Deduction of Mechanisms 4 2 Transition State Theory

![Deduction of Mechanisms 4. 2 Transition State Theory [M(OH 2)6]n+ kex + OH 2 Deduction of Mechanisms 4. 2 Transition State Theory [M(OH 2)6]n+ kex + OH 2](https://slidetodoc.com/presentation_image_h2/9811de8cd2ad3f5e5ddcb8c14471f64d/image-6.jpg)
























![Electron Transfer Reactions Outer sphere electron transfer reactions outer sphere mechanism classical experiment: -[Os(bpy)3]3+ Electron Transfer Reactions Outer sphere electron transfer reactions outer sphere mechanism classical experiment: -[Os(bpy)3]3+](https://slidetodoc.com/presentation_image_h2/9811de8cd2ad3f5e5ddcb8c14471f64d/image-88.jpg)


![Electron Transfer Reactions some more activation parameters for outer sphere reactions Oxidant [Co(bpy)3]3+ [Ru(bpy)3]3+ Electron Transfer Reactions some more activation parameters for outer sphere reactions Oxidant [Co(bpy)3]3+ [Ru(bpy)3]3+](https://slidetodoc.com/presentation_image_h2/9811de8cd2ad3f5e5ddcb8c14471f64d/image-94.jpg)





- Slides: 104

Deduction of Mechanisms 4. 2 Transition State Theory Observations: Reaction rates increase almost ever with increasing T this relation k vs Ea is consistent with the Arrhenius equation k describes the overall reaction rate "constant" A and Ea are considered T independent, mostly correct over a small range of T Transition state theory (TST): = Übergangsfaktor, generally = 1 09. 2021 k. B: Boltzmann h: Planck CHE 323 -FS 18 -T 2 -1

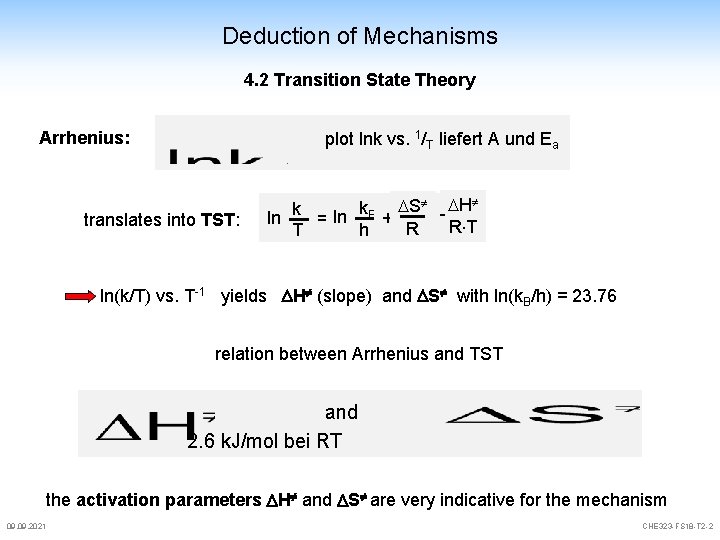
Deduction of Mechanisms 4. 2 Transition State Theory Arrhenius: plot lnk vs. 1/T liefert A und Ea translates into TST: k. B S - H k ln + = ln R T R h T ln(k/T) vs. T-1 yields H (slope) and S with ln(k. B/h) = 23. 76 relation between Arrhenius and TST and 2. 6 k. J/mol bei RT the activation parameters H and S are very indicative for the mechanism 09. 2021 CHE 323 -FS 18 -T 2 -2

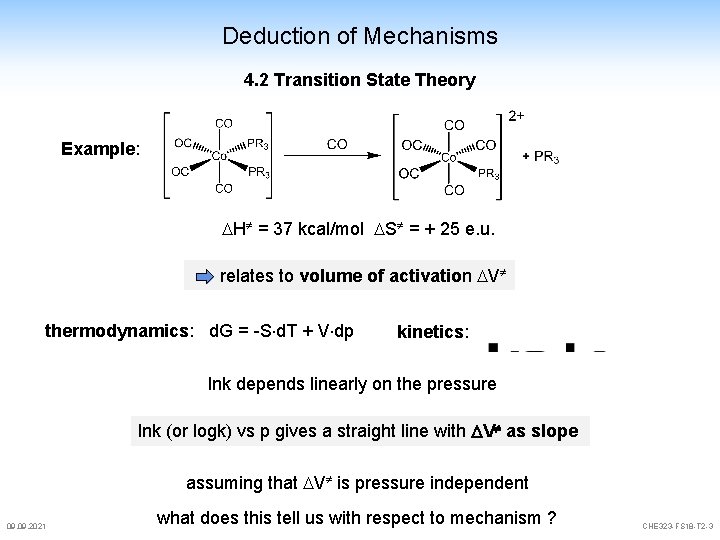
Deduction of Mechanisms 4. 2 Transition State Theory Example: H = 37 kcal/mol S = + 25 e. u. relates to volume of activation V thermodynamics: d. G = -S d. T + V dp kinetics: lnk depends linearly on the pressure lnk (or logk) vs p gives a straight line with V as slope assuming that V is pressure independent 09. 2021 what does this tell us with respect to mechanism ? CHE 323 -FS 18 -T 2 -3

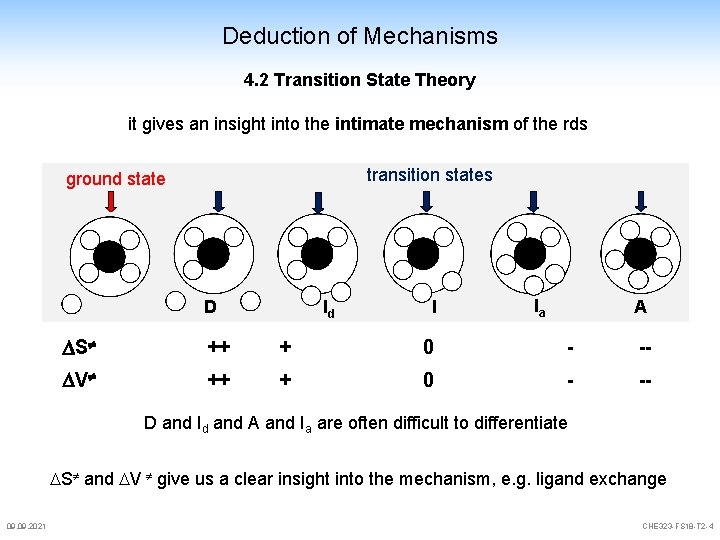
Deduction of Mechanisms 4. 2 Transition State Theory it gives an insight into the intimate mechanism of the rds transition states ground state D Id I Ia A S ++ + 0 - -- V ++ + 0 - -- D and Id and A and Ia are often difficult to differentiate S and V give us a clear insight into the mechanism, e. g. ligand exchange 09. 2021 CHE 323 -FS 18 -T 2 -4

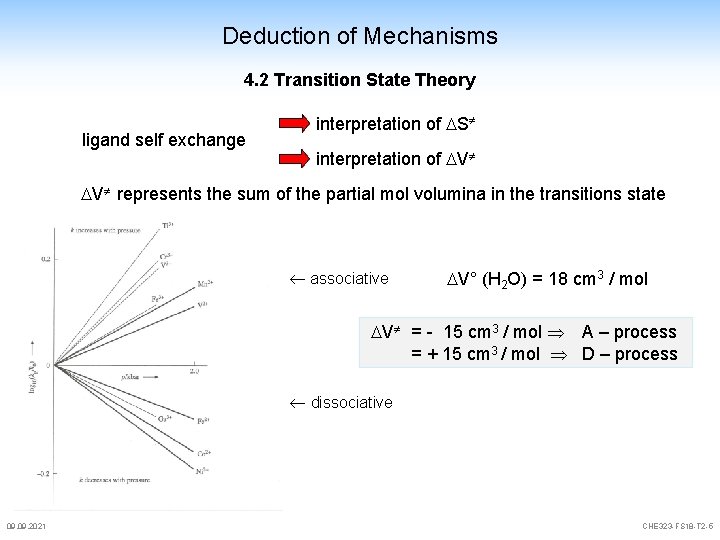
Deduction of Mechanisms 4. 2 Transition State Theory ligand self exchange interpretation of S interpretation of V V represents the sum of the partial mol volumina in the transitions state associative V° (H 2 O) = 18 cm 3 / mol V = - 15 cm 3 / mol A – process = + 15 cm 3 / mol D – process dissociative 09. 2021 CHE 323 -FS 18 -T 2 -5
![Deduction of Mechanisms 4 2 Transition State Theory MOH 26n kex OH 2 Deduction of Mechanisms 4. 2 Transition State Theory [M(OH 2)6]n+ kex + OH 2](https://slidetodoc.com/presentation_image_h2/9811de8cd2ad3f5e5ddcb8c14471f64d/image-6.jpg)
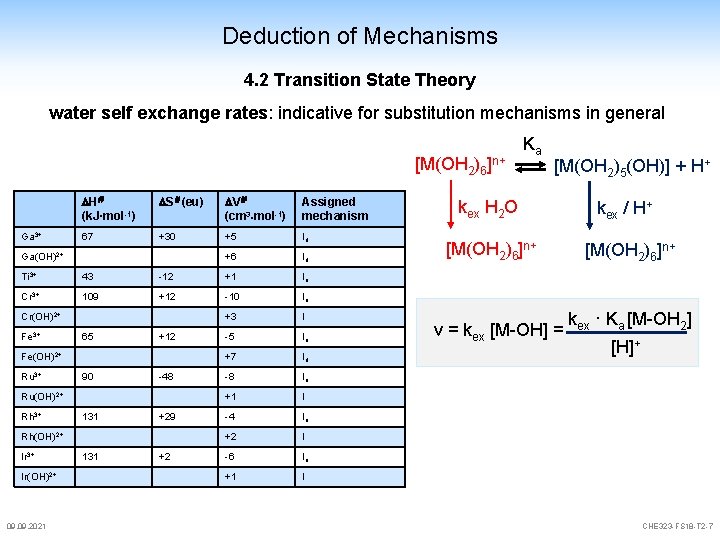
Deduction of Mechanisms 4. 2 Transition State Theory [M(OH 2)6]n+ kex + OH 2 [M(OH 2)5(OH 2)]n+ + H 2 O kex covers an extremely broad range for D processes k = kex „no reaction" reaction Go = O 09. 2021 CHE 323 -FS 18 -T 2 -6

Deduction of Mechanisms 4. 2 Transition State Theory water self exchange rates: indicative for substitution mechanisms in general [M(OH 2)6]n+ Ga 3+ H (k. J·mol-1) S (eu) V (cm 3 mol-1) Assigned mechanism 67 +30 +5 Id +6 Id Ga(OH)2+ Ti 3+ 43 -12 +1 Ia Cr 3+ 109 +12 -10 Ia +3 I -5 Ia +7 Id -8 Ia +1 I -4 Ia +2 I -6 Ia +1 I Cr(OH)2+ Fe 3+ 65 +12 Fe(OH)2+ Ru 3+ 90 -48 Ru(OH)2+ Rh 3+ 131 +29 Rh(OH)2+ Ir 3+ Ir(OH)2+ 09. 2021 131 +2 Ka [M(OH 2)5(OH)] + H+ kex H 2 O kex / H+ [M(OH 2)6]n+ v = kex [M-OH] = kex · Ka [M-OH 2] [H]+ CHE 323 -FS 18 -T 2 -7

Deduction of Mechanisms 4. 2 Transition State Theory back to data interpretation: most reactions are not one step… let's look at a p. H dependent scheme AH+ Ka H+ + A A+B k 2 P p. m. the composed rate constant is: k 2·KA applying TST and van't Hoff 09. 2021 CHE 323 -FS 18 -T 2 -8

Deduction of Mechanisms 4. 2 Transition State Theory back to data interpretation: most reactions are not one step… linearization: we do not get H but - H we must know pre-equilibrium (Ka in this case) to get the "true" H Question: find a situation for which G is negative e. g. the reaction becomes slower with increasing temperature steady state System: k-3 >> k 4 09. 2021 A+B k 3 k-3 I I k 4 P then: CHE 323 -FS 18 -T 2 -9

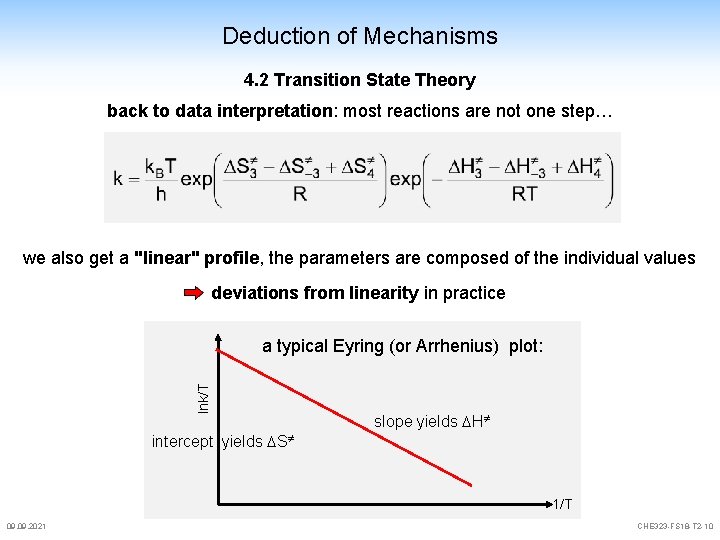
Deduction of Mechanisms 4. 2 Transition State Theory back to data interpretation: most reactions are not one step… we also get a "linear" profile, the parameters are composed of the individual values deviations from linearity in practice lnk/T a typical Eyring (or Arrhenius) plot: slope yields H intercept yields S 1/T 09. 2021 CHE 323 -FS 18 -T 2 -10

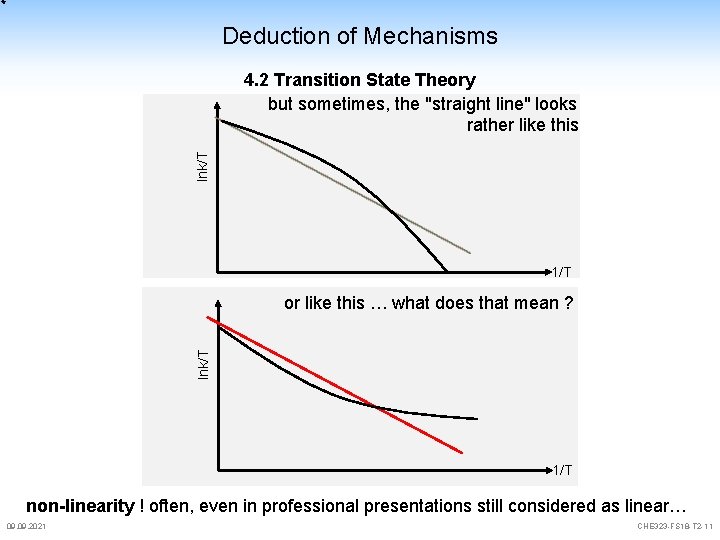
Deduction of Mechanisms lnk/T 4. 2 Transition State Theory but sometimes, the "straight line" looks rather like this 1/T lnk/T or like this … what does that mean ? 1/T non-linearity ! often, even in professional presentations still considered as linear… 09. 2021 CHE 323 -FS 18 -T 2 -11

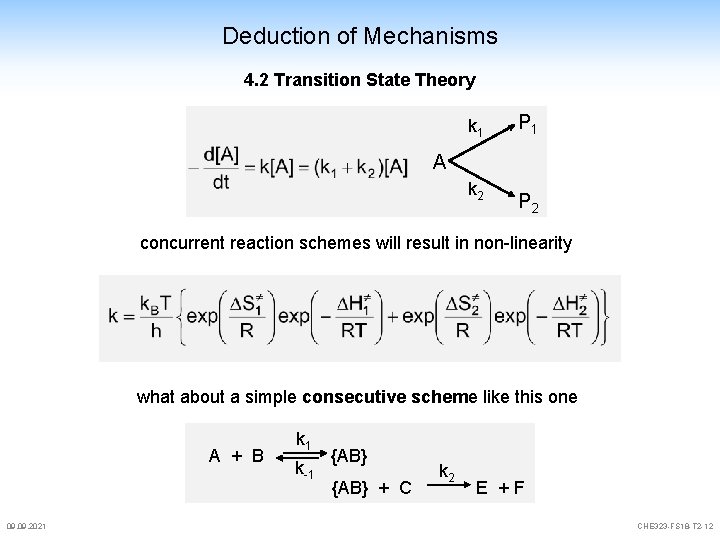
Deduction of Mechanisms 4. 2 Transition State Theory k 1 P 1 A k 2 P 2 concurrent reaction schemes will result in non-linearity what about a simple consecutive scheme like this one A + B 09. 2021 k 1 k-1 {AB} + C k 2 E +F CHE 323 -FS 18 -T 2 -12

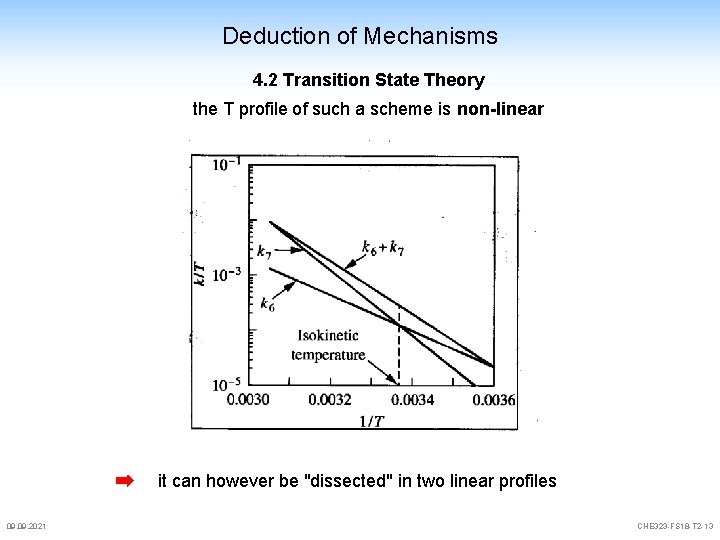
Deduction of Mechanisms 4. 2 Transition State Theory the T profile of such a scheme is non-linear it can however be "dissected" in two linear profiles 09. 2021 CHE 323 -FS 18 -T 2 -13

