Decade of field experience with TB Gene Xpert

Decade of field experience with TB Gene. Xpert IAS 2019 Satellite session Mexico, 22 July 2019 By Jean de Dieu IRAGENA Technical Officer Laboratory WHO/AFRO, HIV/TB and Hepatitis Programme iragenaj@who. int

WHO-endorsed Rapid Diagnostic Test for TB (1) Xpert MTB/RIF, 2010 and 2013 2010 Policy Recommendation Xpert MTB/RIF is recommended rather than conventional microscopy, culture and DST as the initial diagnostic test in adults presumed to have MDR-TB or HIV-associated TB. 2013 Policy Update Xpert MTB/RIF is recommended rather than conventional microscopy and culture as the initial diagnostic test in all adults and children with signs and symptoms of pulmonary and extrapulmonary TB © Gerhard Jörén/ UNITAID 2 2
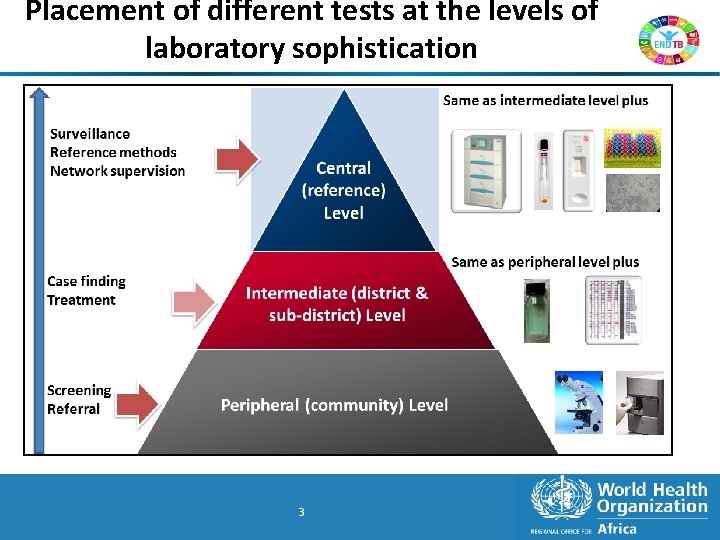
Placement of different tests at the levels of laboratory sophistication 3 3
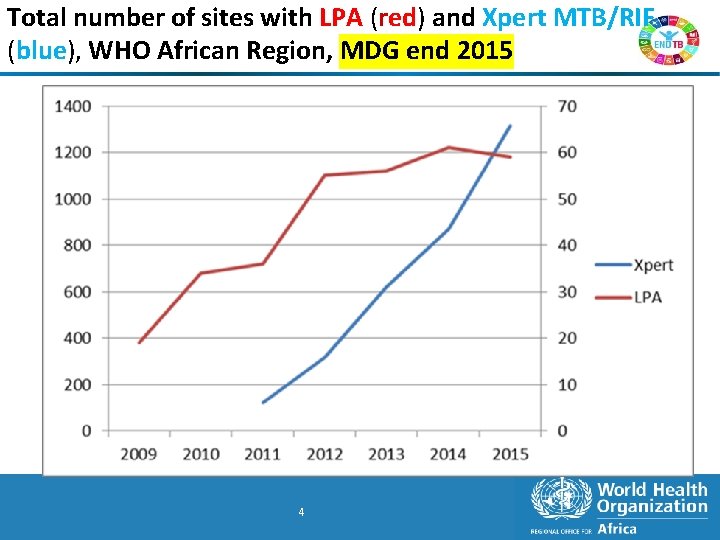
Total number of sites with LPA (red) and Xpert MTB/RIF (blue), WHO African Region, MDG end 2015 4 4

WHO-endorsed Rapid Diagnosti Test for TB (2) Xpert MTB/RIF Ultra, 2017 Xpert MTB/RIF Ultra is recommended rather than conventional microscopy and culture as the initial diagnostic test in all adults and children with signs and symptoms of pulmonary and extrapulmonary TB (CSF, lymph nodes and tissue specimens) S S 5 Ultra has a higher sensitivity for detection of MTB than Xpert MTB/RIF particularly in: S Smear -/Culture + specimens (+17%) S Paediatric, extra-pulmonary specimens (CSF) S HIV + patients whose specimens are frequently paucibacillary The expert group recognized that the impact of increased sensitivity results in decreased specificity for TB detection and becomes a trade-off between increased diagnosis and overtreatment. 5

