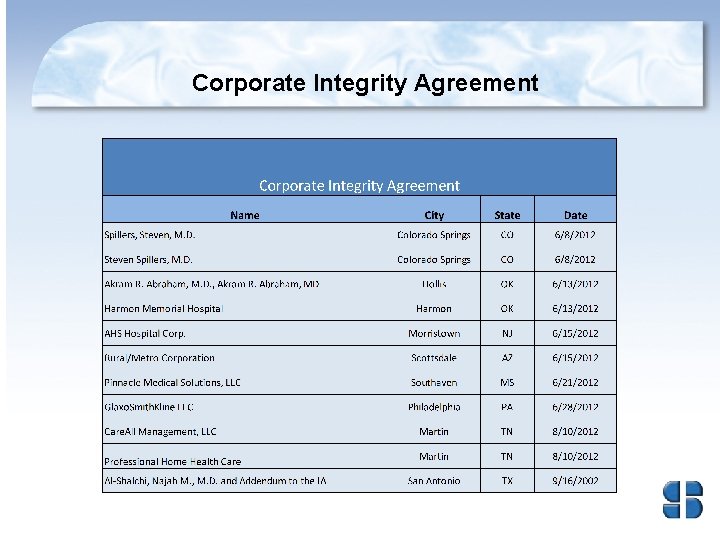
Corporate Integrity Agreements • Corporate integrity agreements (CIAs) are often used by HHS OIG and DOJ with health care providers and other entities as part of the settlement of Federal health care program investigations arising under a variety of civil false claims statues. • The provider or entities agree to the obligations, and in exchange, OIG agrees not to seek their exclusion from participation in Medicare, Medicaid, or other Federal health care programs.

Comprehensive CIA A comprehensive CIA typically lasts 5 years and includes requirements to: • hire a compliance officer/appoint a compliance committee; • develop written standards and policies; • implement a comprehensive employee training program; • retain an independent review organization to conduct annual reviews; • establish a confidential disclosure program; • restrict employment of ineligible persons; • report overpayments, reportable events, and ongoing investigations/legal proceedings; and • provide an implementation report and annual reports to OIG on the status of the entity’s compliance activities. Source: http: //oig. hhs. gov/compliance/corporate-integrity-agreements/index. asp

Health and Human Services – Office of Inspector General • First six months of 2012, HHS OIG reported expected recoveries of about $1. 2 billion consisting of $483. 1 million in audit receivables and $748 million in investigative receivables (which includes $136. 6 million in non-HHS investigative receivables resulting from work in areas such as the States’ shares of Medicaid restitution). • First six months of 2012, thirty-one CIAs signed to include Atlantic Health Care on June 15, 2012. • Providers who wish to voluntarily disclose self-discovered evidence of potential fraud to OIG may do so under the Provider Self-Disclosure Protocol (SDP). (See 63 Fed. Reg. 58, 399. )
Corporate Integrity Agreement
Corporate Integrity Agreement
Corporate Integrity Agreement

Thank You Darryl S. Neier, CFE, MS-ECM Principal in Charge Sobel & Co. , LLC Forensic Accounting and Litigation Service Group

Considerations Regarding Internal Investigations Daniel G. Giaquinto, Esq.

Whether to Commence • Statutory • Discretionary – Triggers/Sources of Information – Seriousness of Allegations – Parallel Investigation – External / Internal factors • Who decides

Purpose • • • Search for the truth Identify Shortcomings/Exposure Guilt/Innocence/Discipline Address Problems/Remedial Measures Governmental Inquiry No Agenda/No Witch Hunt

Who Conducts Investigation • In House Counsel/Other • Regular Outside Counsel • Special Outside Counsel • Need for Credibility is Key

Scope of the Investigation • • Reasonable nexus to initial Information Written or verbal Broad or narrow Focus Ownership of work product Authority to waive privilege Reporting requirements Authorization to expand

Investigative Plan • Identify universe of relevant hard copy and electronic documents /“litigation hold” • Identify relevant employees for Interviews • Order of interviews • Appropriate warnings • Document review to refresh recollection of witness

Treatment of Employees • • • General Notice Document hold and retention notices Cooperation and interviews Upjohn warnings Cannot force waiver of the Fifth Indemnify legal fees

Report Findings • Record of Investigation • Report – Evidence – Standard of proof – Findings Based on Evidence – Conclusions Consistent with Findings – Recommendations Consistent with Findings/Conclusions • Deal with aftermath

Thank You Daniel G. Giaquinto, Esq. Dgiaquinto@Dr. Law. com 800 -445 -0954
- Slides: 25