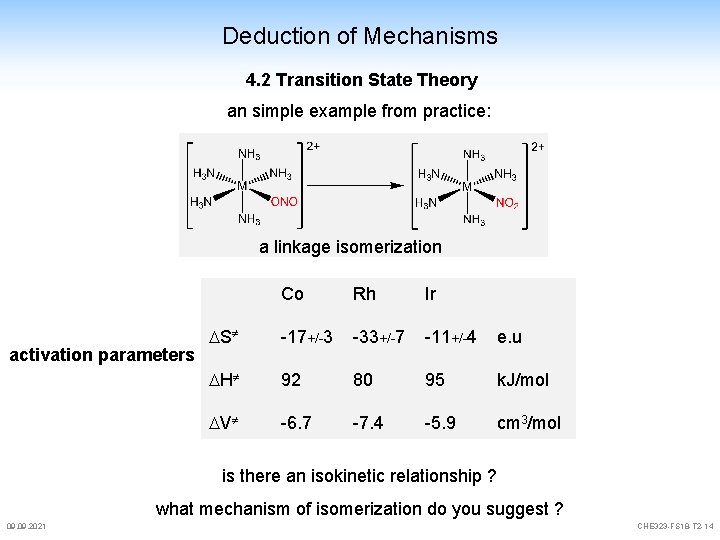
Deduction of Mechanisms 4. 2 Transition State Theory an simple example from practice: a linkage isomerization activation parameters Co Rh Ir S -17+/-3 -33+/-7 -11+/-4 e. u H 92 80 95 k. J/mol V -6. 7 -7. 4 -5. 9 cm 3/mol is there an isokinetic relationship ? what mechanism of isomerization do you suggest ? 09. 2021 CHE 323 -FS 18 -T 2 -14

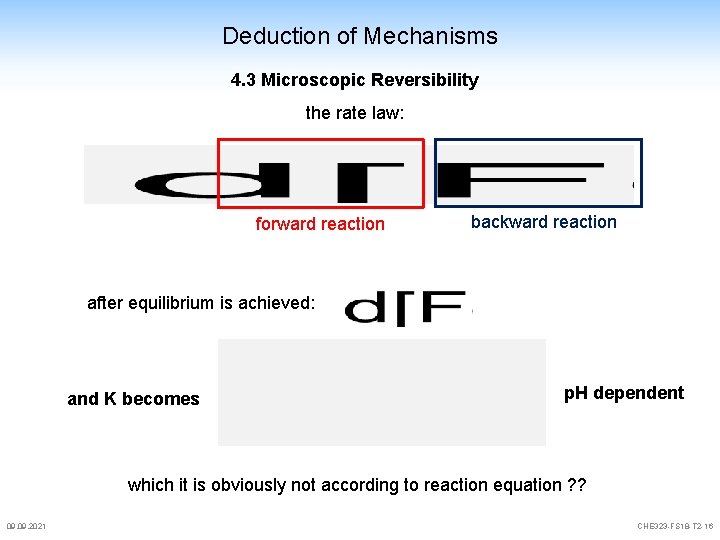
Deduction of Mechanisms 4. 3 Microscopic Reversibility The reaction mechanisms for the forward and the backward reaction are identical, they are mirror images of each other This principle is always valid, even when multiple steps, equilibria or branched reactions are involved if it were not valid, we would run into problems with thermodynamics let's look at the simple reaction [Fe(NCS)(OH 2)5]2+ + H 2 O [Fe(OH 2)6]3+ + NCS- is p. H dependent and can be described with: A 09. 2021 k 1 k-1 I I k 2 k-2 P CHE 323 -FS 18 -T 2 -15

Deduction of Mechanisms 4. 3 Microscopic Reversibility the rate law: forward reaction backward reaction after equilibrium is achieved: and K becomes p. H dependent which it is obviously not according to reaction equation ? ? 09. 2021 CHE 323 -FS 18 -T 2 -16

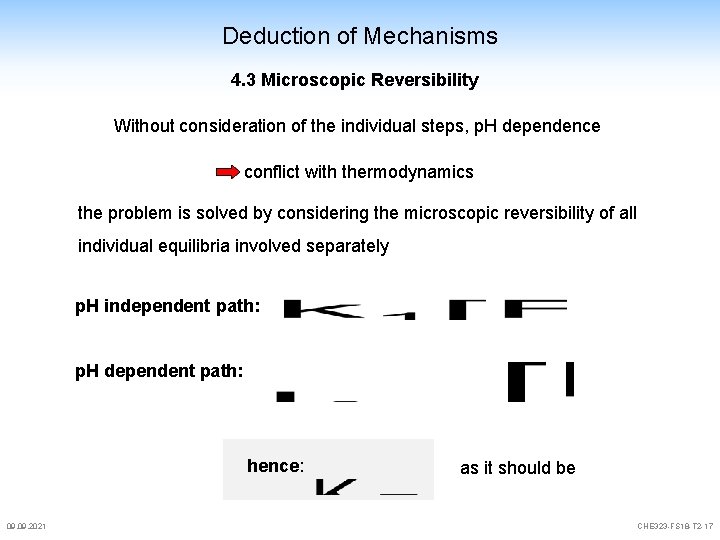
Deduction of Mechanisms 4. 3 Microscopic Reversibility Without consideration of the individual steps, p. H dependence conflict with thermodynamics the problem is solved by considering the microscopic reversibility of all individual equilibria involved separately p. H independent path: p. H dependent path: hence: 09. 2021 as it should be CHE 323 -FS 18 -T 2 -17

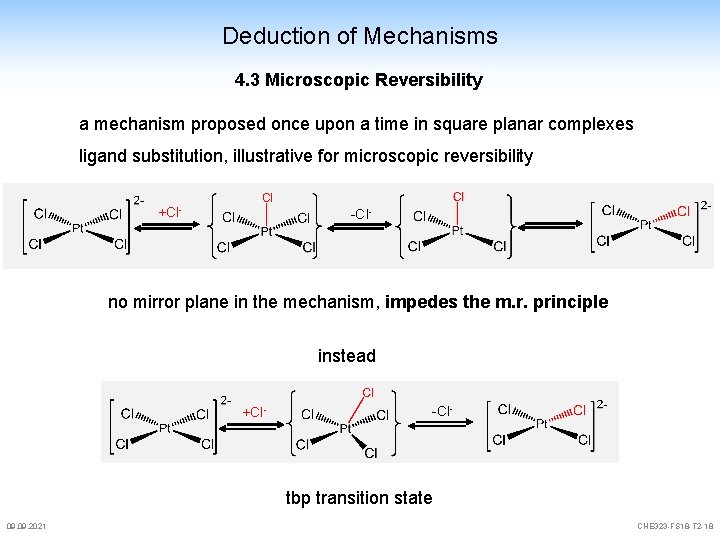
Deduction of Mechanisms 4. 3 Microscopic Reversibility a mechanism proposed once upon a time in square planar complexes ligand substitution, illustrative for microscopic reversibility +Cl- -Cl- no mirror plane in the mechanism, impedes the m. r. principle instead +Cl- -Cl- tbp transition state 09. 2021 CHE 323 -FS 18 -T 2 -18

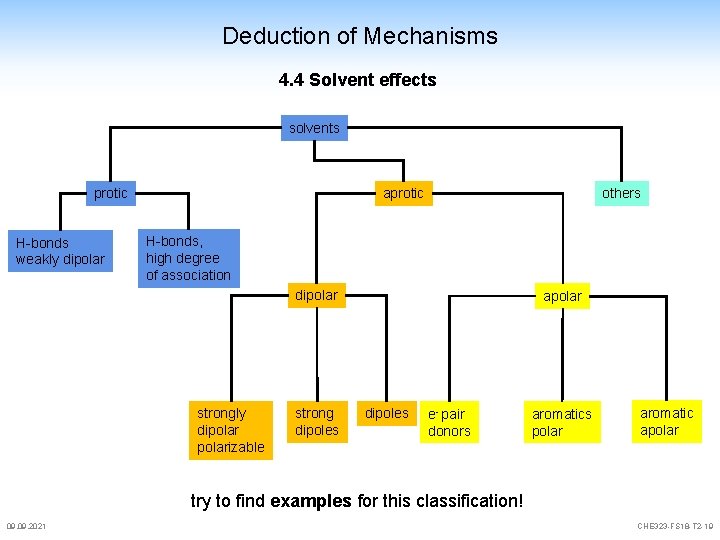
Deduction of Mechanisms 4. 4 Solvent effects solvents protic H-bonds weakly dipolar aprotic others H-bonds, high degree of association dipolar strongly dipolarizable strong dipoles apolar dipoles e- pair donors aromatics polar aromatic apolar try to find examples for this classification! 09. 2021 CHE 323 -FS 18 -T 2 -19

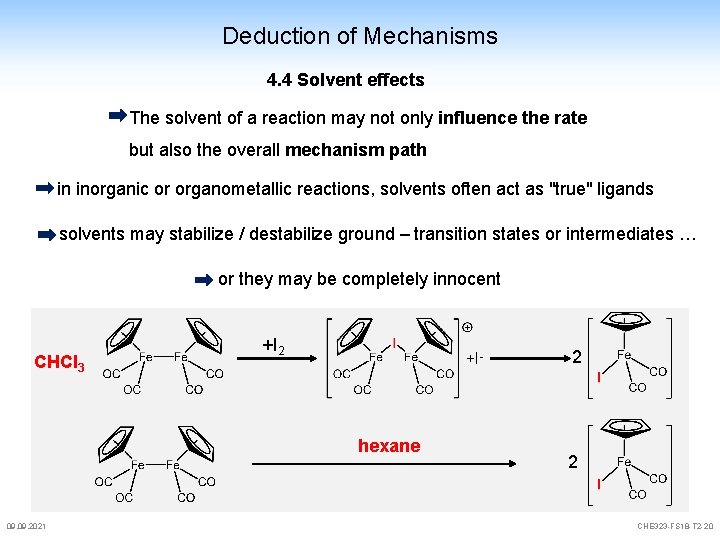
Deduction of Mechanisms 4. 4 Solvent effects The solvent of a reaction may not only influence the rate but also the overall mechanism path in inorganic or organometallic reactions, solvents often act as "true" ligands solvents may stabilize / destabilize ground – transition states or intermediates … or they may be completely innocent CHCl 3 +I 2 +I- hexane 09. 2021 2 2 CHE 323 -FS 18 -T 2 -20

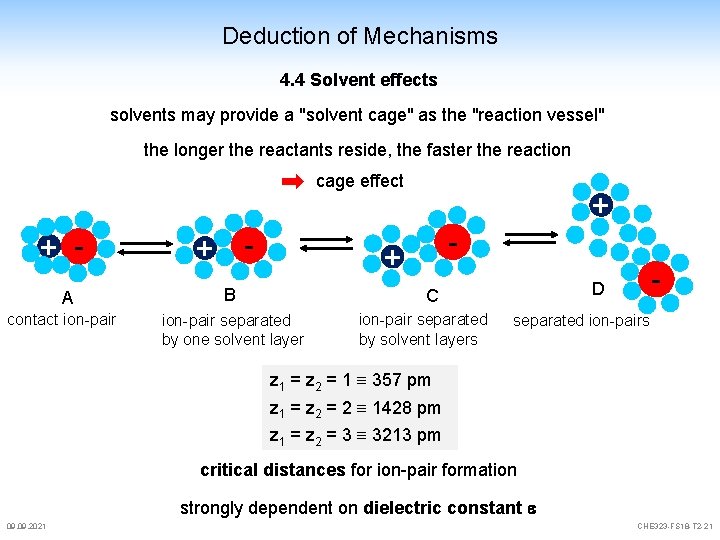
Deduction of Mechanisms 4. 4 Solvent effects solvents may provide a "solvent cage" as the "reaction vessel" the longer the reactants reside, the faster the reaction cage effect + A contact ion-pair - + + - + B ion-pair separated by one solvent layer ion-pair separated by solvent layers - D C separated ion-pairs z 1 = z 2 = 1 357 pm z 1 = z 2 = 2 1428 pm z 1 = z 2 = 3 3213 pm critical distances for ion-pair formation strongly dependent on dielectric constant e 09. 2021 CHE 323 -FS 18 -T 2 -21

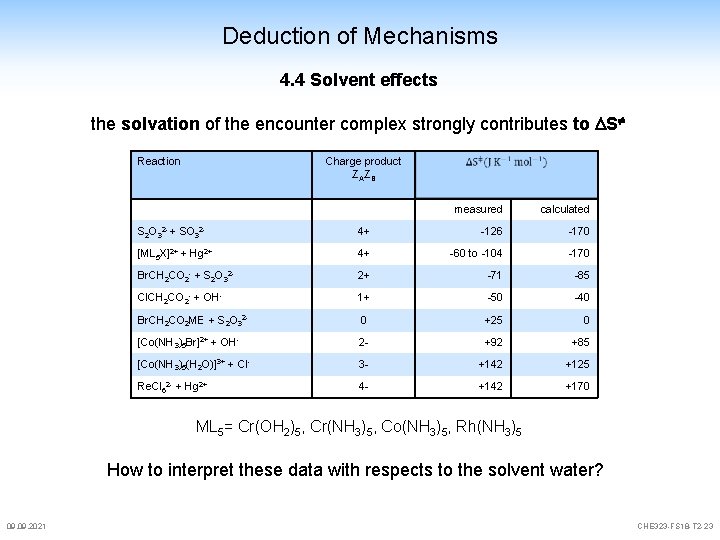
Deduction of Mechanisms 4. 4 Solvent effects residence time in the cage given by the rate of diffusion in water, typically 10 -12 bis 10 -11 sec time to collide 10 – 100 times consequence: short lived intermediates can not be detected since the immediately react further the entity of compounds in a cage is called "encounter complex" the solvation of the encounter complex strongly contributes to S 09. 2021 Reaction Reactands activated compound SN 1 SN 2 R—X+ Y + R—X Y- + R—X Y + R—X+ Y- + R—X+ Rd+·····Xd+ Yd+·····R······Xd. Yd-······R······Xd+ increased polarity ++ ++ -CHE 323 -FS 18 -T 2 -22

Deduction of Mechanisms 4. 4 Solvent effects the solvation of the encounter complex strongly contributes to S Reaction Charge product ZAZB measured calculated S 2 O 32 - + SO 32 - 4+ -126 -170 [ML 5 X]2+ + Hg 2+ 4+ -60 to -104 -170 Br. CH 2 CO 2 - + S 2 O 32 - 2+ -71 -85 Cl. CH 2 CO 2 - + OH- 1+ -50 -40 Br. CH 2 CO 2 ME + S 2 O 32 - 0 +25 0 [Co(NH 3)5 Br]2+ + OH- 2 - +92 +85 [Co(NH 3)5(H 2 O)]3+ + Cl- 3 - +142 +125 Re. Cl 62 - + Hg 2+ 4 - +142 +170 ML 5= Cr(OH 2)5, Cr(NH 3)5, Co(NH 3)5, Rh(NH 3)5 How to interpret these data with respects to the solvent water? 09. 2021 CHE 323 -FS 18 -T 2 -23

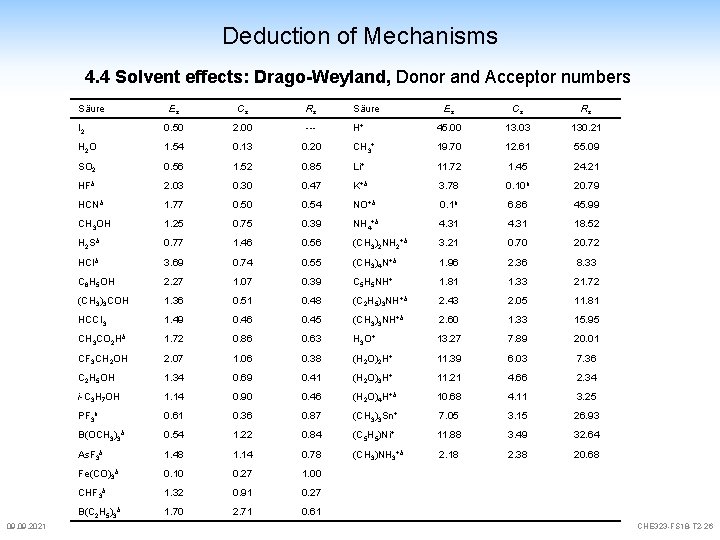
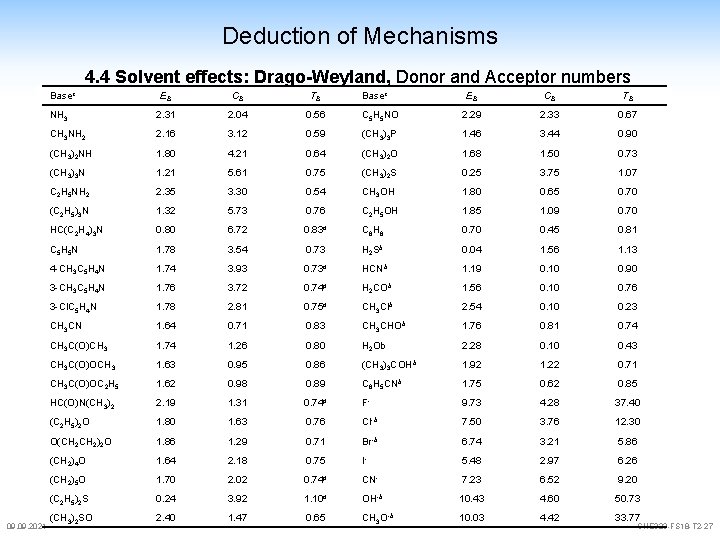
Deduction of Mechanisms 4. 4 Solvent effects donor/acceptor numbers are an indication for the behaviour of solvents Gutmann: B + Sb. Cl 5 B-Sb. Cl 5 DZ(Sb. Cl 5) = -DH useful to compare relative characters of solvents no obvious relationship to e Drago: concept doesn't consider hard/soft (electrostatic – covalent) new definition Drago und Weyland: -D H = E a E b + C a C b E = electrostatic, C = covalent a=acid, b=base 09. 2021 CHE 323 -FS 18 -T 2 -24