Areas of diagnostic integration Additional Xpert assays, 2018 • Xpert cartridges for other diseases and conditions can be used on the same Gene. Xpert instrument as for Xpert MTB/RIF • WHO Prequalified: Xpert HIV-1 Qual assay (EID), Xpert Hepatitis C and HIV Viral Load assays • Potential areas for integration: Testing site personnel, trainings, maintenance, supply systems, specimen referral, quality assurance, etc. See WHO Information Note on adoption and use of multi-disease testing devices (Download the document on www. who. int/tb) 6 6
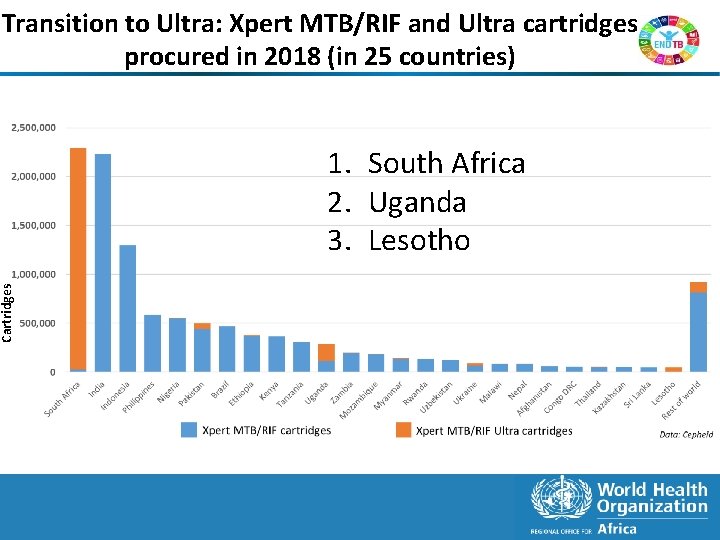
Transition to Ultra: Xpert MTB/RIF and Ultra cartridges procured in 2018 (in 25 countries) Cartridges 1. South Africa 2. Uganda 3. Lesotho 7
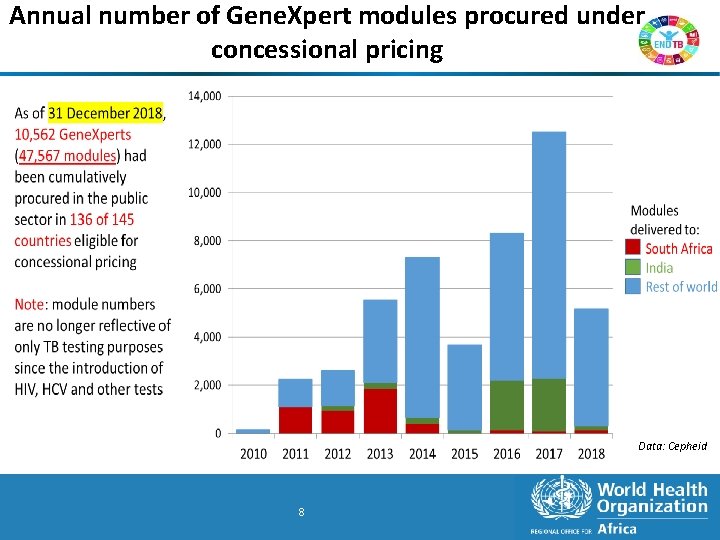
Annual number of Gene. Xpert modules procured under concessional pricing Data: Cepheid 8 8
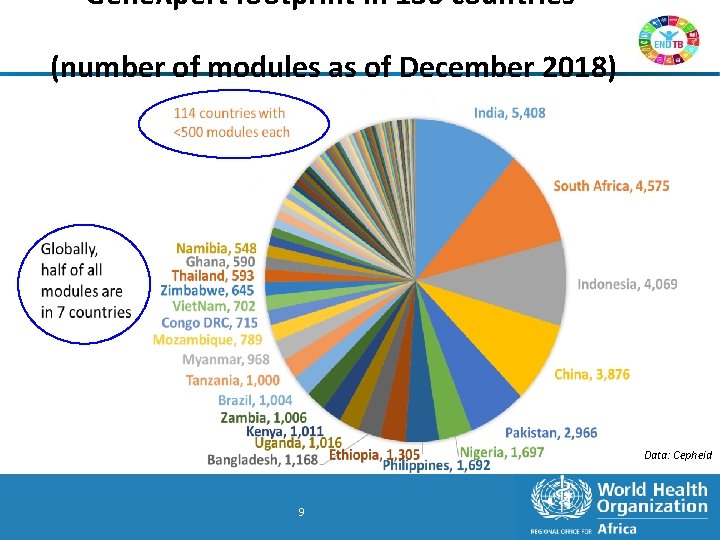
Gene. Xpert footprint in 136 countries (number of modules as of December 2018) Data: Cepheid 9 9
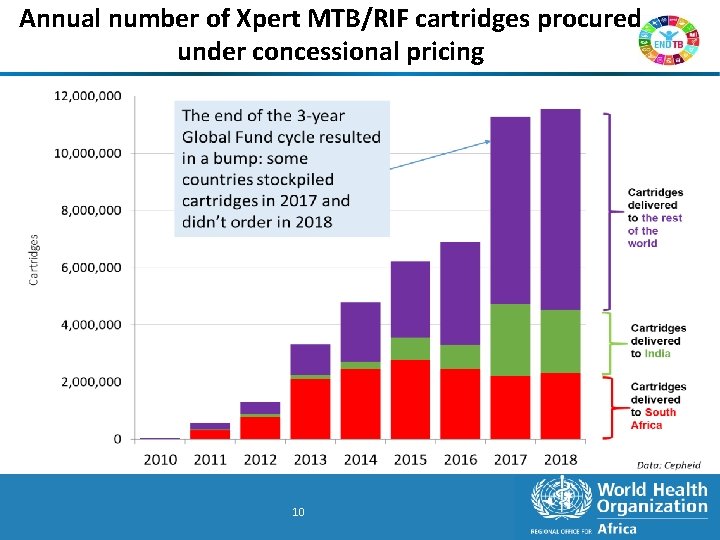
Annual number of Xpert MTB/RIF cartridges procured under concessional pricing 10 10

