Declaration of Helsinki Adopted by 18 th WMA

Declaration of Helsinki Adopted by 18 th WMA General Assembly, Helsinki, June 1964 Amended by: • • 29 th WMA General Assembly, Tokyo, Japan, October 1975 35 th WMA General Assembly, Venice, Italy, October 1983 41 st WMA General Assembly, Hong Kong, September 1989 48 th WMA General Assembly, Somerset West, Republic of South Africa, October 1996 • 52 nd WMA General Assembly, Edinburgh, Scotland, October 2000 • 53 rd WMA General Assembly, Washington 2002 (Note of Clarification on paragraph 29 added) • 55 th WMA General Assembly, Tokyo 2004 (Note of Clarification on Paragraph 30 added) 59 th WMA General Assembly, Seoul, October 2008 Wednesday, September 9, 2020 Writecon, 2009, KEMH 1

CONSORT Statement Consolidated Standards of Reporting Trials Wednesday, September 9, 2020 Evidence-based, minimum set of recommendations for reporting RCTs Writecon, 2009, KEMH 2

International Society for Medical Publication Professionals Good publication practice for communicating company sponsored medical research: the GPP 2 guidelines Graf et al. BMJ (30 th Nov) 2009; 339: b 4330/doi: 10. 1136 Wednesday, September 9, 2020 Writecon, 2009, KEMH 3

GPP-2 Updated element s include: Wednesday, September 9, 2020 • Defining the roles of authors, sponsors, and other contributors • Establishing a publication steering committee • Confirmation of the role of professional medical writers Writecon, 2009, KEMH 4

New or updated codes of good practices in publications since 2003 • International Society for Medical Publication Professionals (www. ismpp. org): the role of the professional medical writer • American Medical Writers Association (www. amwa. org): the contribution of medical writers to scientific publications • Committee on Publication Ethics (http: //publicationethics. org) • Council of Science Editors (www. councilscienceeditors. org) white paper on promoting integrity in scientific journal publications • European Medical Writers Association (www. emwa. org ) Guidelines on the role of medical writers in developing peer reviewed publications • EQUATOR Network (www. equator-network. org ) Reporting guidelines— for example, CONSORT, STROBE, QUOROM/PRISMA, STARD, MOOSE • International Committee of Medical Journal Editors (www. icmje. org) Uniform requirements for manuscripts submitted to biomedical journals: writing and editing for biomedical publication Wednesday, September 9, 2020 Writecon, 2009, KEMH 5

Reporting Ethics Urmila Thatte, Dept. of Clinical Pharmacology urmilathatte@gmail. com

Introduction: Why? Para 5 Medical progress is based on research that ultimately must include studies involving human subjects. Populations that are underrepresented in medical research should be provided appropriate access to participation in research. Wednesday, September 9, 2020 Writecon, 2009, KEMH 7

Why? In whom? Para 17 Medical research involving a disadvantaged or vulnerable population or community is only justified if • the research is responsive to the health needs and priorities of this population or community and • there is a reasonable likelihood that this population stands to benefit from the results of the research. Wednesday, September 9, 2020 Writecon, 2009, KEMH 8
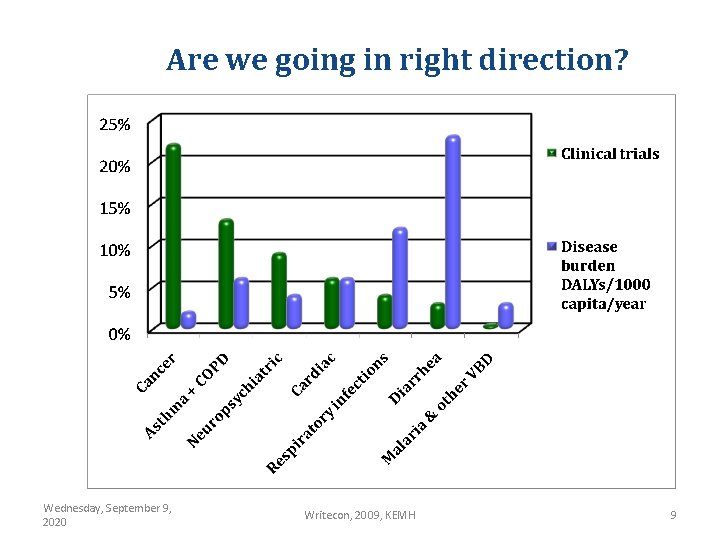
Are we going in right direction? Wednesday, September 9, 2020 Writecon, 2009, KEMH 9

