Decay Rate Contents Probability of decay and Activity
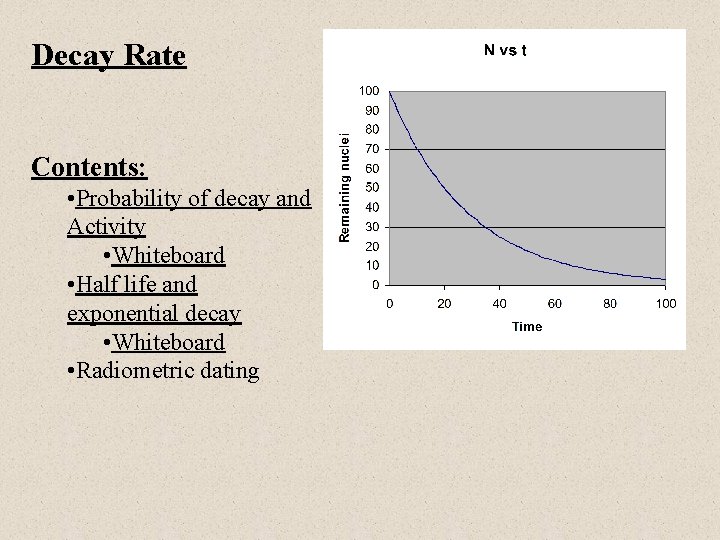
Decay Rate Contents: • Probability of decay and Activity • Whiteboard • Half life and exponential decay • Whiteboard • Radiometric dating

Probability and activity N - Number of un-decayed nuclei (number) - Per second probability of a nuclei decaying (s-1) A - Activity - decays/sec (Becquerels (Bq) = s-1) A = - N/ t = N TOC

Example - Radon 222 has an atomic mass of 222. 02. How many grams of it do you have if your activity is 8. 249 x 1016 decays/sec, and your decay probability is 2. 098 x 10 -6 s-1? (4) NA = 6. 02 x 1023 atoms/mol n = N/NA n = (grams you have)/(molar mass) A = N 8. 249 x 1016 s-1 = (2. 098 x 10 -6 s-1)N N = 3. 93184 x 1022 nuclei n = (3. 93184 x 1022)/(6. 02 x 1023 mol-1) = 0. 065312954 grams = n(molar mass) = (0. 065312954)(222. 02) = 14. 5 g

Whiteboards: Activity and decay probability 1|2 TOC

What is the activity if you have a of 3. 19 x 10 -10 s-1, and you have 5. 12 x 1023 undecayed nuclei? A = N = (3. 19 x 10 -10 s-1)(5. 12 x 1023) = 1. 63 x 1014 decays/s (or s-1) 1. 63 x 1014 decays/sec W

What is the if 1. 27 x 1020 un-decayed nuclei generate 1420 decays per second? A = N 1420 s-1 = (1. 27 x 1020) = 1. 12 x 10 -17 s-1 W

Half life and exponential decay - Per second probability of a nuclei decaying (s-1) N - Number of un-decayed nuclei (number) A = Activity - decays/sec (s-1) A = - N/ t N = Noe- t - Exponential decay No - Original value N - Value at time t t - Elapsed time Diff EQ… A = Noe- t TOC
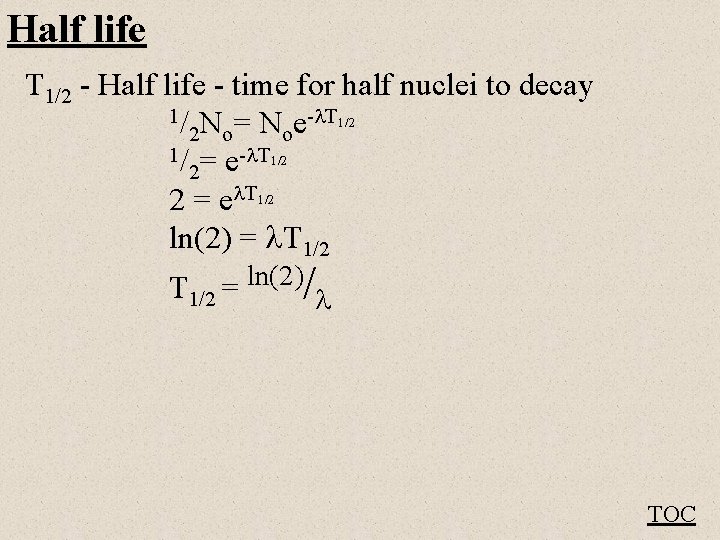
Half life T 1/2 - Half life - time for half nuclei to decay 1/ N = N e- T 1/2 2 o o 1/ = e- T 1/2 2 2 = e T 1/2 ln(2) = T 1/2 = ln(2)/ TOC
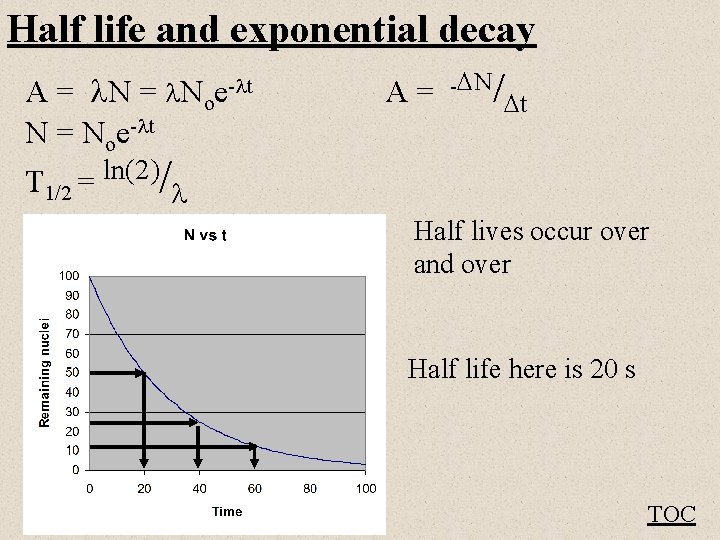
Half life and exponential decay A = Noe- t A = - N/ t N = Noe- t T 1/2 ln(2) = / Half lives occur over and over Half life here is 20 s TOC