Deduction of Mechanisms 4. 4 Solvent effects new definition Drago und Weyland: I 2 + Pyridin -D H = E a E b + C a C b I 2·C 5 H 5 N -DHber = (0. 5 · 1. 78) + (2. 0 · 3. 54) = 7. 97 kcal/mol (-DHexp = 7. 8 kcal/mol) "acidic" solvents which tend to interact electrostatically (large Es) interact preferentially with bases with large Eb and acids which prefer covalent interaction (large Ca) interact with bases of large Cb 09. 2021 CHE 323 -FS 18 -T 2 -25

Deduction of Mechanisms 4. 4 Solvent effects: Drago-Weyland, Donor and Acceptor numbers Säure 09. 2021 Es Cs Rs Säure Es Cs Rs I 2 0. 50 2. 00 --- H+ 45. 00 13. 03 130. 21 H 2 O 1. 54 0. 13 0. 20 CH 3+ 19. 70 12. 61 55. 09 SO 2 0. 56 1. 52 0. 85 Li+ 11. 72 1. 45 24. 21 HFb 2. 03 0. 30 0. 47 K+b 3. 78 0. 10 b 20. 79 HCNb 1. 77 0. 50 0. 54 NO+b 0. 1 b 6. 86 45. 99 CH 3 OH 1. 25 0. 75 0. 39 NH 4+b 4. 31 18. 52 H 2 Sb 0. 77 1. 46 0. 56 (CH 3)2 NH 2+b 3. 21 0. 70 20. 72 HCIb 3. 69 0. 74 0. 55 (CH 3)4 N+b 1. 96 2. 36 8. 33 C 6 H 5 OH 2. 27 1. 07 0. 39 C 5 H 5 NH+ 1. 81 1. 33 21. 72 (CH 3)3 COH 1. 36 0. 51 0. 48 (C 2 H 5)3 NH+b 2. 43 2. 05 11. 81 HCCI 3 1. 49 0. 46 0. 45 (CH 3)3 NH+b 2. 60 1. 33 15. 95 CH 3 CO 2 Hb 1. 72 0. 86 0. 63 H 3 O+ 13. 27 7. 89 20. 01 CF 3 CH 2 OH 2. 07 1. 06 0. 38 (H 2 O)2 H+ 11. 39 6. 03 7. 36 C 2 H 5 OH 1. 34 0. 69 0. 41 (H 2 O)3 H+ 11. 21 4. 66 2. 34 i-C 3 H 7 OH 1. 14 0. 90 0. 46 (H 2 O)4 H+b 10. 68 4. 11 3. 25 PF 3 b 0. 61 0. 36 0. 87 (CH 3)3 Sn+ 7. 05 3. 15 26. 93 B(OCH 3)3 b 0. 54 1. 22 0. 84 (C 5 H 5)Ni+ 11. 88 3. 49 32. 64 As. F 3 b 1. 48 1. 14 0. 78 (CH 3)NH 3+b 2. 18 2. 38 20. 68 Fe(CO)3 b 0. 10 0. 27 1. 00 CHF 3 b 1. 32 0. 91 0. 27 B(C 2 H 5)3 b 1. 70 2. 71 0. 61 CHE 323 -FS 18 -T 2 -26

Deduction of Mechanisms 4. 4 Solvent effects: Drago-Weyland, Donor and Acceptor numbers Basec 09. 2021 EB CB TB NH 3 2. 31 2. 04 0. 56 CH 3 NH 2 2. 16 3. 12 (CH 3)2 NH 1. 80 (CH 3)3 N Basec EB CB TB C 5 H 5 NO 2. 29 2. 33 0. 67 0. 59 (CH 3)3 P 1. 46 3. 44 0. 90 4. 21 0. 64 (CH 3)2 O 1. 68 1. 50 0. 73 1. 21 5. 61 0. 75 (CH 3)2 S 0. 25 3. 75 1. 07 C 2 H 5 NH 2 2. 35 3. 30 0. 54 CH 3 OH 1. 80 0. 65 0. 70 (C 2 H 5)3 N 1. 32 5. 73 0. 76 C 2 H 5 OH 1. 85 1. 09 0. 70 HC(C 2 H 4)3 N 0. 80 6. 72 0. 83 d C 6 H 6 0. 70 0. 45 0. 81 C 5 H 5 N 1. 78 3. 54 0. 73 H 2 Sb 0. 04 1. 56 1. 13 4 -CH 3 C 5 H 4 N 1. 74 3. 93 0. 73 d HCNb 1. 19 0. 10 0. 90 3 -CH 3 C 5 H 4 N 1. 76 3. 72 0. 74 d H 2 COb 1. 56 0. 10 0. 76 3 -Cl. C 5 H 4 N 1. 78 2. 81 0. 75 d CH 3 Clb 2. 54 0. 10 0. 23 CH 3 CN 1. 64 0. 71 0. 83 CH 3 CHOb 1. 76 0. 81 0. 74 CH 3 C(O)CH 3 1. 74 1. 26 0. 80 H 2 Ob 2. 28 0. 10 0. 43 CH 3 C(O)OCH 3 1. 63 0. 95 0. 86 (CH 3)3 COHb 1. 92 1. 22 0. 71 CH 3 C(O)OC 2 H 5 1. 62 0. 98 0. 89 C 6 H 5 CNb 1. 75 0. 62 0. 85 HC(O)N(CH 3)2 2. 19 1. 31 0. 74 d F- 9. 73 4. 28 37. 40 (C 2 H 5)2 O 1. 80 1. 63 0. 76 Cl-b 7. 50 3. 76 12. 30 O(CH 2)2 O 1. 86 1. 29 0. 71 Br-b 6. 74 3. 21 5. 86 (CH 2)4 O 1. 64 2. 18 0. 75 I- 5. 48 2. 97 6. 26 (CH 2)5 O 1. 70 2. 02 0. 74 d CN- 7. 23 6. 52 9. 20 (C 2 H 5)2 S 0. 24 3. 92 1. 10 d OH-b 10. 43 4. 60 50. 73 (CH 3)2 SO 2. 40 1. 47 0. 65 CH 3 O-b 10. 03 4. 42 33. 77 CHE 323 -FS 18 -T 2 -27

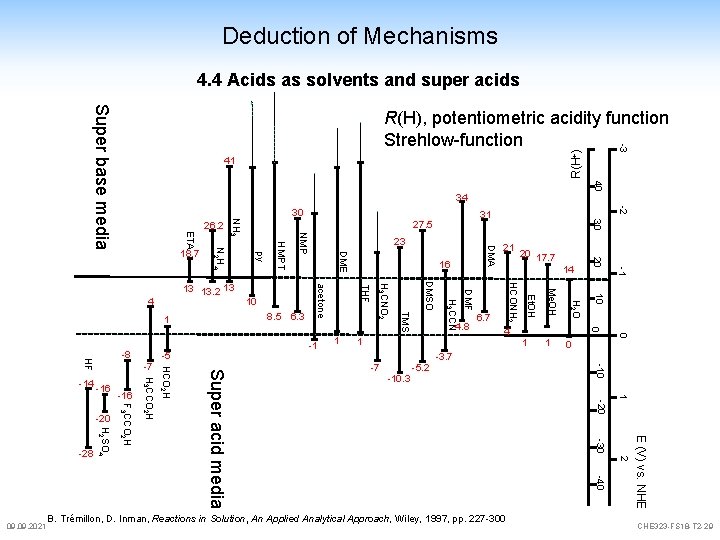
Deduction of Mechanisms 4. 4 Acids as solvents and super acids most Brønsted acids show auto dissociation, like water p. H-scale and value defined by the degree of auto dissociation therefore, each acid has its individual p. H-scale all acid/base pairs which are inside these limits act as electrolyte all acid/base pairs outside these limits will be levelled by the acid solvent CH 3 COOH is an acid in water and an electrolyte CH 3 COOH in H 2 SO 4 is a base, base strength levelled by [HSO 4]more practical: dissolution of amide H 2 N- is levelled by base strength of [OH]since outside the p. H scale of water 09. 2021 CHE 323 -FS 18 -T 2 -28

Deduction of Mechanisms 4. 4 Acids as solvents and super acids Super base media -2 -1 0 0 H 2 O 1 10 Me. OH 1 0 4 Et. OH 6. 7 20 20 17. 7 14 HCONH 2 4. 8 1 21 DMF H 3 CCN DMSO TMS H 3 CNO 2 THF acetone 1 DMA DME 16 30 NMP HMPT -3. 7 -5. 2 -7 -10. 3 1 -20 2 -40 E (V) vs. NHE -30 B. Trémillon, D. Inman, Reactions in Solution, An Applied Analytical Approach, Wiley, 1997, pp. 227 -300 -10 H 3 CCO 2 H F 3 CCO 2 H 09. 2021 H 2 SO 4 -28 -16 23 -1 Super acid media -7 10 8. 5 6. 3 -5 31 27. 5 py 13 13. 2 13 HCO 2 H HF -8 30 NH 3 N 2 H 4 ETA 18. 7 1 -20 40 34 4 -16 R(H+) 41 26. 2 -3 R(H), potentiometric acidity function Strehlow-function CHE 323 -FS 18 -T 2 -29

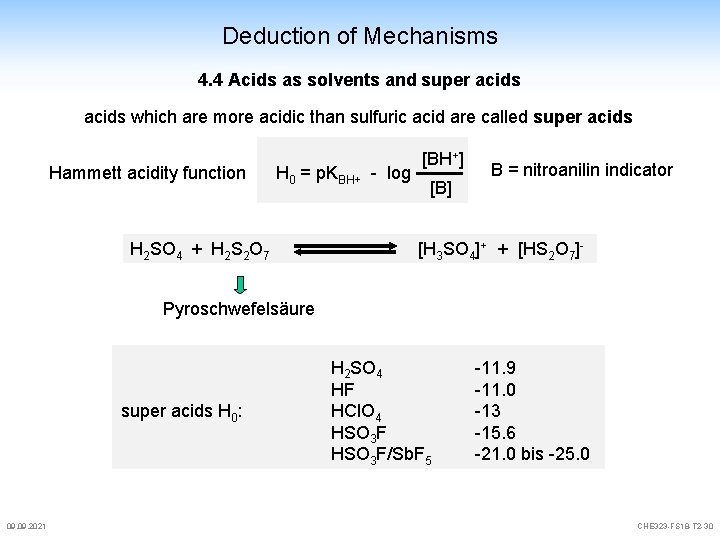
Deduction of Mechanisms 4. 4 Acids as solvents and super acids which are more acidic than sulfuric acid are called super acids Hammett acidity function H 0 = p. KBH+ - log H 2 SO 4 + H 2 S 2 O 7 [BH+] [B] B = nitroanilin indicator [H 3 SO 4]+ + [HS 2 O 7]- Pyroschwefelsäure super acids H 0: 09. 2021 H 2 SO 4 HF HCl. O 4 HSO 3 F/Sb. F 5 -11. 9 -11. 0 -13 -15. 6 -21. 0 bis -25. 0 CHE 323 -FS 18 -T 2 -30

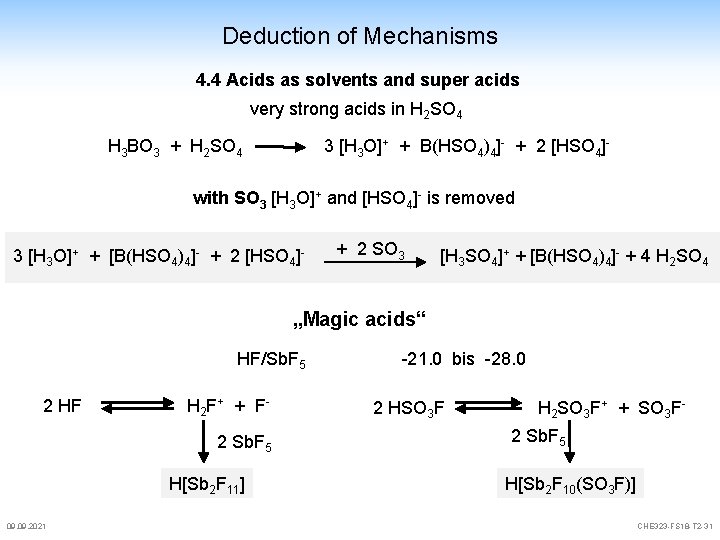
Deduction of Mechanisms 4. 4 Acids as solvents and super acids very strong acids in H 2 SO 4 H 3 BO 3 + H 2 SO 4 3 [H 3 O]+ + B(HSO 4)4]- + 2 [HSO 4]- with SO 3 [H 3 O]+ and [HSO 4]- is removed 3 [H 3 O]+ + [B(HSO 4)4]- + 2 [HSO 4]- + 2 SO 3 [H 3 SO 4]+ + [B(HSO 4)4]- + 4 H 2 SO 4 „Magic acids“ HF/Sb. F 5 2 HF H 2 F + + F 2 Sb. F 5 H[Sb 2 F 11] 09. 2021 -21. 0 bis -28. 0 2 HSO 3 F H 2 SO 3 F+ + SO 3 F 2 Sb. F 5 H[Sb 2 F 10(SO 3 F)] CHE 323 -FS 18 -T 2 -31

Deduction of Mechanisms 4. 4 Acids as solvents and super acids - the strongest known acids are based on Carborane anions the conjugate base of the super acid may not coordinate or oxidize need for delocalization over many atoms principle behind synthesis: Brønsted acidity induced by Lewis acidity H-LB + LS-A LS-LB + H-A a classical hard-soft problem extrem starke LS: Silylkationen {R 3 Si}+ starke LB: Cl- A: extrem schwaches Nucleophil e. g. Ch. Reed, Angew. Chem. Int. Ed. 2004, 43, 5352 09. 2021 CHE 323 -FS 18 -T 2 -32

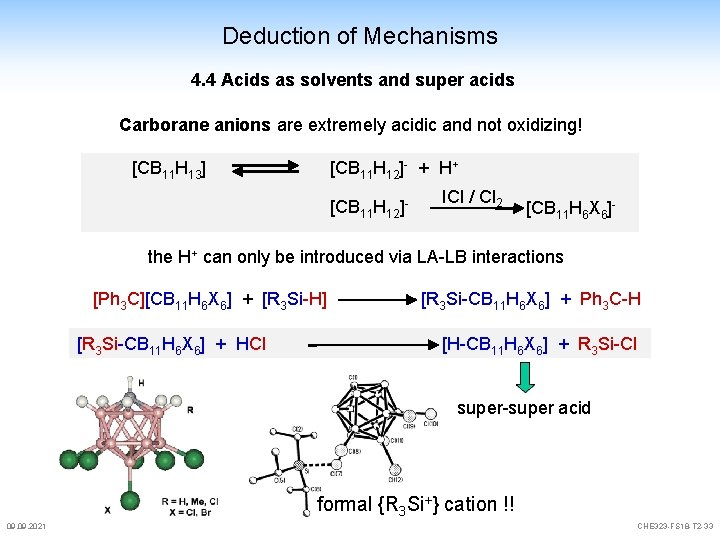
Deduction of Mechanisms 4. 4 Acids as solvents and super acids Carborane anions are extremely acidic and not oxidizing! [CB 11 H 13] [CB 11 H 12]- + H+ [CB 11 H 12]- ICl / Cl 2 [CB 11 H 6 X 6]- the H+ can only be introduced via LA-LB interactions [Ph 3 C][CB 11 H 6 X 6] + [R 3 Si-H] [R 3 Si-CB 11 H 6 X 6] + HCl [R 3 Si-CB 11 H 6 X 6] + Ph 3 C-H [H-CB 11 H 6 X 6] + R 3 Si-Cl super-super acid formal {R 3 Si+} cation !! 09. 2021 CHE 323 -FS 18 -T 2 -33

Deduction of Mechanisms 4. 4 Acids as solvents and super acids what can we do with super-super acids? Au. F 3 + 6 Xe + 3 H+ [Au. Xe 4]2+ + [Xe 2]2+ + 3 HF HF/Sb. F 5 mechanistic principle: super-super acid protonates Au. F 3 + 3 H+ [Au(FH)3]3+ very strong oxidant 2[Au(FH)3]3+ + 6 Xe [Au. Xe 4]2+ + [Xe 2]2+ + 3 HF [Sb 2 F 11]- is the counter-ion 09. 2021 S. Seidel, K. Seppelt, Science 2000, 290, 117. T. Drews, K. Seppelt, Angew. Chem. Int. Ed. 1997, 36, 273 CHE 323 -FS 18 -T 2 -34

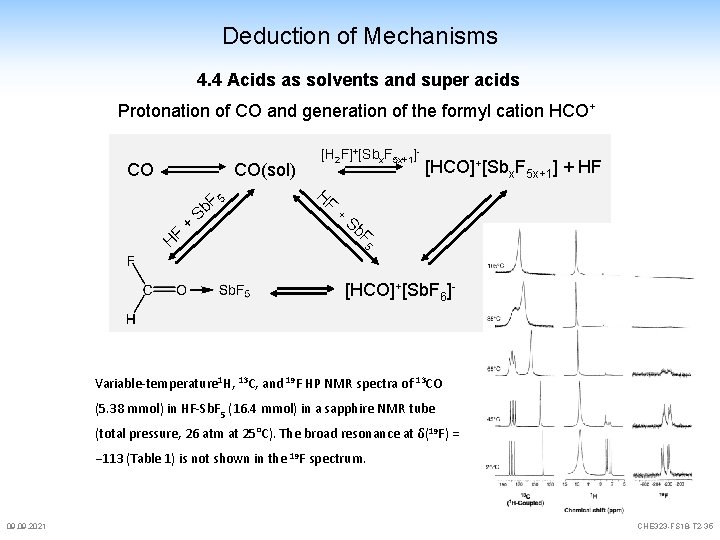
Deduction of Mechanisms 4. 4 Acids as solvents and super acids Protonation of CO and generation of the formyl cation HCO+ CO CO(sol) F H + S 5 b. F [H 2 F]+[Sbx. F 5 x+1]- HF + [HCO]+[Sbx. F 5 x+1] + HF Sb F 5 [HCO]+[Sb. F 6]- Variable-temperature 1 H, 13 C, and 19 F HP NMR spectra of 13 CO (5. 38 mmol) in HF-Sb. F 5 (16. 4 mmol) in a sapphire NMR tube (total pressure, 26 atm at 25°C). The broad resonance at δ(19 F) = − 113 (Table 1) is not shown in the 19 F spectrum. 09. 2021 CHE 323 -FS 18 -T 2 -35