Introduction: Why? Para 12 Medical research involving human subjects must • be based on a thorough knowledge of the scientific literature, • other relevant sources of information, • and adequate laboratory and, as appropriate, animal experimentation. Wednesday, September 9, 2020 Writecon, 2009, KEMH 10

CONSORT Statement “. . . it is unethical to expose human subjects unnecessarily to the risks of research”. Wednesday, September 9, 2020 Do we need this research? Writecon, 2009, KEMH 11
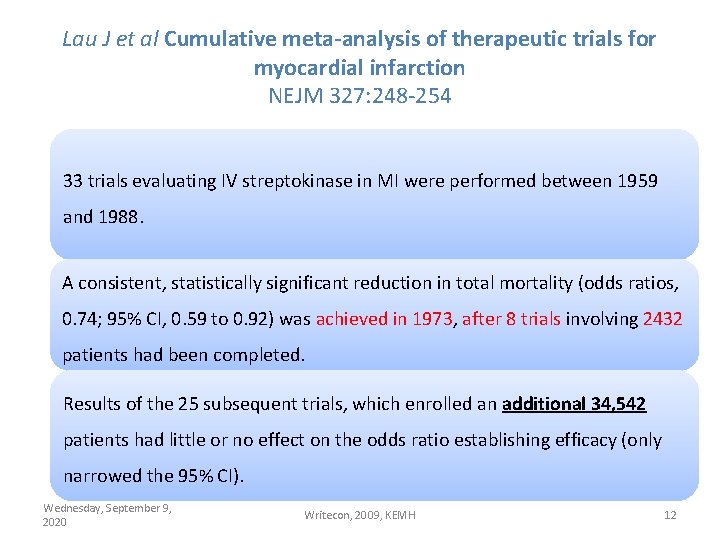
Lau J et al Cumulative meta-analysis of therapeutic trials for myocardial infarction NEJM 327: 248 -254 33 trials evaluating IV streptokinase in MI were performed between 1959 and 1988. A consistent, statistically significant reduction in total mortality (odds ratios, 0. 74; 95% CI, 0. 59 to 0. 92) was achieved in 1973, after 8 trials involving 2432 patients had been completed. Results of the 25 subsequent trials, which enrolled an additional 34, 542 patients had little or no effect on the odds ratio establishing efficacy (only narrowed the 95% CI). Wednesday, September 9, 2020 Writecon, 2009, KEMH 12