Half life and exponential decay A = Noe- t A = - N/ t N = Noe- t T 1/2 ln(2) = / Example: Bi 211 has a half life of 128. 4 s. What is the per-second probability of a nuclei decaying? If you start out with 32 grams of Bi 211, how much is left after 385. 2 s? After what time is there 23 grams left? What is the activity when there is 23 grams left? (m = 210. 987 u) Use formulas Cheat (385. 2 s = 3 half lives) TOC

Example: Bi 211 has a half life of 128. 4 s. What is the per-second probability of a nuclei decaying? If you start out with 32 grams of Bi 211, how much is left after 385. 2 s? After what time is there 23 grams left? What is the activity when there is 23 grams left? (m = 210. 987 u) so the = ln 2/128. 4 = 0. 005398343 s-1. and the N (just use grams) at 385. 2 seconds would be: N = Noe- t = (32 g)e-(0. 005398343 s-1)(385. 2 s) = 4. 0 g (How come it is exact? ? ? – 385. 2 = 3*128. 4 so it is exactly 3 half lives. look for that on tests and stuff. ) Use the same formula for finding the time it will be 23 g (less than one half life 128. 4 s – right? ) N = Noe- t (23 g) = (32 g)e-(0. 005398343 s-1)t so t = 61 s Activity is just given by A = N, so we use chemistry to find N: N = (6. 02 x 1023 atoms/mol)(23 g)/210. 987 g/mol) = 6. 56249 E+22 atoms so A = N = (0. 005398343 s-1)(6. 56249 E+22 atoms) = 3. 54266 E+20 counts per second

Whiteboards: Half Life and Decay 1|2|3|4|5|6 TOC

Oregonium has a decay probability of 8. 91 x 10 -8 s-1. What is its half life in days? T 1/2 = ln(2)/(8. 91 x 10 -8 s-1) = 7779429. 636 s = 90. 0 days W

What is the nuclear decay probability of a substance that has a half life of 96. 23 minutes? T 1/2 = ln(2)/ - (in data packet) T 1/2 = (96. 23 min)(60 sec/min) = 5773. 8 s = ln(2)/(5773. 8 s) = 0. 0001201 s-1 W

Oregonium has a decay probability of 8. 91 x 10 -8 s-1. If you have 1250 grams of Oregonium initially, how many grams do you have after 30. 00 days? (x 24 x 3600) N = Noe- t - (in data packet) N = (1250) e-(8. 91 x 10 -8)(30*24*3600) N = 992 g W

Tualatonium has a half life of 12 seconds. If you start with 64 grams of it, how much remains after a minute? (Cheat) 60 seconds is exactly 5 half lives, so divide in half five times 64/25 = 2. 0 grams W

Tigardium has a half life of 8. 34 seconds. The initial activity is 1350 counts/second, after what time is the activity 125 counts/sec? = ln(2)/T 1/2 = 0. 083111173 s-1 N = Noe- t - (in data packet) N/No = e- t ln(N/No) = - t ln(No/N) = t ln(No/N)/ = t = ln(1350/125)/(0. 083111173 s-1) t = 28. 6 s W

A certain substance has an activity of 1245 counts/sec initially, and an activity of 938 counts/second after exactly 3. 00 minutes. What is the half life of the substance? N = Noe- t - (in data packet) N/No = e- t ln(N/No) = - t ln(No/N) = t ln(No/N)/t = = ln(1245/938)/(180. s) =. 001573 s-1 T 1/2 = ln(2)/(. 001573 s-1) = 441 s W

Radiometric dating Know original proportion of unstable nuclei Measure current proportion Use N = Noe- t to calculate elapsed time Carbon 14 dating: C 14 created in atmosphere (T 1/2 = 5730 Y) Living things absorb C 14 in known amounts They die, and quit absorbing C 14 Rocks can be dated Hardening forms nearly pure crystals Magma is product of fission within the earth Lead - Earth’s age - Uranium/Lead - Clean room - lead in fuel. TOC
- Slides: 19