Deduction of Mechanisms 4. 4 Acids as solvents and super acids Beside CO, super acids also protonate alkanes, but also small molecules like N 2, Ar a. o. fullerene [H-CB 11 H 6 X 6] + C 60 [CB 11 H 6 X 6][HC 60] benzene derivatives [H-CB 11 H 6 X 6] + C 6 H 6 [CB 11 H 6 X 6][C 6 H 7] 09. 2021 CHE 323 -FS 18 -T 2 -36

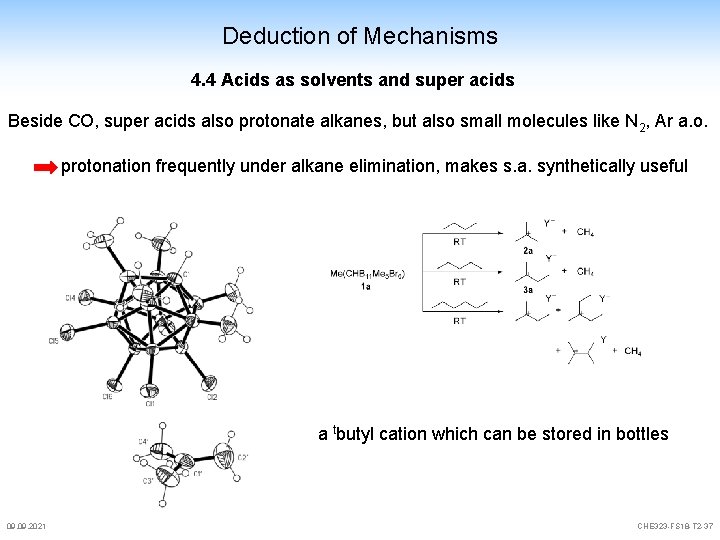
Deduction of Mechanisms 4. 4 Acids as solvents and super acids Beside CO, super acids also protonate alkanes, but also small molecules like N 2, Ar a. o. protonation frequently under alkane elimination, makes s. a. synthetically useful a tbutyl cation which can be stored in bottles 09. 2021 CHE 323 -FS 18 -T 2 -37

Deduction of Mechanisms 4. 5 Diffusion controlled reactions if the rate of a reaction is given by the collision probability, it runs at maximum speed. such a reaction runs at a diffusion controlled rate the frequency is defined by the Fick law of diffusion the relevant relationship is (Smoluchowski) r. AB is the reaction cross section, DAB = DA + DB the diffusion constant, a typical range for DAB is 2 10 -9 m 2 sec-1, for r. A 0. 2 nm or 2 10 -10 m depends on the viscosity of the solvent the resulting rate constant is kdc 6 109 M-1 sec-1 09. 2021 CHE 323 -FS 18 -T 2 -38

Deduction of Mechanisms 4. 5 Diffusion controlled reactions kdc does not depend significantly on the reaction partners a big molecule diffuses slower but may have a larger reaction cross section for other solvents, the diffusion constant can be calculated according to Einstein-Stokes Solvent /10 -4 kg m-1 s-1 kdc/L mol-1 s-1 n-Pentane Diethyl ether Acetone Benzene Water Acetic acid Benzonitrile Ethylene glycol Cyclohexanol Glycerol 2. 15 2. 22 3. 16 6. 03 8. 98 11. 6 14. 5 136 410 9450 3. 1· 1010 3. 0· 1010 2. 1· 1010 1. 1· 1010 7. 4· 109 5. 7· 109 4. 6· 109 4. 9· 108 1. 6· 108 7. 0· 106 resulting activation energy from kdc 09. 2021 k. B·T DA = 6·p·r. A· if v. A v. B mit = Viskosität Ea 4 - 20 k. J/mol CHE 323 -FS 18 -T 2 -39

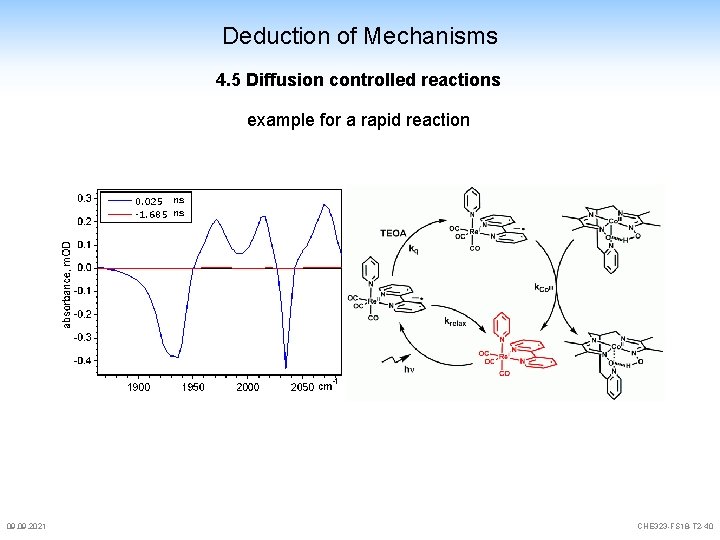
Deduction of Mechanisms 4. 5 Diffusion controlled reactions example for a rapid reaction 09. 2021 CHE 323 -FS 18 -T 2 -40

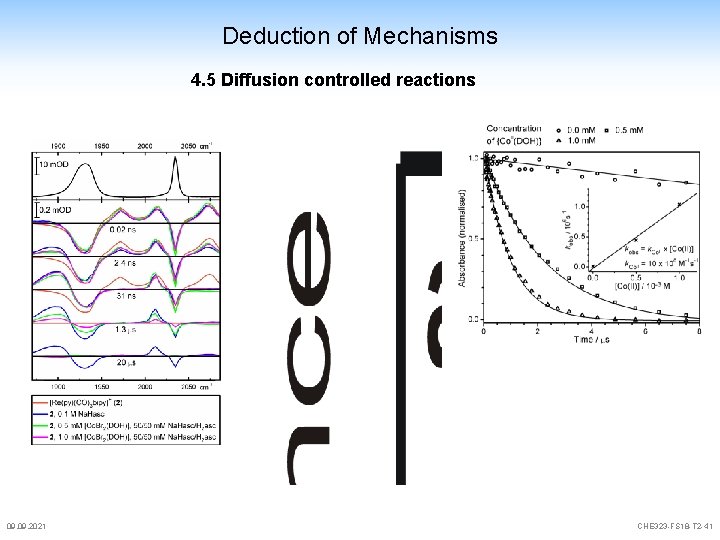
Deduction of Mechanisms 4. 5 Diffusion controlled reactions 09. 2021 CHE 323 -FS 18 -T 2 -41

Deduction of Mechanisms 4. 6 Kinetic Isotope Effect Exchanging isotopes is often a good hint to a reaction mechanism and the relevance of individual elementary steps If a reaction rate depends on the isotope (H/D), then this step is rate determining An isotopic effect is generally observable with H/D but is very difficult to detect with other isotopes e. g. 12 C/13 C vibrational energy of a molecule: En=(n+1/2) h harmonic oscillator: if force constants for R-H and R-D are equal: 09. 2021 CHE 323 -FS 18 -T 2 -42

Deduction of Mechanisms 4. 6 Kinetic Isotope Effect Principle of kie: Difference of activation energy is equal to the difference of ground state energies since R-H(D) bonds must be broken k A E or ln k. H = ln H AD R T D from Arrhenius: and for R = C follows that k. H/k. D = 7. 8 accordingly: 09. 2021 Bond H/cm-1 k. H/k. D C-H O-H N-H S-H 2900 3300 3100 2600 7. 8 10. 3 8. 9 6. 3 CHE 323 -FS 18 -T 2 -43

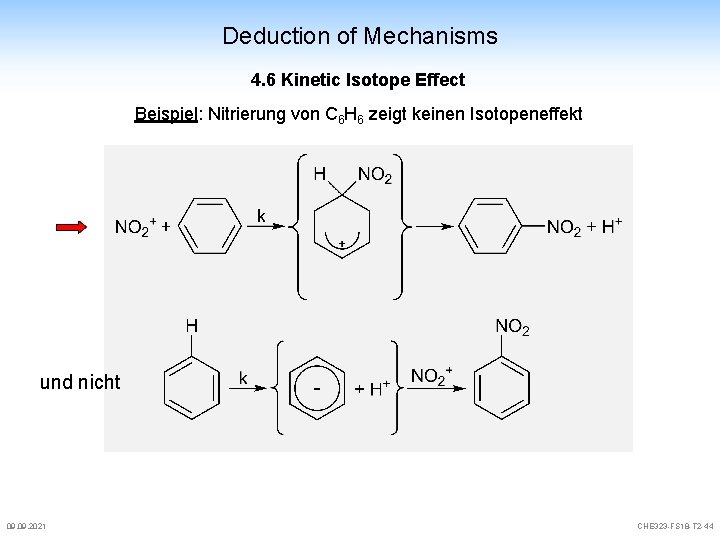
Deduction of Mechanisms 4. 6 Kinetic Isotope Effect Beispiel: Nitrierung von C 6 H 6 zeigt keinen Isotopeneffekt und nicht 09. 2021 CHE 323 -FS 18 -T 2 -44

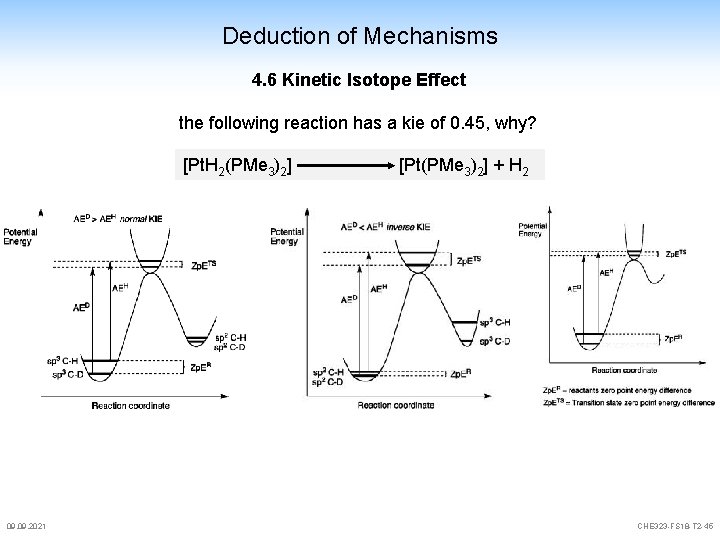
Deduction of Mechanisms 4. 6 Kinetic Isotope Effect the following reaction has a kie of 0. 45, why? [Pt. H 2(PMe 3)2] 09. 2021 [Pt(PMe 3)2] + H 2 CHE 323 -FS 18 -T 2 -45

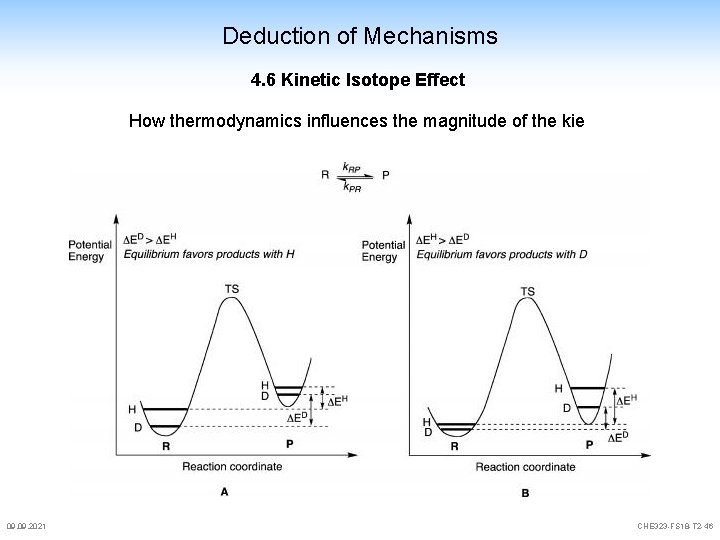
Deduction of Mechanisms 4. 6 Kinetic Isotope Effect How thermodynamics influences the magnitude of the kie 09. 2021 CHE 323 -FS 18 -T 2 -46

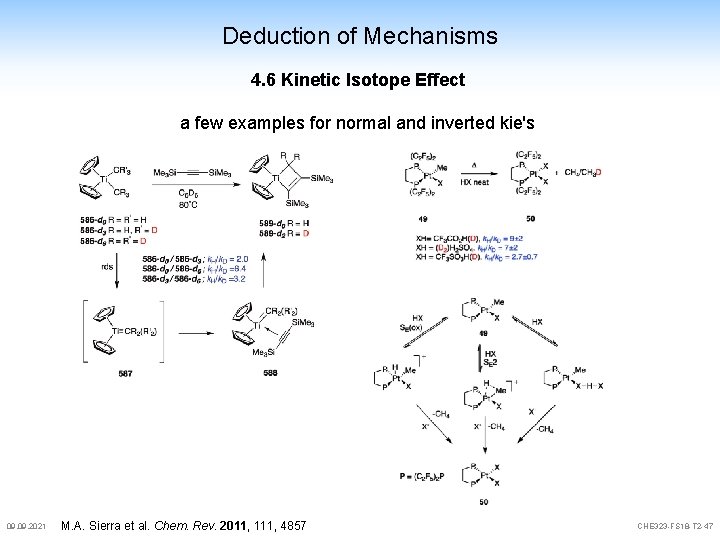
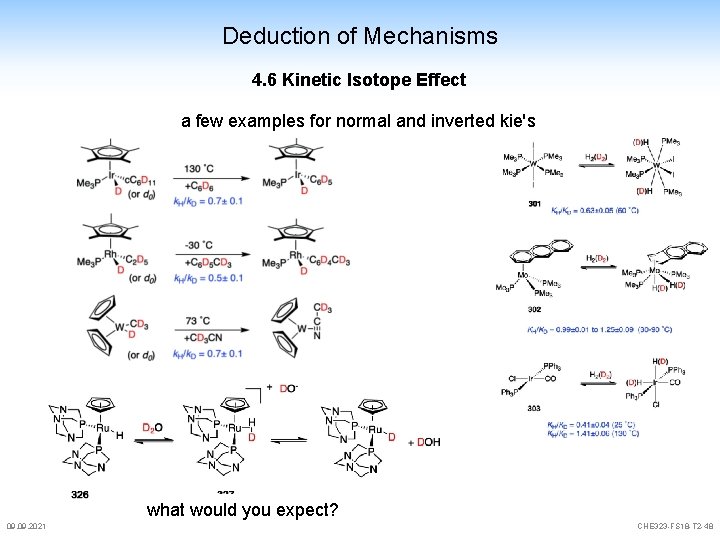
Deduction of Mechanisms 4. 6 Kinetic Isotope Effect a few examples for normal and inverted kie's 09. 2021 M. A. Sierra et al. Chem. Rev. 2011, 111, 4857 CHE 323 -FS 18 -T 2 -47

Deduction of Mechanisms 4. 6 Kinetic Isotope Effect a few examples for normal and inverted kie's what would you expect? 09. 2021 CHE 323 -FS 18 -T 2 -48

5. Linear Free Energy Relationships LFER Kinetic rate laws describe the time course of a reaction as a function of the elementary steps a relationship between kinetics and thermodynamics of a reaction is called a Linear Free Energy Relationship LFER are a semi quantitative way of “systematizing” our ideas about the similarity of reactions. G and G are correlated, although they have sometimes nothing to do with each other only sets of closely related reactions should be compared in that way 09. 2021 CHE 323 -FS 18 -T 2 -49

Linear Free Energy Relationships LFER only sets of closely related reactions should be compared in that way the most useful examples have well-defined domains of applicability, such as the Hammett equation, the Brønsted catalysis law and the Marcus equation (see later). an LFER is of the general form lnk = m ln. Kc + b since lnk = ln k T - G* h R T G 0 ln. Kc = - R T G* = m G 0 + b' whereas the parameters m and b' may tell us something about the mechanism 09. 2021 CHE 323 -FS 18 -T 2 -50

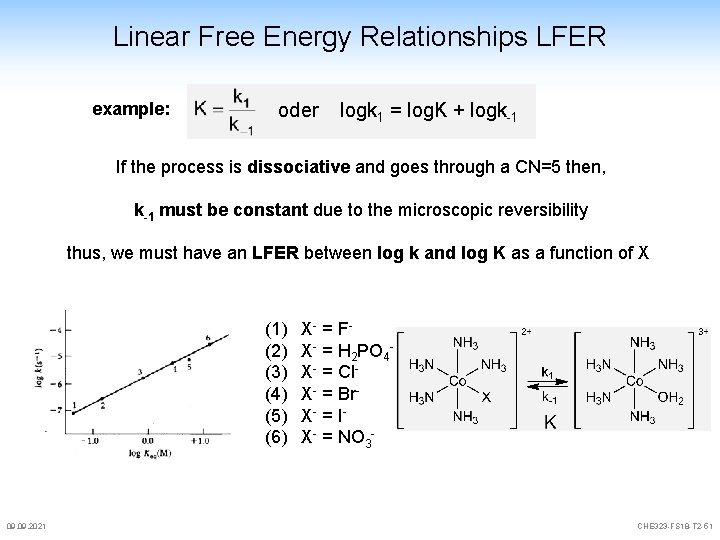
Linear Free Energy Relationships LFER example: oder logk 1 = log. K + logk-1 If the process is dissociative and goes through a CN=5 then, k-1 must be constant due to the microscopic reversibility thus, we must have an LFER between log k and log K as a function of X (1) (2) (3) (4) (5) (6) 09. 2021 X- = F X- = H 2 PO 4 X- = Cl. X- = Br. X- = I X- = NO 3 - CHE 323 -FS 18 -T 2 -51