Methods: Protocol Para 14 Protocol should include information regarding • conflicts of interest, • incentives for subjects and • provisions for treating and/or compensating subjects who are harmed as a consequence of participation in the research study. • post-study access by study subjects to interventions identified as beneficial in the study or access to other appropriate care or benefits. Wednesday, September 9, 2020 Writecon, 2009, KEMH 13
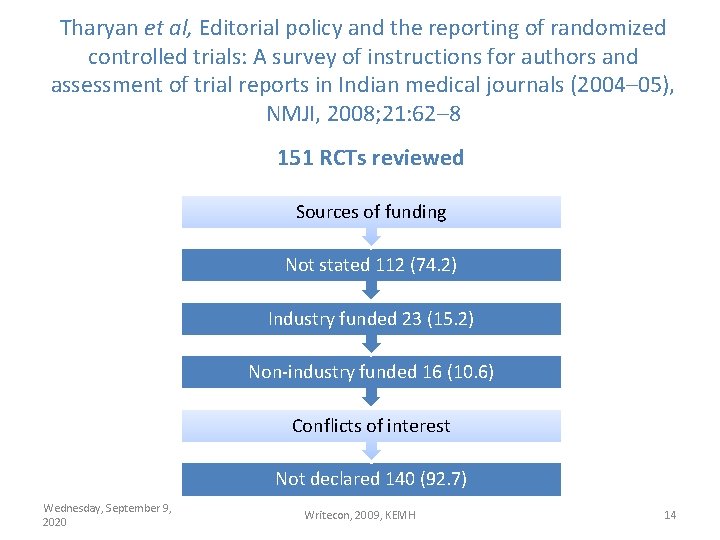
Tharyan et al, Editorial policy and the reporting of randomized controlled trials: A survey of instructions for authors and assessment of trial reports in Indian medical journals (2004– 05), NMJI, 2008; 21: 62– 8 151 RCTs reviewed Sources of funding Not stated 112 (74. 2) Industry funded 23 (15. 2) Non-industry funded 16 (10. 6) Conflicts of interest Not declared 140 (92. 7) Wednesday, September 9, 2020 Writecon, 2009, KEMH 14

Methods - CONSORT Eligibility criteria Wednesday, September 9, 2020 Setting To assess the external validity (generalisability) of the results Single/Multicentric: ethics committee “shopping” Method of recruitment, such as by referral or selfselection (for example, through advertisements). ? Responsibility of sponsors Writecon, 2009, KEMH 15
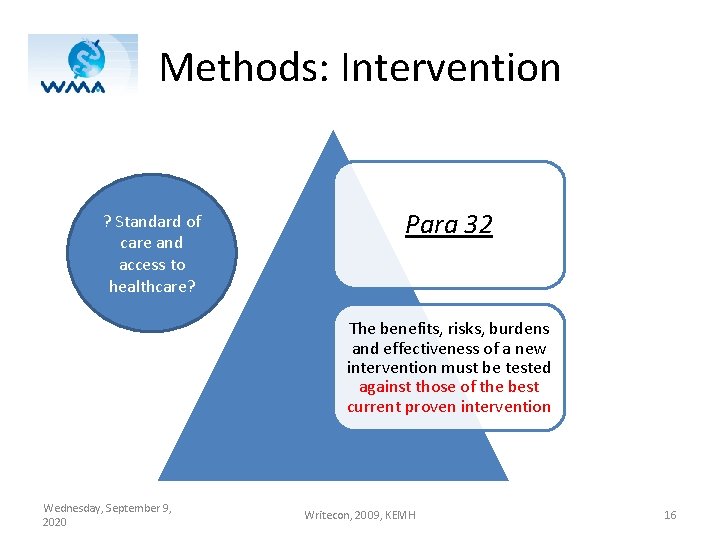
Methods: Intervention ? Standard of care and access to healthcare? Para 32 The benefits, risks, burdens and effectiveness of a new intervention must be tested against those of the best current proven intervention Wednesday, September 9, 2020 Writecon, 2009, KEMH 16

“Country profiles of Environmental Burden of Disease” by WHO (2009) http: //www. who. int/quantifying_ehimpacts/national/coun tryprofile/india. pdf Wednesday, September 9, 2020 Population of 1. 1 billion 35% officially below the international poverty line Under- 5 mortality rate 76/1000 live births Life expectancy: 63 years (2006) Writecon, 2009, KEMH 17

Access to medicines? Expenditure on health: 5 -6% of total expenditure 50% on drugs Govt. Spends between 4% of GDP on health 2 -10% of health budget on drugs (approximately Rs. 2000 crores) Wednesday, September 9, 2020 Writecon, 2009, KEMH 18

Trials registered in ctri (accessed 2 nd December 2009) and disease burden estimates Wednesday, September 9, 2020 Writecon, 2009, KEMH 19

Intervention: Standard of care? • Universal standard of care? • Access to health care? • Why are trials being undertaken? Wednesday, September 9, 2020 Writecon, 2009, KEMH 20

Methods: Intervention 212 of the trials at ctri are placebo controlled Para 32 • Use of placebo/ no treatment acceptable where no current proven intervention exists • Where for compelling and scientifically sound methodological reasons the use of placebo is necessary to determine the efficacy or safety of an intervention • Patients who receive placebo /no treatment will not be subject to any risk of serious or irreversible harm. Extreme care must be taken to avoid abuse of this option. Wednesday, September 9, 2020 Writecon, 2009, KEMH 21

Methods: Intervention (CONSORT) Blinding/Masking Wednesday, September 9, 2020 Translations (? placebo, ? blinded) Writecon, 2009, KEMH 22

Methods Para 19 • Every clinical trial must be registered in a publicly accessible database before recruitment of the first subject Para 30 • Authors have a duty to make publicly available the results of their research on human subjects and are accountable for the completeness and accuracy of their reports. Wednesday, September 9, 2020 Writecon, 2009, KEMH 23
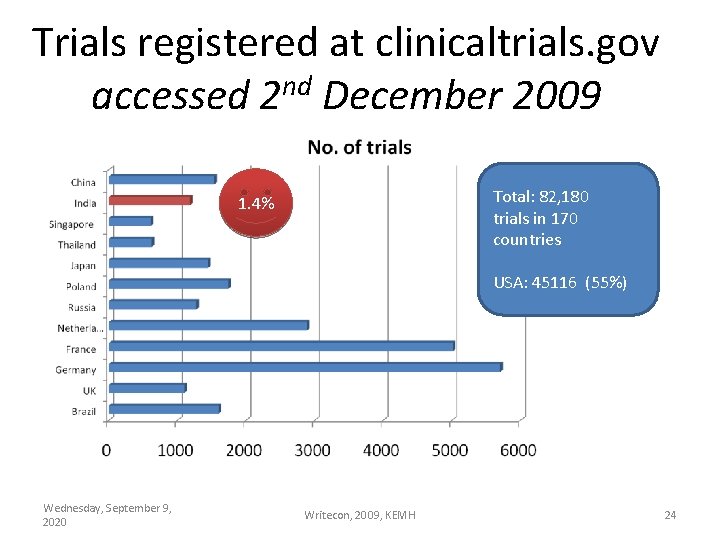
Trials registered at clinicaltrials. gov nd accessed 2 December 2009 Total: 82, 180 trials in 170 countries 1. 4% USA: 45116 (55%) Wednesday, September 9, 2020 Writecon, 2009, KEMH 24
Investigator Initiated trials (USA) Voluntary: Investigator to submit trial data to Clinical. Trials. gov using the Protocol Registration System (PRS). A trial must be • approved by a human subject review board and • must conform to the regulations of the appropriate national health authorities. Wednesday, September 9, 2020 Writecon, 2009, KEMH 25