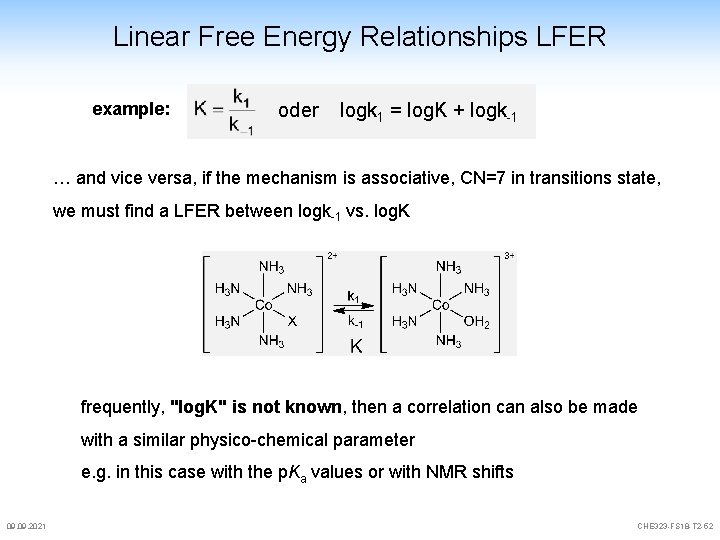
Linear Free Energy Relationships LFER example: oder logk 1 = log. K + logk-1 … and vice versa, if the mechanism is associative, CN=7 in transitions state, we must find a LFER between logk-1 vs. log. K frequently, "log. K" is not known, then a correlation can also be made with a similar physico-chemical parameter e. g. in this case with the p. Ka values or with NMR shifts 09. 2021 CHE 323 -FS 18 -T 2 -52

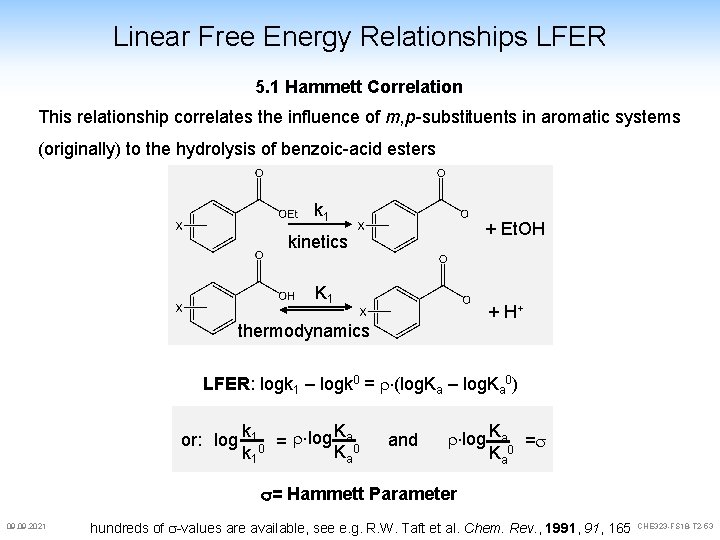
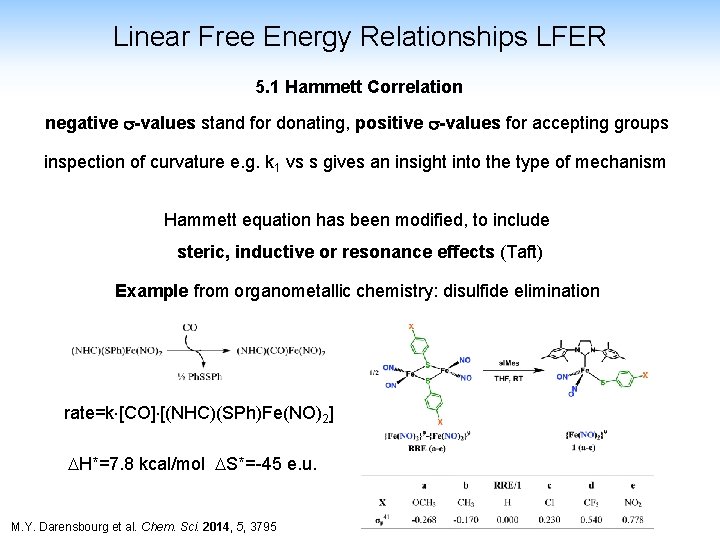
Linear Free Energy Relationships LFER 5. 1 Hammett Correlation This relationship correlates the influence of m, p-substituents in aromatic systems (originally) to the hydrolysis of benzoic-acid esters k 1 + Et. OH kinetics K 1 + H+ thermodynamics LFER: logk 1 – logk 0 = r (log. Ka – log. Ka 0) K or: log k 10 = r log a 0 Ka k 1 and r log Ka 0 =s Ka s= Hammett Parameter 09. 2021 hundreds of s-values are available, see e. g. R. W. Taft et al. Chem. Rev. , 1991, 165 CHE 323 -FS 18 -T 2 -53

Linear Free Energy Relationships LFER 5. 1 Hammett Correlation negative s-values stand for donating, positive s-values for accepting groups inspection of curvature e. g. k 1 vs s gives an insight into the type of mechanism Hammett equation has been modified, to include steric, inductive or resonance effects (Taft) Example from organometallic chemistry: disulfide elimination rate=k [CO] [(NHC)(SPh)Fe(NO)2] H*=7. 8 kcal/mol S*=-45 e. u. M. Y. Darensbourg et al. Chem. Sci. 2014, 5, 3795

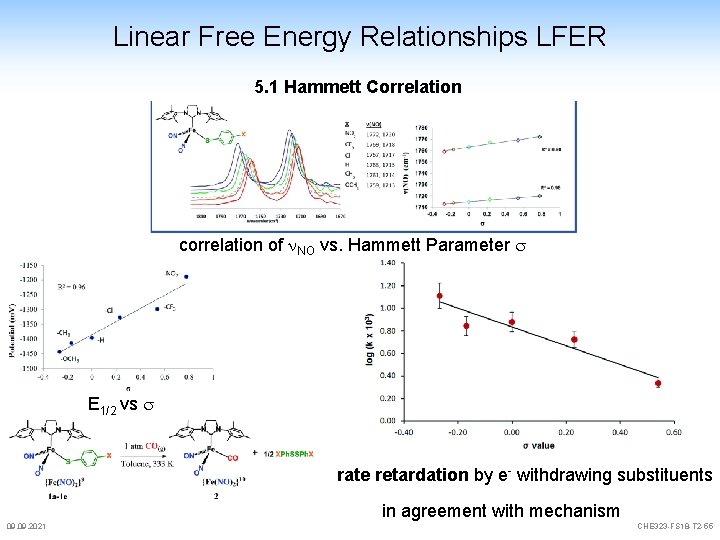
Linear Free Energy Relationships LFER 5. 1 Hammett Correlation correlation of NO vs. Hammett Parameter s E 1/2 vs s rate retardation by e- withdrawing substituents in agreement with mechanism 09. 2021 CHE 323 -FS 18 -T 2 -55

Linear Free Energy Relationships LFER 5. 2 Ligand Field Stabilization Energies Pro memoria: Classification of substitution mechanisms M-X M+ + Y - M-Y Hughes-Ingold: SN 1 - Langford-Gray: M-X + Y M -- X -- Y D M-Y + X Hughes-Ingold: SN 1 - Langford-Gray: Id / Ia M-X + Y Y -- M -- X M-Y + X Hughes-Ingold: SN 2 - Langford-Gray: A 09. 2021 CHE 323 -FS 18 -T 2 -56

Linear Free Energy Relationships LFER 5. 2 Ligand Field Stabilization Energies A or D mechanisms may show different LFER, the LFSE plays a role intimate mechanisms if in between must be elucidated from a series of experiments, e. g. with LFER relations nomenclature: 09. 2021 - dissociative activation Id influence of leaving group - associative activation Ia influence of entering group CHE 323 -FS 18 -T 2 -57

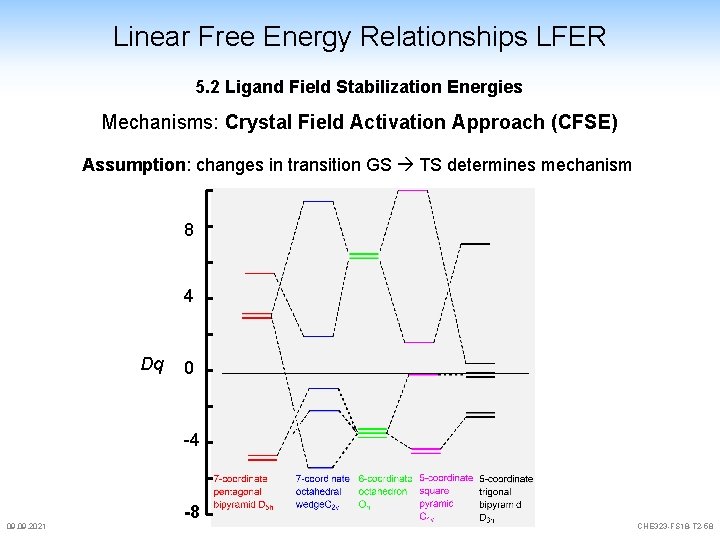
Linear Free Energy Relationships LFER 5. 2 Ligand Field Stabilization Energies Mechanisms: Crystal Field Activation Approach (CFSE) Assumption: changes in transition GS TS determines mechanism 8 4 Dq 0 -4 09. 2021 -8 CHE 323 -FS 18 -T 2 -58

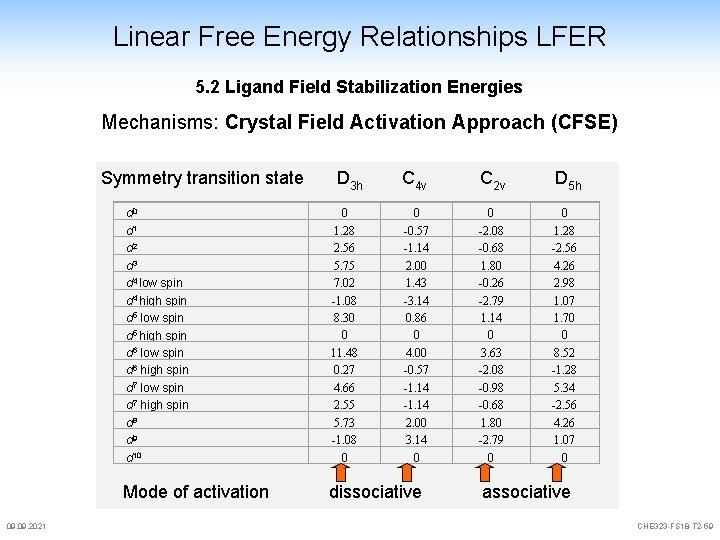
Linear Free Energy Relationships LFER 5. 2 Ligand Field Stabilization Energies Mechanisms: Crystal Field Activation Approach (CFSE) Symmetry transition state 09. 2021 D 3 h C 4 v C 2 v D 5 h 0 -0. 57 -1. 14 2. 00 1. 43 -3. 14 0. 86 0 4. 00 -0. 57 -1. 14 2. 00 3. 14 0 0 -2. 08 -0. 68 1. 80 -0. 26 -2. 79 1. 14 0 3. 63 -2. 08 -0. 98 -0. 68 1. 80 -2. 79 0 0 1. 28 -2. 56 4. 26 2. 98 1. 07 1. 70 0 8. 52 -1. 28 5. 34 -2. 56 4. 26 1. 07 0 d 1 d 2 d 3 d 4 low spin d 4 high spin d 5 low spin d 5 high spin d 6 low spin d 6 high spin d 7 low spin d 7 high spin d 8 d 9 d 10 0 1. 28 2. 56 5. 75 7. 02 -1. 08 8. 30 0 11. 48 0. 27 4. 66 2. 55 5. 73 -1. 08 0 Mode of activation dissociative associative CHE 323 -FS 18 -T 2 -59

Linear Free Energy Relationships LFER 5. 2 Ligand Field Stabilization Energies Crystal field activation energy (CFAE) is (almost) always positive. CFAE is zero for d 0, d 5 hs. d 10 (if we don't have a spin crossover) d 3, d 6 (ls), d 8 inert centres d 4 , d 7 , d 9 gain energy in other states Jahn-Teller states square pyramide favoured (KZ = 5) capped octahedral favored(KZ = 7) Analyse von CFAE is not quantitative, but enables a certain prediction 09. 2021 CHE 323 -FS 18 -T 2 -60

Linear Free Energy Relationships LFER 5. 3 Substitution in square planar complexes square planar, specially stabilized are d 8 systems Mn(-I) Fe(0) Co(I) Rh(I) Ir(I) Ni(II) Pd(II) Pt(II) Cu(III) Ag(III) Au(III) Pd 2+ about 105 time more reactive than Pt 2+ Classical reaction: cis-[Pd. Cl 2(SMe 2)2] trans-[Pd. Cl 2(SMe 2)2] v = k 1 [Pd. Cl 2(Me 2 S)2] [Me 2 S] and trans-[Pt. Cl 2(SMe 2)2] + Me 2 S* trans-[Pt. Cl 2(SMe 2)] + Me 2 S v = k 1 [Pt. Cl 2(Me 2 S)2] [Me 2 S] 09. 2021 CHE 323 -FS 18 -T 2 -61

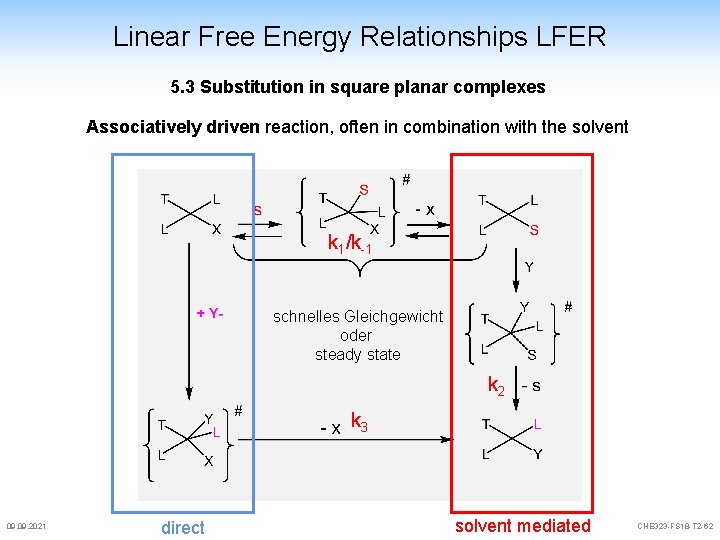
Linear Free Energy Relationships LFER 5. 3 Substitution in square planar complexes Associatively driven reaction, often in combination with the solvent k 1/k-1 schnelles Gleichgewicht oder steady state k 2 k 3 09. 2021 direct solvent mediated CHE 323 -FS 18 -T 2 -62

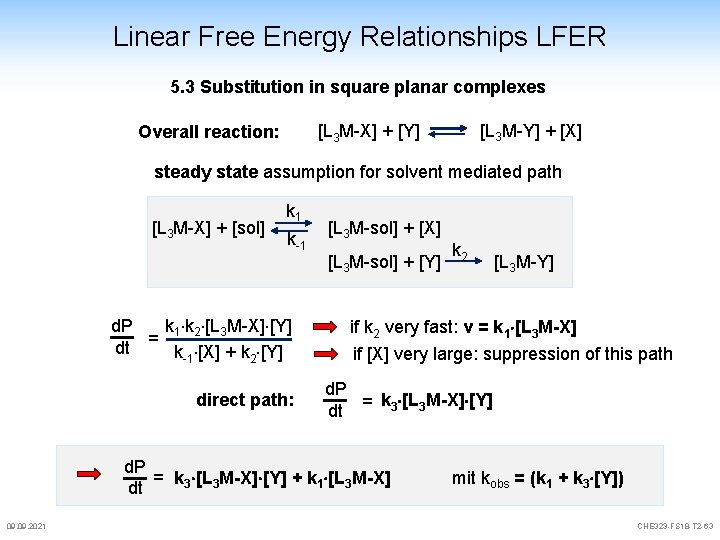
Linear Free Energy Relationships LFER 5. 3 Substitution in square planar complexes [L 3 M-Y] + [X] [L 3 M-X] + [Y] Overall reaction: steady state assumption for solvent mediated path [L 3 M-X] + [sol] k 1 k-1 k 2 [L 3 M-X] [Y] d. P dt = k [X] + k [Y] -1 2 direct path: [L 3 M-sol] + [X] [L 3 M-sol] + [Y] [L 3 M-Y] if k 2 very fast: v = k 1 [L 3 M-X] if [X] very large: suppression of this path d. P k [L M-X] [Y] dt = 3 3 d. P = k 3 [L 3 M-X] [Y] + k 1 [L 3 M-X] dt 09. 2021 k 2 mit kobs = (k 1 + k 3 [Y]) CHE 323 -FS 18 -T 2 -63

Linear Free Energy Relationships LFER 5. 3 Substitution in square planar complexes [L 3 M-Y] + [X] [L 3 M-X] + [Y] Overall reaction: prior equilibrium assumption for solvent mediated path [L 3 M-X] + [sol] Ksol [L 3 M-sol] + [X] [L 3 M-sol] + [Y] [L 3 M-X] = A; K= [L 3 M-sol] [X] [L 3 M-X] [sol] v= 09. 2021 f. B = k 2 K [A] [Y] [sol] K [sol] + [X] [L 3 M-sol] = B; [B] [A] + [B] k 2 [L 3 M-Y] = C K [sol] = K [sol] + [X] [B] = f. B [A] if K very large: v = k 2 [A][Y] if K small: suppression of this path CHE 323 -FS 18 -T 2 -64