U. S. Public Law 110 -85 (FDAAA) • The Food and Drug Administration Amendments Act of 2007 (FDAAA or US Public Law 11085) was passed on September 27, 2007. Wednesday, September 9, 2020 Writecon, 2009, KEMH • The law requires mandatory registration and results reporting for certain clinical trials of drugs, biologics, and devices. 26
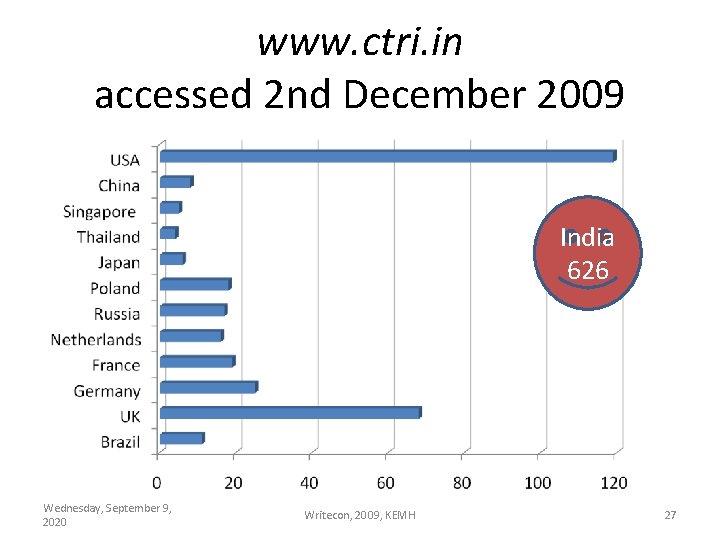
www. ctri. in accessed 2 nd December 2009 India 626 Wednesday, September 9, 2020 Writecon, 2009, KEMH 27

India launched Clinical Trials Registry 20 Jul 2007 1 st trial registered 29 th August 2007 302 till June 15 th 2009 (14 per month) Wednesday, September 9, 2020 Writecon, 2009, KEMH 28
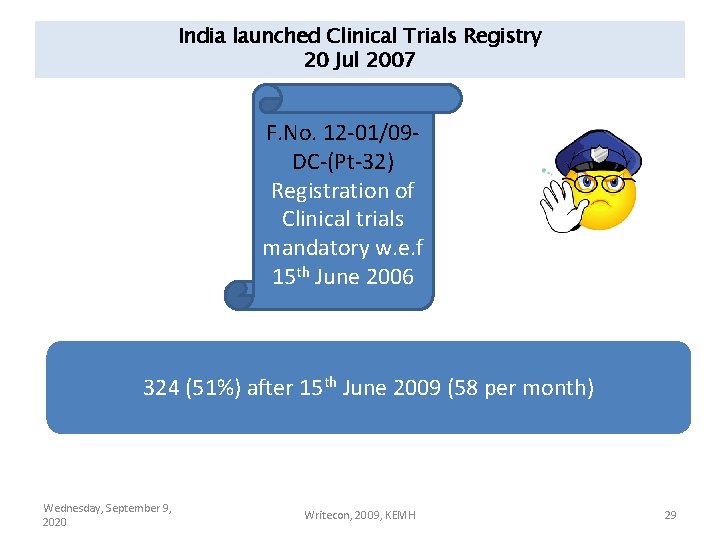
India launched Clinical Trials Registry 20 Jul 2007 F. No. 12 -01/09 DC-(Pt-32) Registration of Clinical trials mandatory w. e. f 15 th June 2006 324 (51%) after 15 th June 2009 (58 per month) Wednesday, September 9, 2020 Writecon, 2009, KEMH 29

Methods Para 23 Protect the privacy of research subjects and confidentiality of their personal information and to minimize the impact of the study on their physical, mental and social integrity. Para 11 It is the duty of physicians who participate in medical research to protect the life, health, dignity, integrity, right to self-determination, privacy, and confidentiality of personal information of research subjects Wednesday, September 9, 2020 Writecon, 2009, KEMH 30

ICMR Guidelines, 2006 IX. Researcher’s relations with the media and publication Clear consent for publication be obtained besides the consent for participation in research Preferably be obtained on two separate occasions and not as a blanket one at the commencement of the study. Wednesday, September 9, 2020 Writecon, 2009, KEMH 31

ICMR Guidelines, 2006 IX. Researcher’s relations with the media and publication Maintenance of confidentiality in case of publication / presentation of photographs/ slides / videos of participant (s), prior consent should be obtained. Identification features camouflaged. Wednesday, September 9, 2020 Writecon, 2009, KEMH 32