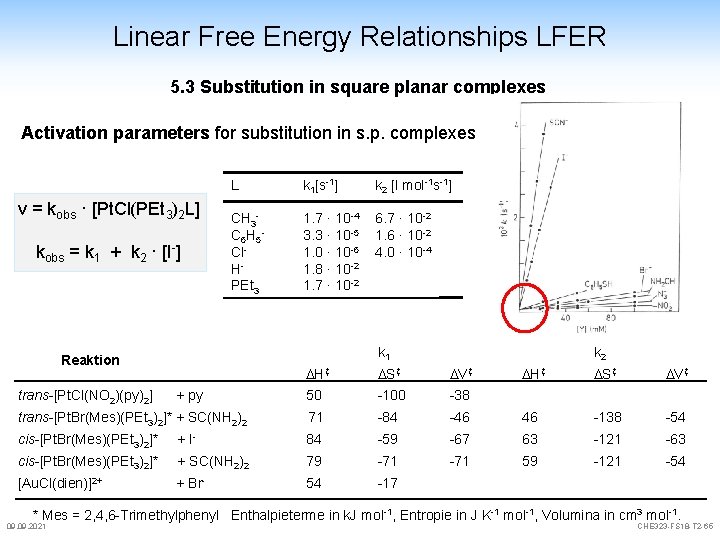
Linear Free Energy Relationships LFER 5. 3 Substitution in square planar complexes Activation parameters for substitution in s. p. complexes v = kobs · [Pt. Cl(PEt 3)2 L] kobs = k 1 + k 2 · [I-] L k 1[s-1] k 2 [l mol-1 s-1] CH 3 C 6 H 5 Cl. HPEt 3 1. 7 · 10 -4 3. 3 · 10 -5 1. 0 · 10 -6 1. 8 · 10 -2 1. 7 · 10 -2 6. 7 · 10 -2 1. 6 · 10 -2 4. 0 · 10 -4 k 1 Reaktion k 2 H‡ S‡ V‡ 50 -100 -38 trans-[Pt. Br(Mes)(PEt 3)2]* + SC(NH 2)2 71 -84 cis-[Pt. Br(Mes)(PEt 3)2]* + I- 84 cis-[Pt. Br(Mes)(PEt 3)2]* + SC(NH 2)2 [Au. Cl(dien)]2+ + Br- trans-[Pt. Cl(NO 2)(py)2] + py H‡ S‡ V‡ -46 46 -138 -54 -59 -67 63 -121 -63 79 -71 59 -121 -54 54 -17 * Mes = 2, 4, 6 -Trimethylphenyl Enthalpieterme in k. J mol-1, Entropie in J K-1 mol-1, Volumina in cm 3 mol-1. 09. 2021 CHE 323 -FS 18 -T 2 -65

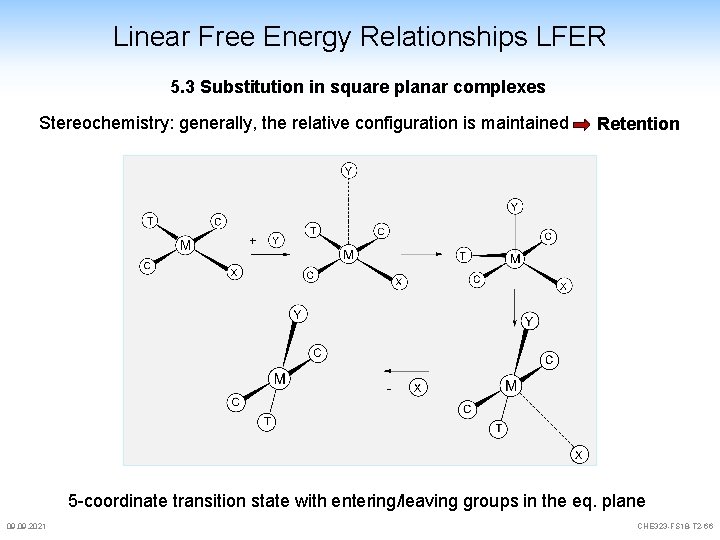
Linear Free Energy Relationships LFER 5. 3 Substitution in square planar complexes Stereochemistry: generally, the relative configuration is maintained Retention 5 -coordinate transition state with entering/leaving groups in the eq. plane 09. 2021 CHE 323 -FS 18 -T 2 -66

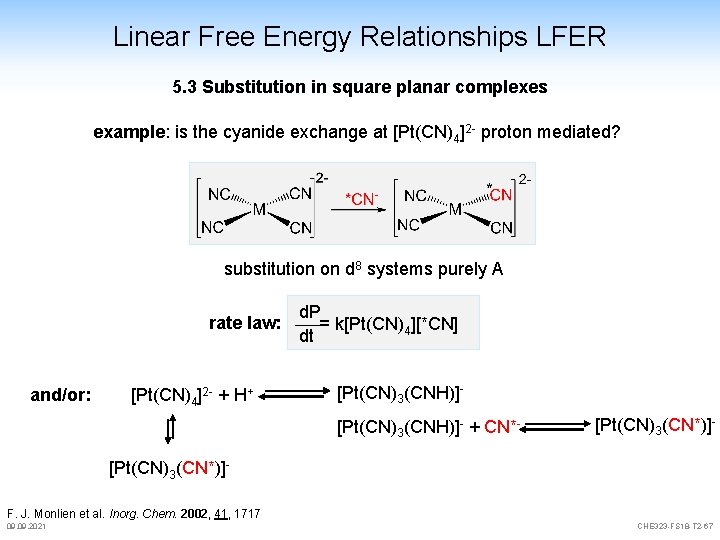
Linear Free Energy Relationships LFER 5. 3 Substitution in square planar complexes example: is the cyanide exchange at [Pt(CN)4]2 - proton mediated? substitution on d 8 systems purely A rate law: and/or: [Pt(CN)4]2 - + H+ d. P = k[Pt(CN)4][*CN] dt [Pt(CN)3(CNH)]- + CN*- [Pt(CN)3(CN*)]F. J. Monlien et al. Inorg. Chem. 2002, 41, 1717 09. 2021 CHE 323 -FS 18 -T 2 -67

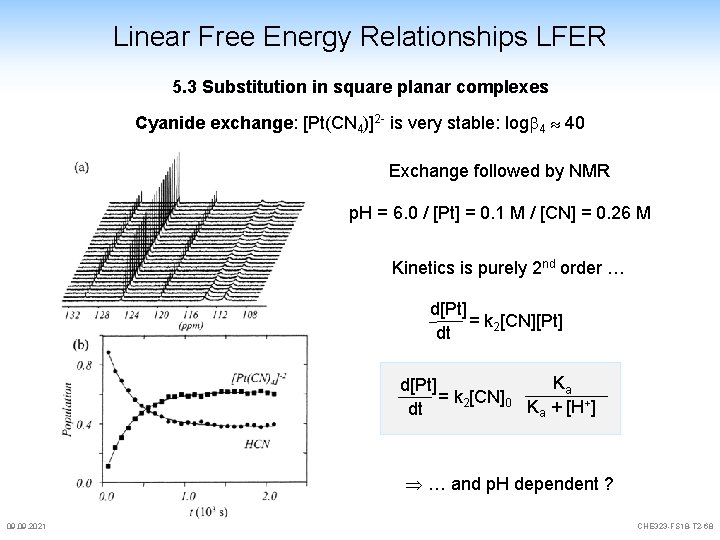
Linear Free Energy Relationships LFER 5. 3 Substitution in square planar complexes Cyanide exchange: [Pt(CN 4)]2 - is very stable: log 4 40 Exchange followed by NMR p. H = 6. 0 / [Pt] = 0. 1 M / [CN] = 0. 26 M Kinetics is purely 2 nd order … d[Pt] = k 2[CN][Pt] dt Ka d[Pt] = k 2[CN]0 Ka + [H+] dt … and p. H dependent ? 09. 2021 CHE 323 -FS 18 -T 2 -68

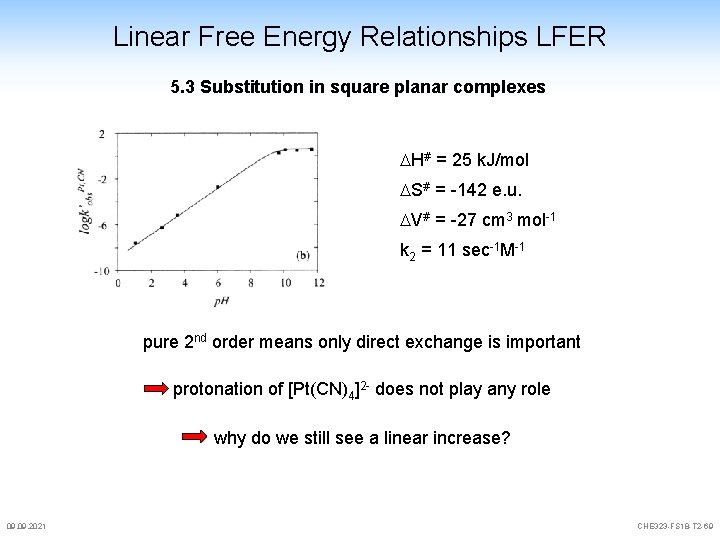
Linear Free Energy Relationships LFER 5. 3 Substitution in square planar complexes H# = 25 k. J/mol S# = -142 e. u. V# = -27 cm 3 mol-1 k 2 = 11 sec-1 M-1 pure 2 nd order means only direct exchange is important protonation of [Pt(CN)4]2 - does not play any role why do we still see a linear increase? 09. 2021 CHE 323 -FS 18 -T 2 -69

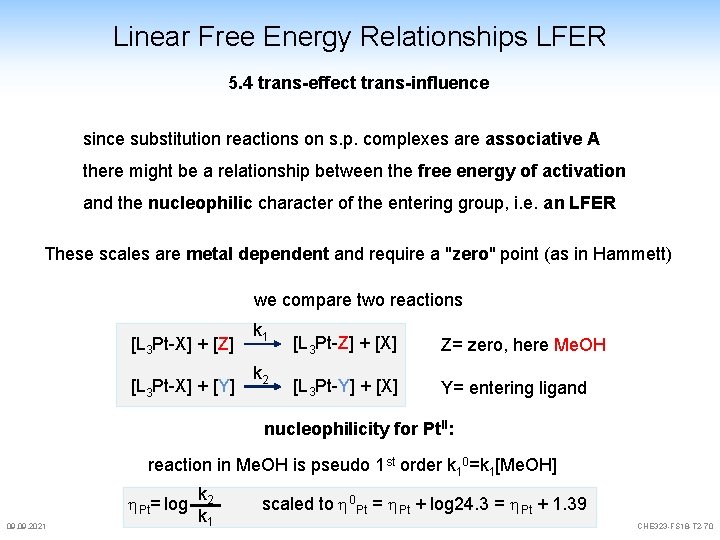
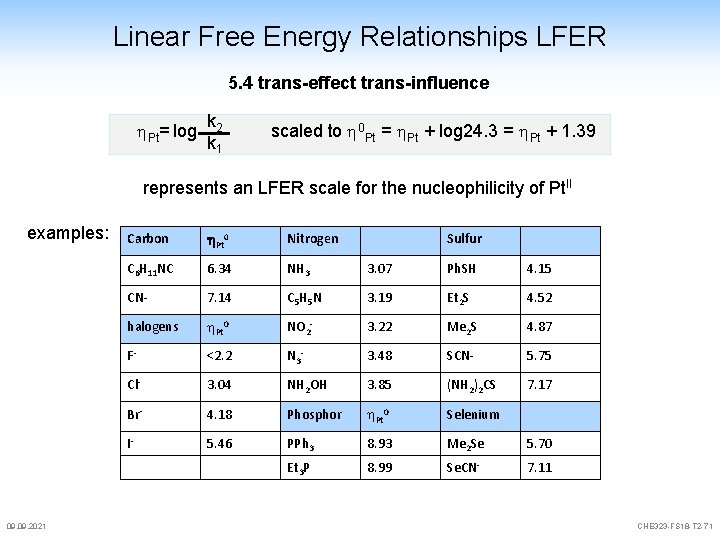
Linear Free Energy Relationships LFER 5. 4 trans-effect trans-influence since substitution reactions on s. p. complexes are associative A there might be a relationship between the free energy of activation and the nucleophilic character of the entering group, i. e. an LFER These scales are metal dependent and require a "zero" point (as in Hammett) we compare two reactions [L 3 Pt-X] + [Z] [L 3 Pt-X] + [Y] k 1 k 2 [L 3 Pt-Z] + [X] Z= zero, here Me. OH [L 3 Pt-Y] + [X] Y= entering ligand nucleophilicity for Pt. II: reaction in Me. OH is pseudo 1 st order k 10=k 1[Me. OH] Pt= log 09. 2021 k 2 k 1 scaled to 0 Pt = Pt + log 24. 3 = Pt + 1. 39 CHE 323 -FS 18 -T 2 -70

Linear Free Energy Relationships LFER 5. 4 trans-effect trans-influence Pt= log k 2 k 1 scaled to 0 Pt = Pt + log 24. 3 = Pt + 1. 39 represents an LFER scale for the nucleophilicity of Pt. II examples: 09. 2021 Carbon h. Pt 0 Nitrogen C 6 H 11 NC 6. 34 NH 3 3. 07 Ph. SH 4. 15 CN- 7. 14 C 5 H 5 N 3. 19 Et 2 S 4. 52 halogens Pt 0 NO 2 - 3. 22 Me 2 S 4. 87 F- <2. 2 N 3 - 3. 48 SCN- 5. 75 Cl- 3. 04 NH 2 OH 3. 85 (NH 2)2 CS 7. 17 Br- 4. 18 Phosphor Pt 0 Selenium I- 5. 46 PPh 3 8. 93 Me 2 Se 5. 70 Et 3 P 8. 99 Se. CN- 7. 11 Sulfur CHE 323 -FS 18 -T 2 -71

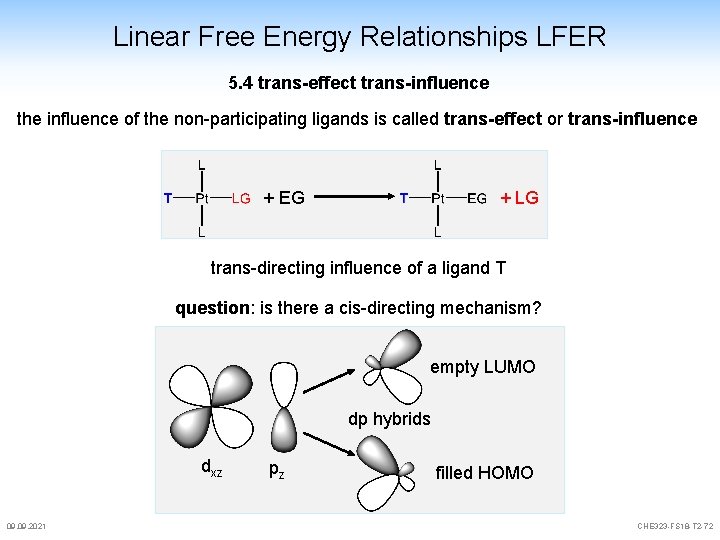
Linear Free Energy Relationships LFER 5. 4 trans-effect trans-influence the influence of the non-participating ligands is called trans-effect or trans-influence + EG + LG trans-directing influence of a ligand T question: is there a cis-directing mechanism? empty LUMO dp hybrids dxz 09. 2021 pz filled HOMO CHE 323 -FS 18 -T 2 -72

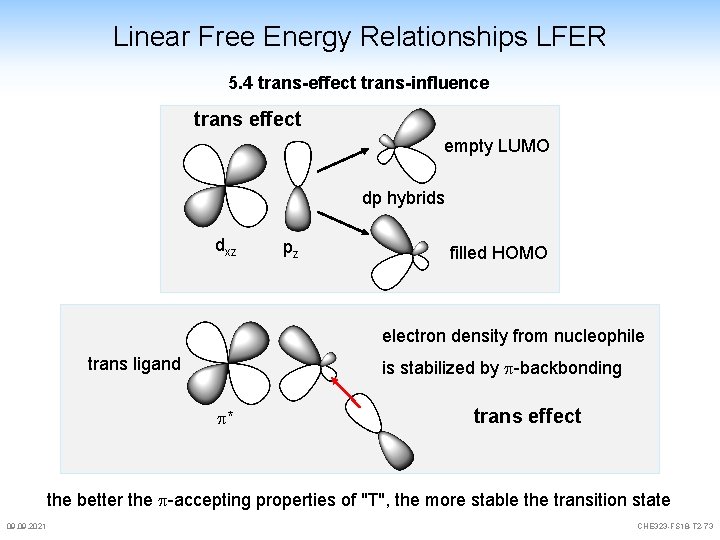
Linear Free Energy Relationships LFER 5. 4 trans-effect trans-influence trans effect empty LUMO dp hybrids dxz pz filled HOMO electron density from nucleophile trans ligand is stabilized by p-backbonding p* trans effect the better the p-accepting properties of "T", the more stable the transition state 09. 2021 CHE 323 -FS 18 -T 2 -73

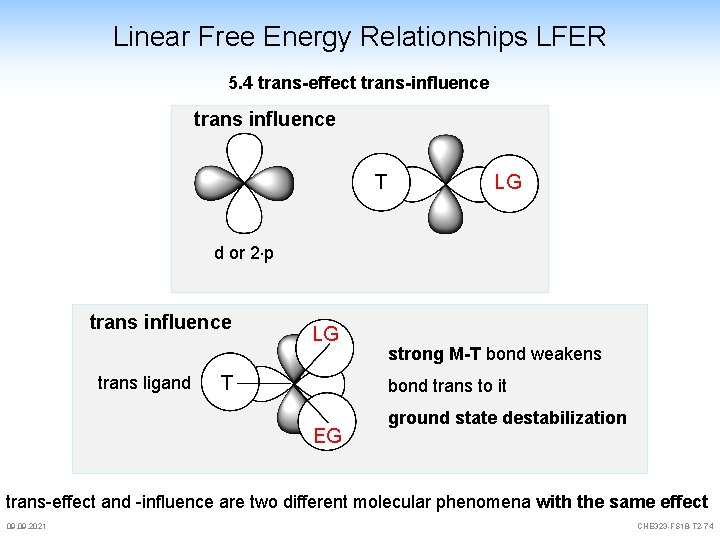
Linear Free Energy Relationships LFER 5. 4 trans-effect trans-influence trans influence T LG d or 2 p trans influence trans ligand LG T strong M-T bond weakens bond trans to it EG ground state destabilization trans-effect and -influence are two different molecular phenomena with the same effect 09. 2021 CHE 323 -FS 18 -T 2 -74

Linear Free Energy Relationships LFER 5. 4 trans-effect trans-influence weakens ground state binding energy of M-LG mirrored by bond-lengths 09. 2021 trans-ligand PR 3 Pt-Cl (Å) Cl- PEt 3 2. 294 C 2 F 5 - PMe. Ph 2 2. 361 C 6 H 5 - PPh 3 2. 408 CH 3 - PMe. Ph 2 2. 412 (H 3 C)3 Si. CH 2 - PMe 2 Ph 2. 415 H- PEt. Ph 2 2. 422 Ph 2 Me. Si- PMe. Ph 2 2. 45 CHE 323 -FS 18 -T 2 -75

Linear Free Energy Relationships LFER 5. 4 trans-effect trans-influence the trans influence mirrors an LFER since destabilization of ground state directly translate into acceleration of substitution trans influence goes about along with s-donating properties, i. e. nucleophilicity CO < H 2 O < NH 3 < NH 2 R < C 2 H 4 < Cl- < Me 2 SO < Br< Me 2 S < As. Ph 3 < PPh 3 < CH 3 - < H- < Si(CH 3) 3 to disentangle from the kinetic trans effect, rate measurements are required since the effect comes from the transition state stabilization 09. 2021 CHE 323 -FS 18 -T 2 -76

Linear Free Energy Relationships LFER 5. 4 trans-effect trans-influence to disentangle from the kinetic trans effect, rate measurements are required since the effect comes from the transition state stabilization + 09. 2021 Trans-ligand k 1 x 103 (s-1) C 2 H 4 6800 too fast CO 1210 too fast P(OMe)3 365 5240 PPh 3 27 635 PEt 3 26 495 PBu 3 23 380 PMe 3 15 275 As. Et 3 1. 7 39 Me 2 SO 0. 1 2. 8 CHE 323 -FS 18 -T 2 -77

Linear Free Energy Relationships LFER 5. 4 trans-effect trans-influence + trans effect series: H 2 O < NH 3 < Cl- < Br- < I- [NO 2]- < Me 2 S < Et 2 S < Me 2 SO < As. Et 3 < Ph- < Me- < PPh 3 < PMe 3 < P(OMe)3 H- << CO CN-< C 2 H 4 09. 2021 Trans-ligand k 1 x 103 (s-1) C 2 H 4 6800 too fast CO 1210 too fast P(OMe)3 365 5240 PPh 3 27 635 PEt 3 26 495 PBu 3 23 380 PMe 3 15 275 As. Et 3 1. 7 39 Me 2 SO 0. 1 2. 8 CHE 323 -FS 18 -T 2 -78

Linear Free Energy Relationships LFER 5. 4 trans-effect trans-influence trans-effect useful for syntheses + NH 3 cis ! + Cl- + NH 3 trans ! how to prepare? 09. 2021 CHE 323 -FS 18 -T 2 -79

Electron Transfer Reactions Redox reactions are ubiquitous in chemistry How are electrons transferred from the oxidant to the reductant? We discern two different type of electron transfer reactions: inner sphere mechanism outer sphere mechanism 09. 2021 ACIV-FS 18 -80

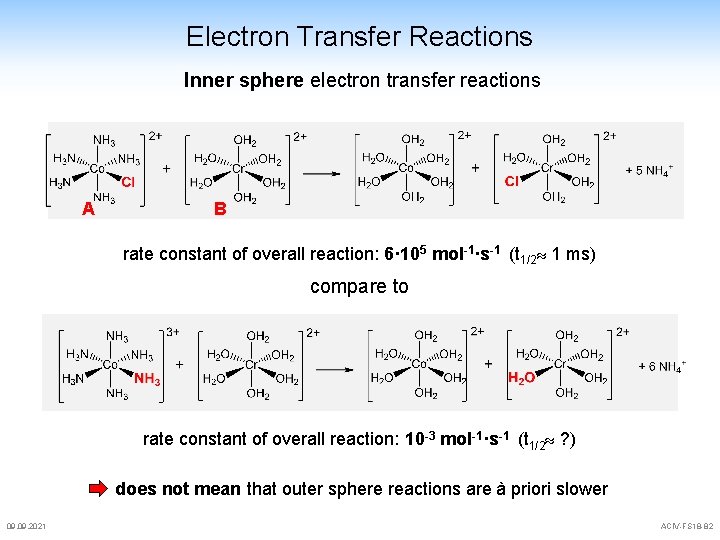
Electron Transfer Reactions Taube's famous Experiment A B rate constant of overall reaction: 6· 105 mol-1·s-1 (t 1/2 1 ms) puzzling: substitution rate Cl- for H 2 O in A is 2· 10 -6 s-1 equally puzzling: substitution rate of H 2 O for Cl- in B is 3· 10 -8 s-1 Cl- can not bind to Cr. III after the redox process only explanation: Cl- must bridge the two centres before and during e--transfer inner sphere electron transfer 09. 2021 ACIV-FS 18 -81

Electron Transfer Reactions Inner sphere electron transfer reactions A B rate constant of overall reaction: 6· 105 mol-1·s-1 (t 1/2 1 ms) compare to rate constant of overall reaction: 10 -3 mol-1·s-1 (t 1/2 ? ) does not mean that outer sphere reactions are à priori slower 09. 2021 ACIV-FS 18 -82

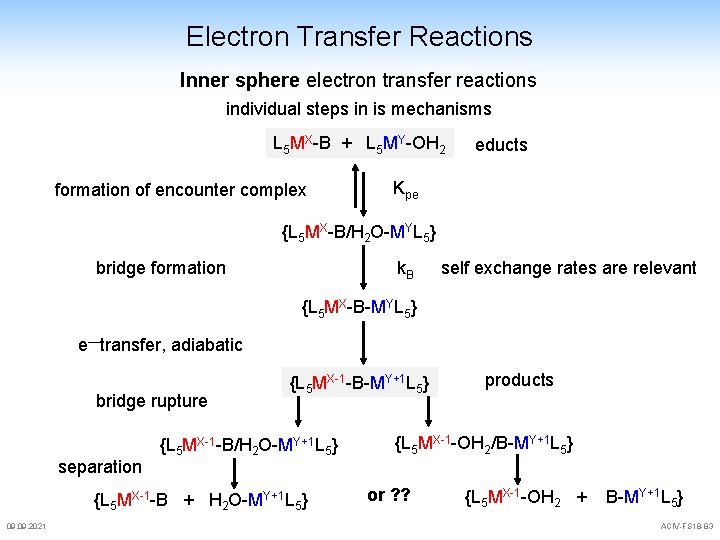
Electron Transfer Reactions Inner sphere electron transfer reactions individual steps in is mechanisms L 5 MX-B + L 5 MY-OH 2 formation of encounter complex educts Kpe {L 5 MX-B/H 2 O-MYL 5} bridge formation k. B self exchange rates are relevant {L 5 MX-B-MYL 5} e—transfer, adiabatic bridge rupture separation {L 5 MX-1 -B-MY+1 L 5} {L 5 MX-1 -B/H 2 O-MY+1 L 5} {L 5 MX-1 -B + H 2 O-MY+1 L 5} 09. 2021 products {L 5 MX-1 -OH 2/B-MY+1 L 5} or ? ? {L 5 MX-1 -OH 2 + B-MY+1 L 5} ACIV-FS 18 -83

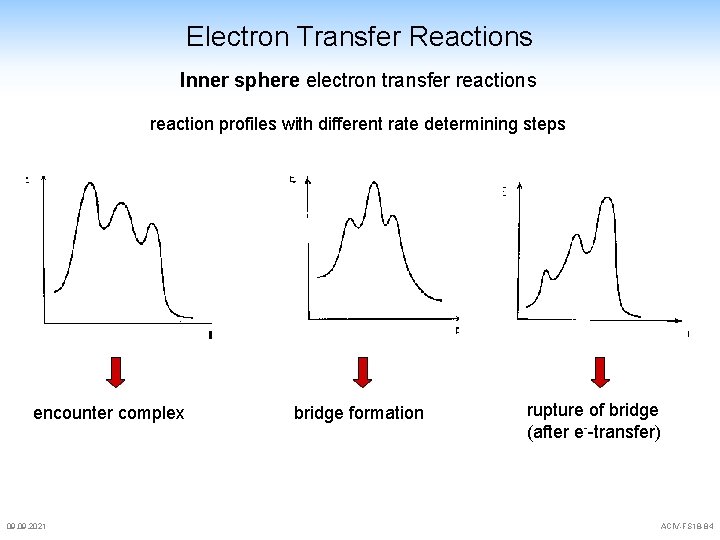
Electron Transfer Reactions Inner sphere electron transfer reactions reaction profiles with different rate determining steps encounter complex 09. 2021 bridge formation rupture of bridge (after e--transfer) ACIV-FS 18 -84

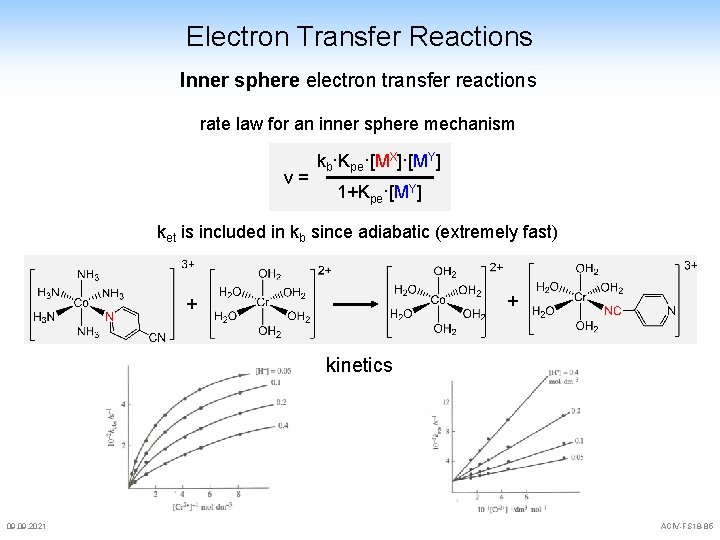
Electron Transfer Reactions Inner sphere electron transfer reactions rate law for an inner sphere mechanism v= kb·Kpe·[MX]·[MY] 1+Kpe·[MY] ket is included in kb since adiabatic (extremely fast) + + kinetics 09. 2021 ACIV-FS 18 -85

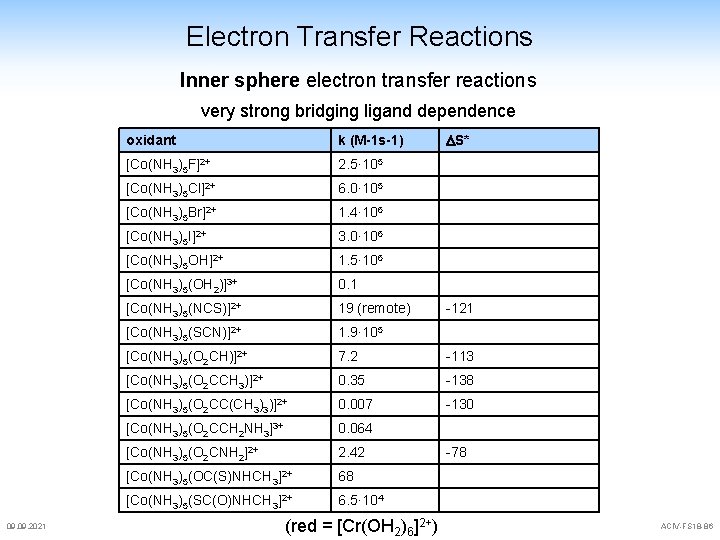
Electron Transfer Reactions Inner sphere electron transfer reactions very strong bridging ligand dependence 09. 2021 S* oxidant k (M-1 s-1) [Co(NH 3)5 F]2+ 2. 5· 105 [Co(NH 3)5 Cl]2+ 6. 0· 105 [Co(NH 3)5 Br]2+ 1. 4· 106 [Co(NH 3)5 I]2+ 3. 0· 106 [Co(NH 3)5 OH]2+ 1. 5· 106 [Co(NH 3)5(OH 2)]3+ 0. 1 [Co(NH 3)5(NCS)]2+ 19 (remote) [Co(NH 3)5(SCN)]2+ 1. 9· 105 [Co(NH 3)5(O 2 CH)]2+ 7. 2 -113 [Co(NH 3)5(O 2 CCH 3)]2+ 0. 35 -138 [Co(NH 3)5(O 2 CC(CH 3)3)]2+ 0. 007 -130 [Co(NH 3)5(O 2 CCH 2 NH 3]3+ 0. 064 [Co(NH 3)5(O 2 CNH 2]2+ 2. 42 [Co(NH 3)5(OC(S)NHCH 3]2+ 68 [Co(NH 3)5(SC(O)NHCH 3]2+ 6. 5· 104 (red = [Cr(OH 2)6]2+) -121 -78 ACIV-FS 18 -86

Electron Transfer Reactions Inner sphere electron transfer reactions criteria for inner sphere reactions: reasonable bridging ligands (typically with a lone pair) conductive ligands if the e--transfer is equal or slower than rates of substitution (if rates of e--transfer are faster than e. g. ligand self exchange then it must be os) if reactions are faster than predicted by Marcus-Hush correlation (see later) HSAB aspects 09. 2021 ACIV-FS 18 -87
![Electron Transfer Reactions Outer sphere electron transfer reactions outer sphere mechanism classical experiment Osbpy33 Electron Transfer Reactions Outer sphere electron transfer reactions outer sphere mechanism classical experiment: -[Os(bpy)3]3+](https://slidetodoc.com/presentation_image_h2/9811de8cd2ad3f5e5ddcb8c14471f64d/image-88.jpg)
Electron Transfer Reactions Outer sphere electron transfer reactions outer sphere mechanism classical experiment: -[Os(bpy)3]3+ does not racemize at all addition of L- [Os(bpy)3]2+ leads to very rapid racemization how does this work? 09. 2021 ACIV-FS 18 -88

Electron Transfer Reactions Outer sphere electron transfer reactions L 5 MX-B + L 5 MY-OH 2 educts Kpe formation of encounter complex {L 5 MX-B/H 2 O-MYL 5} e--transfer k. ET {L 5 MX-1 -B/H 2 O-MY+1 L 5} separation {L 5 MX-1 -B + H 2 O-MY+1 L 5} products rate laws follow a typical Langford-Gray mechanism v= 09. 2021 k. ET·Kpe·[MX]·[MY] 1+Kpe ·[MY] kobs = much simpler than inner sphere !! k. ET·Kpe· [MY] 1+Kpe·[MY] ACIV-FS 18 -89

Electron Transfer Reactions Outer sphere electron transfer reactions this relation ship leads to typical saturation kinetic behaviour encounter complex formation example: [Co(NH 3)5 OH 2]3+ + [Fe(CN)6]4 - fast [Co(NH 3)5 OH 23+][Fe(CN)64 -] KOS = 1500 M-1 activation – electron transfer - separation [Co(NH 3)5 OH 23+][Fe(CN)64 -] [Co(NH 3)5 OH 22+][Fe(CN)63 -] k. ET = 1. 9· 10 -1 s-1, t 1/2 = 4 sec 09. 2021 [Co(OH 2)6]2++ [Fe(CN)6]3 ACIV-FS 18 -90

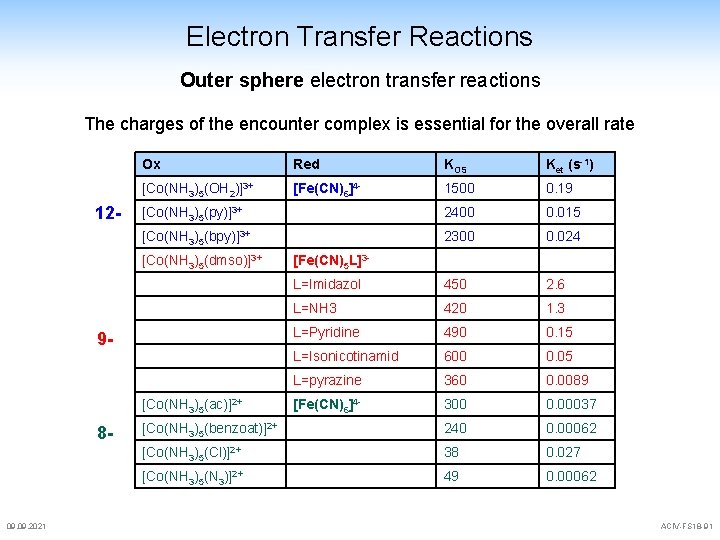
Electron Transfer Reactions Outer sphere electron transfer reactions The charges of the encounter complex is essential for the overall rate 12 - Ox Red KOS Ket (s-1) [Co(NH 3)5(OH 2)]3+ [Fe(CN)6]4 - 1500 0. 19 [Co(NH 3)5(py)]3+ 2400 0. 015 [Co(NH 3)5(bpy)]3+ 2300 0. 024 L=Imidazol 450 2. 6 L=NH 3 420 1. 3 L=Pyridine 490 0. 15 L=Isonicotinamid 600 0. 05 L=pyrazine 360 0. 0089 [Fe(CN)6]4 - 300 0. 00037 [Co(NH 3)5(benzoat)]2+ 240 0. 00062 [Co(NH 3)5(Cl)]2+ 38 0. 027 [Co(NH 3)5(N 3)]2+ 49 0. 00062 [Co(NH 3)5(dmso)]3+ 9 - [Co(NH 3)5(ac)]2+ 8 - 09. 2021 [Fe(CN)5 L]3 - ACIV-FS 18 -91

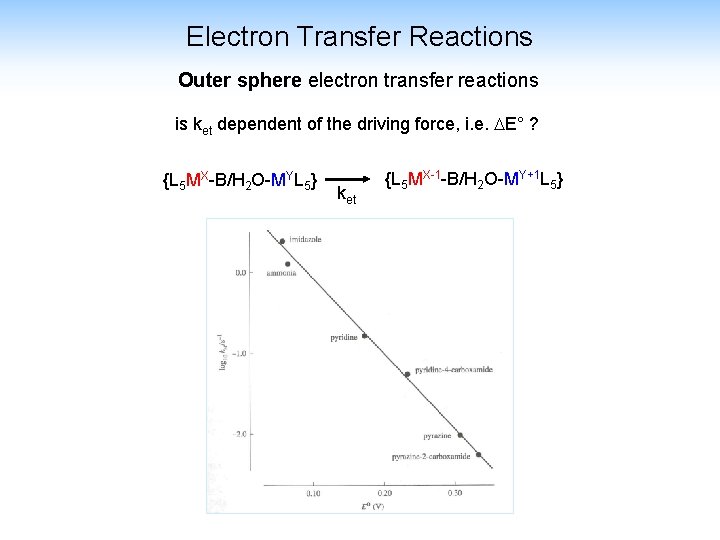
Electron Transfer Reactions Outer sphere electron transfer reactions is ket dependent of the driving force, i. e. E° ? {L 5 MX-B/H 2 O-MYL 5} ket {L 5 MX-1 -B/H 2 O-MY+1 L 5}