Twin pillars of human protection Independent Review Informed Consent Para 15 Wednesday, September 9, 2020 Para 24 33 Writecon, 2009, KEMH

Bavdekar SB, Gogtay NJ, Wagh S. Reporting ethical processes in two Indian journals. Indian J Med Sci 2008; 62: 134 -40 132 manuscripts reviewed from Indian Pediatrics and Indian J of Pediatrics 29. 5% reported having obtained approval from the ethics committee. 47% indicated that informed consent was obtained from parents or lawful guardians. Neither ethical approval nor informed consent was mentioned in 34% published articles reporting prospective studies. Only 15% reported that children's assent was obtained. Only four eligible studies reported ethics committee's approval, informed consent, as well as assent. Wednesday, September 9, 2020 Writecon, 2009, KEMH 34
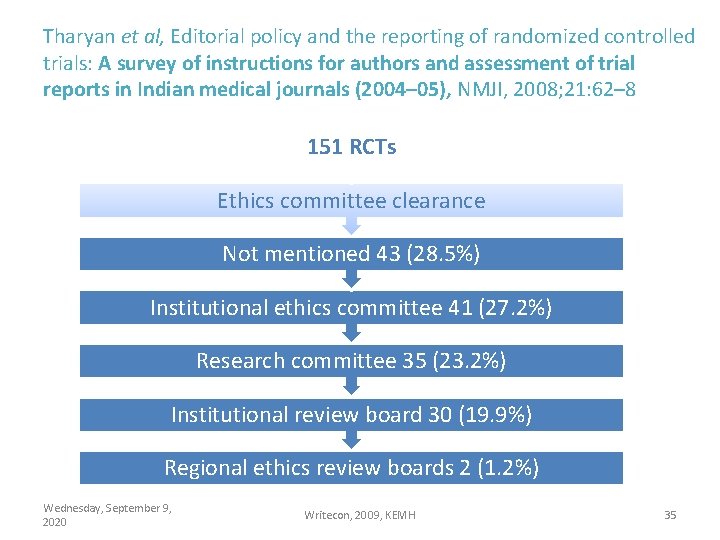
Tharyan et al, Editorial policy and the reporting of randomized controlled trials: A survey of instructions for authors and assessment of trial reports in Indian medical journals (2004– 05), NMJI, 2008; 21: 62– 8 151 RCTs Ethics committee clearance Not mentioned 43 (28. 5%) Institutional ethics committee 41 (27. 2%) Research committee 35 (23. 2%) Institutional review board 30 (19. 9%) Regional ethics review boards 2 (1. 2%) Wednesday, September 9, 2020 Writecon, 2009, KEMH 35
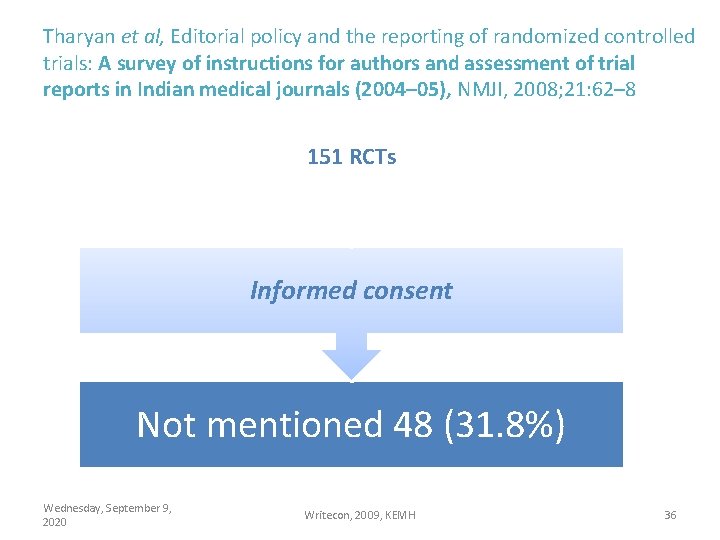
Tharyan et al, Editorial policy and the reporting of randomized controlled trials: A survey of instructions for authors and assessment of trial reports in Indian medical journals (2004– 05), NMJI, 2008; 21: 62– 8 151 RCTs Informed consent Not mentioned 48 (31. 8%) Wednesday, September 9, 2020 Writecon, 2009, KEMH 36