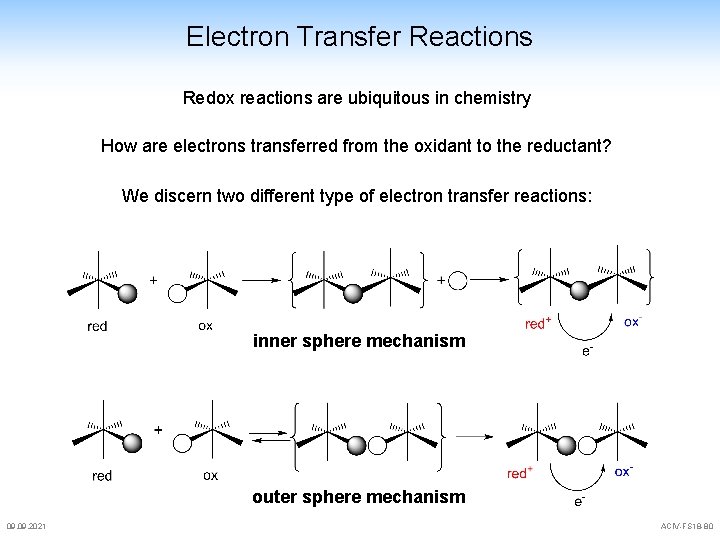
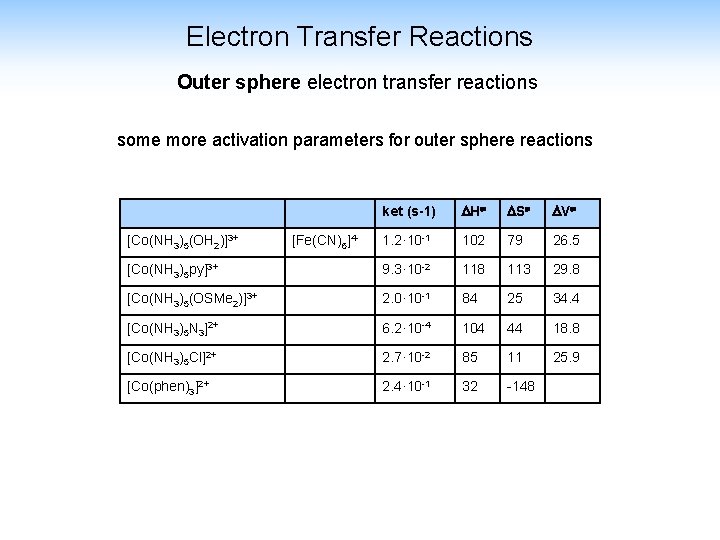
Electron Transfer Reactions Outer sphere electron transfer reactions some more activation parameters for outer sphere reactions ket (s-1) H S V 1. 2· 10 -1 102 79 26. 5 [Co(NH 3)5 py]3+ 9. 3· 10 -2 118 113 29. 8 [Co(NH 3)5(OSMe 2)]3+ 2. 0· 10 -1 84 25 34. 4 [Co(NH 3)5 N 3]2+ 6. 2· 10 -4 104 44 18. 8 [Co(NH 3)5 Cl]2+ 2. 7· 10 -2 85 11 25. 9 [Co(phen)3]2+ 2. 4· 10 -1 32 -148 [Co(NH 3)5(OH 2)]3+ [Fe(CN)6]4 -
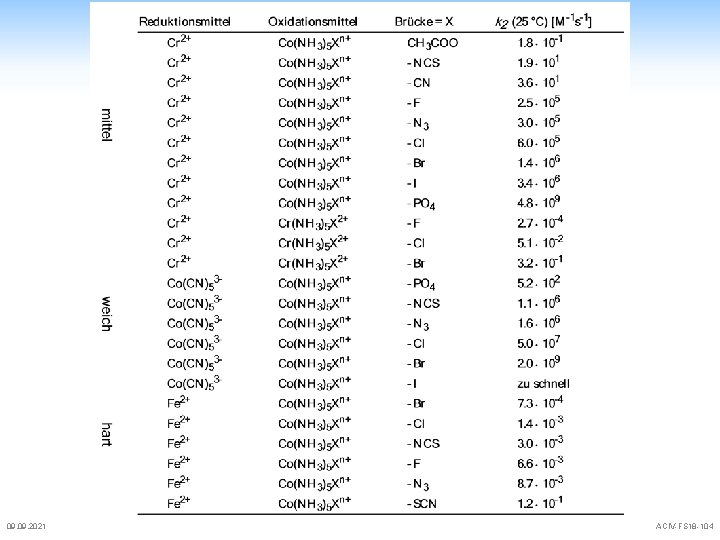
![Electron Transfer Reactions some more activation parameters for outer sphere reactions Oxidant Cobpy33 Rubpy33 Electron Transfer Reactions some more activation parameters for outer sphere reactions Oxidant [Co(bpy)3]3+ [Ru(bpy)3]3+](https://slidetodoc.com/presentation_image_h2/9811de8cd2ad3f5e5ddcb8c14471f64d/image-94.jpg)
Electron Transfer Reactions some more activation parameters for outer sphere reactions Oxidant [Co(bpy)3]3+ [Ru(bpy)3]3+ [Co(phen)3]3+ [Co(bpy)3]3+ [Co(phen)3]3+ [Co(NH 3)6]3+ [Fe(H 2 O)6]3+ [Ru(NH 3)5 py]3+ [Co(phen)3]3+ [Co(terpy)2]3+ [Co(NH 3)6]3+ [Ni(bpy)3]3+ [Co(en)3]3+ [Fe(bpy)3]3+ [Co(edta)][Ir. Cl 3]2[Co(ox)3]3[Fe(CN)6]309. 2021 Reductant [Cr(phen)3]2+ [Ru(NH 3)6]2+ [Co(terpy)2]2+ [Ru(NH 3)5 py]2+ [Ru(NH 3)6]2+ [Ru(NH 3)5 py]2+ [V(H 2 O)6]2+ [Cr(H 2 O)6]2+ [Fe(H 2 O)6]2+ [V(pic)3][Co(edta)]2[Fe(pdta)]2 [Ru(CN)6]4[Ru(NH 3)6]2+ [Co(phen)3]2+ k (M-1 s-1) 2. 0· 108 3. 7· 109 4. 2· 102 1. 5· 104 3. 0· 101 1. 9· 103 1. 1· 10 -2 7. 8· 104 3. 0· 105 4. 0· 103 3. 8· 103 1. 0· 10 -3 6. 7· 106 3. 1· 103 3. 3· 104 1. 3· 101 6. 6· 104 1. 8· 10 -1 6. 0· 106 H (k. J mol-1) S (J K-1 mol-1) 27. 6 18 21 21 56 20 0 16 7. 9 -100 -105 -155 -100 -1 -84 -138 -121 -150 7 52 29 30 19 45 -92 -4 -63 -128 -88 -108 ACIV-FS 18 -94

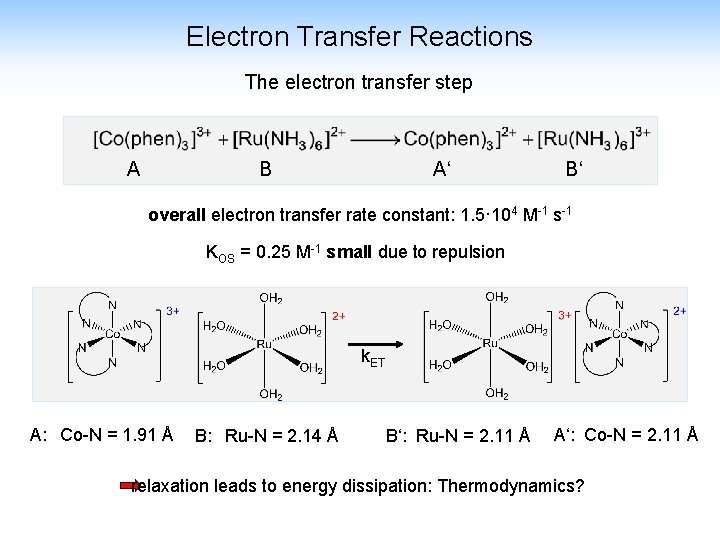
Electron Transfer Reactions The electron transfer step A B A‘ B‘ overall electron transfer rate constant: 1. 5· 104 M-1 s-1 KOS = 0. 25 M-1 small due to repulsion k. ET A: Co-N = 1. 91 Å B: Ru-N = 2. 14 Å B‘: Ru-N = 2. 11 Å A‘: Co-N = 2. 11 Å relaxation leads to energy dissipation: Thermodynamics?

Electron Transfer Reactions The electron transfer step electron transfer must obey the Franck-Condon principles electron transfer occurs after structural changes which can be illustrated with a potential diagram reaction profile describes precursor p (x=0) and product (x=1), e--transfer probability=1 where they intersect 09. 2021 G dependent on G° and lp/ls !! ACIV-FS 18 -96

Electron Transfer Reactions The electron transfer step electron transfer must obey the Franck-Condon principles different possibilities 09. 2021 ACIV-FS 18 -97

Electron Transfer Reactions The electron transfer step The Marcus equation for outer sphere e--transfer reactions os reaction: A- + B A + B- k. AB A- + A* A + A*- k. AA* B- + B* B + B*- k. BB* e--self exchange rates The different states can be expressed as follows: G AA = G (A) + G (A-) – G°(A-) Kinetics: G BB = G (B) + G (B-) – G°(B-) G AB = G (A-) + G (B) – G°(A-) Thermodynamics: G°AB = G°(B-) + G°(A) – G°(B) – G°(A-)

Electron Transfer Reactions The electron transfer step with some algebra (up to you) and the principle of microscopic reversibility we arrive at: G AB = G (AA) + G (BB) + G°(AB) if this is translated into rate constants with Marcus correlation - G ij kij= Zij·exp RT k. AB= (k. AA·k. BB·KAB·F) ½ F= Z 2 AB ZAA·ZBB if the charges remain the same left and right, F 1 e. g. Fe 3+ + Cr 2+ Fe 2+ + Cr 3+ i, j=A, B

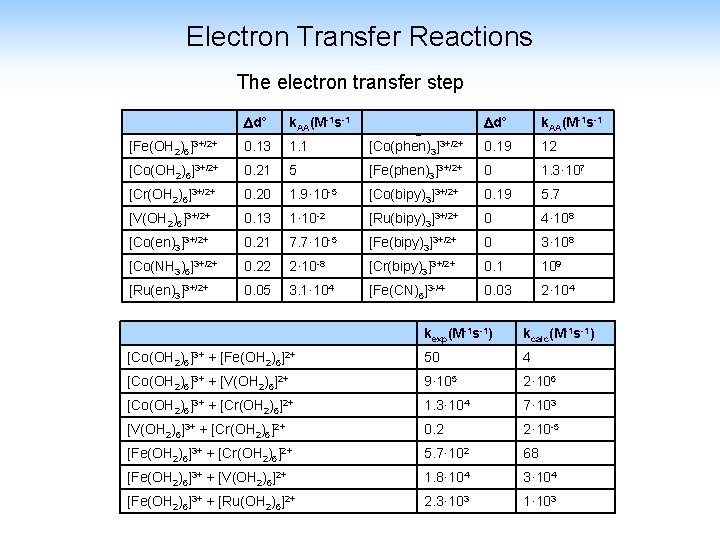
Electron Transfer Reactions The electron transfer step -1 s-1 d° k. AA(Mself electron exchange rates d° k. AA(M-1 s-1 [Fe(OH 2)6]3+/2+ 0. 13 1. 1 [Co(phen)3]3+/2+ 0. 19 12 [Co(OH 2)6]3+/2+ 0. 21 5 [Fe(phen)3]3+/2+ 0 1. 3· 107 [Cr(OH 2)6]3+/2+ 0. 20 1. 9· 10 -5 [Co(bipy)3]3+/2+ 0. 19 5. 7 [V(OH 2)6]3+/2+ 0. 13 1· 10 -2 [Ru(bipy)3]3+/2+ 0 4· 108 [Co(en)3]3+/2+ 0. 21 7. 7· 10 -5 [Fe(bipy)3]3+/2+ 0 3· 108 [Co(NH 3)6]3+/2+ 0. 22 2· 10 -8 [Cr(bipy)3]3+/2+ 0. 1 109 [Ru(en)3]3+/2+ 0. 05 3. 1· 104 [Fe(CN)6]3 -/4 - 0. 03 2· 104 kexp(M-1 s-1) kcalc(M-1 s-1) [Co(OH 2)6]3+ + [Fe(OH 2)6]2+ 50 4 [Co(OH 2)6]3+ + [V(OH 2)6]2+ 9· 105 2· 106 [Co(OH 2)6]3+ + [Cr(OH 2)6]2+ 1. 3· 104 7· 103 [V(OH 2)6]3+ + [Cr(OH 2)6]2+ 0. 2 2· 10 -5 [Fe(OH 2)6]3+ + [Cr(OH 2)6]2+ 5. 7· 102 68 [Fe(OH 2)6]3+ + [V(OH 2)6]2+ 1. 8· 104 3· 104 [Fe(OH 2)6]3+ + [Ru(OH 2)6]2+ 2. 3· 103 1· 103

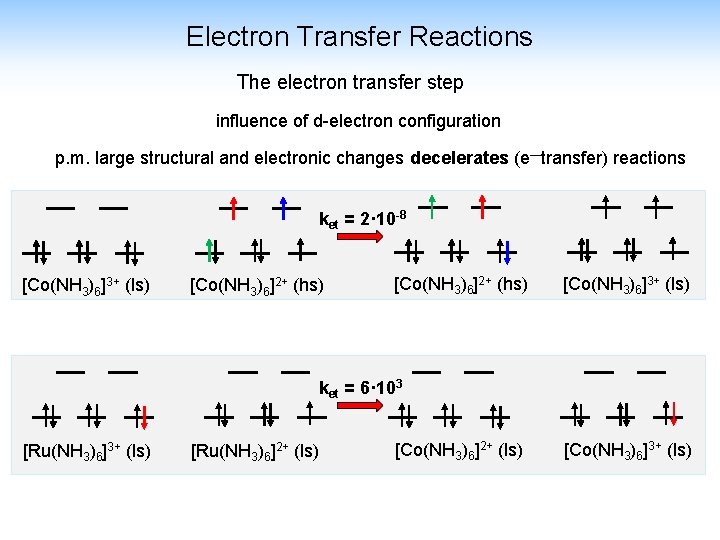
Electron Transfer Reactions The electron transfer step influence of d-electron configuration p. m. large structural and electronic changes decelerates (e—transfer) reactions ket = 2· 10 -8 [Co(NH 3)6]3+ (ls) [Co(NH 3)6]2+ (hs) [Co(NH 3)6]3+ (ls) ket = 6· 103 [Ru(NH 3)6]3+ (ls) [Ru(NH 3)6]2+ (ls) [Co(NH 3)6]3+ (ls)

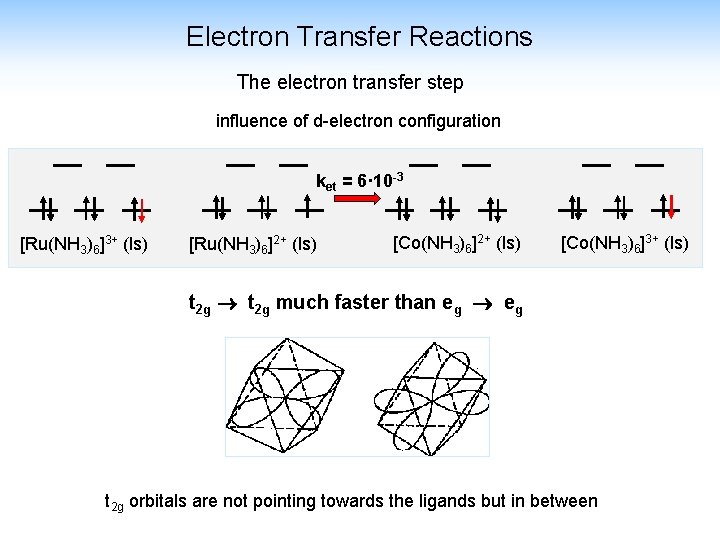
Electron Transfer Reactions The electron transfer step influence of d-electron configuration ket = 6· 10 -3 [Ru(NH 3)6]3+ (ls) [Ru(NH 3)6]2+ (ls) [Co(NH 3)6]3+ (ls) t 2 g much faster than eg t 2 g orbitals are not pointing towards the ligands but in between

Electron Transfer Reactions The electron transfer step influence of d-electron configuration kexp(M-1 s-1) kcalc(M-1 s-1) 4 (t 2 g)4(eg)2 (t 2 g)3(eg)2 [Fe(phen)3]2+ + [Fe(phen)3]3+ 3 · 107 (t 2 g)6 (t 2 g)5 [Ru(NH 3)6]2+ + [Ru(NH 3)6]3+ 8. 2 · 102 (t 2 g)6 (t 2 g)5 [Ru(phen)3]2+ + [Ru(phen)3]3+ > 107 (t 2 g)6 (t 2 g)5 [Co(H 2 O)6]2+ + [Co(H 2 O)6]3+ 5 (t 2 g)5(eg) (t 2 g)6 [Co(NH 3)6]2+ + [Co(NH 3)6]3+ 2· 10 -8 (t 2 g)5(eg)2 (t 2 g)6 [Co(en)3]2+ + [Co(en)3]3+ 1. 4 10 -4 (t 2 g)5(eg)2 (t 2 g)6 [Co(phen)3]2+ + [Co(phen)3]3+ 1. 1 (t 2 g)5(eg)2 (t 2 g)6 [Fe(H 2 O)6]2+ + [Fe(H 2 O)6]3+

09. 2021 ACIV-FS 18 -104