Bavdekar SB, Gogtay NJ, Chavan R. Reporting ethical processes: Survey of 'instructions to authors' provided by Indian journals. Indian J Med Sci 2009; 63: 260 -2 No. of Journals not providing instructions regarding reporting of: ethics committee approval: 38/100 Informed Consent: 56/100 Assent : 97/100 Wednesday, September 9, 2020 Writecon, 2009, KEMH 37

Methods Para 25 • For medical research using identifiable human material or data, physicians must normally seek consent for the collection, analysis, storage and/or reuse. • Waiver conditions defined (must seek approval of a research ethics committee). Wednesday, September 9, 2020 Writecon, 2009, KEMH 38

Para 30 Communicating results. . . • Authors, editors and publishers all have ethical obligations with regard to the publication of the results of research. Wednesday, September 9, 2020 • Authors have a duty to make publicly available the results of their research on human subjects and are accountable for the completeness and accuracy of their reports. Writecon, 2009, KEMH • Negative and inconclusive as well as positive results should be published or otherwise made publicly available. 39

“The file-drawer problem” (Rosenthal, 1979) Publication bias described by Sterling (1959): 97% of studies published in four major psychology journals provided statistically significant results Wednesday, September 9, 2020 1995: same author concluded publication bias unchanged Writecon, 2009, KEMH 40

Extent of publication bias in different categories of research cohorts: a meta-analysis of empirical studies BMC Medical Research Methodology 2009, 9: 79 doi: 10. 1186/1471 -2288 -9 -79 The pooled odds ratio of publication of studies with positive results, compared to those without positive results (publication bias) was • 2. 78 (95% CI: 2. 10 to 3. 69) • 5. 00 (95% CI: 2. 01 to 12. 45) in trials submitted to regulatory authority, • 1. 70 (95% CI: 1. 44 to 2. 02) in abstracts Wednesday, September 9, 2020 Writecon, 2009, KEMH 41

Turner et al, Selective Publication of Antidepressant Trials and Its Influence on Apparent Efficacy, NEJM, 2008; 358: 252 -260 74 studies examined FDA deemed 38/74 (51%) positive, 37/38 published 36/74 studies (49%) deemed negative (24) or questionable (12) by FDA 3/36 studies published as not positive 22/36 not published 11 were published as positive Wednesday, September 9, 2020 12 times as likely Writecon, 2009, KEMH 42 Studies that the FDA judged as positive were approximately

Turner et al, Selective Publication of Antidepressant Trials and Its Influence on Apparent Efficacy, NEJM, 2008; 358: 252 -260 Total of 12, 564 patients participated in these trials Data from 3449 patients (27%) were not published. Additional 1843 patients (15%) data were reported as positive in conflict with the FDA outcome Wednesday, September 9, 2020 Writecon, 2009, KEMH 43

Results and further Para 33 At the conclusion of the study, Patients entered into the study are entitled to be informed about the outcome of the study and to share any benefits that result from it, for example, access to interventions identified as beneficial in the study or to other appropriate care or benefits. Wednesday, September 9, 2020 Writecon, 2009, KEMH 44

Declaration of Helsinki, 2008 Para 30 Wednesday, September 9, 2020 Reports of research not in accordance with the principles of this Declaration should not be accepted for publication. Writecon, 2009, KEMH Bavdekar et al – 60/100 Journals Instructions did not mention the need for mention that study was in accordance with Do. H 45

Albert Einstein • Relativity applies to physics, not ethics
- Slides: 46