Countries procuring the most Xpert MTB/RIF cartridges and Gene. Xpert modules, cumulatively since 2010 11 Country Cartridges Country Modules South Africa 15, 344, 020 India 5, 408 India 6, 908, 250 South Africa 4, 575 Indonesia 2, 047, 550 Indonesia 4, 069 Brazil 1, 690, 930 China 3, 876 Nigeria 1, 482, 090 Pakistan 2, 966 Kenya 1, 476, 050 Nigeria 1, 697 Philippines 1, 457, 510 Philippines 1, 692 Pakistan 1, 456, 020 Ethiopia 1, 305 Ethiopia 997, 240 Bangladesh 1, 168 Tanzania 995, 900 Uganda 1, 016 Uganda 942, 790 Kenya 1, 011 Bangladesh 746, 060 Zambia 1, 006 Data: Cepheid

Countries procuring the most Xpert MTB/RIF cartridges and Gene. Xpert modules in 2018 12 Country Cartridges Country Modules South Africa 2, 293, 020 Indonesia 926 India 2, 230, 340 Mozambique 430 Indonesia 1, 296, 750 Zambia 328 Philippines 584, 230 Myanmar 277 Nigeria 551, 550 India 204 Pakistan 495, 470 Philippines 176 Brazil 471, 250 Ukraine 176 Ethiopia 375, 400 Bangladesh 172 Kenya 363, 950 Benin 162 Tanzania 308, 500 Thailand 144 Uganda 288, 150 Namibia 140 Zambia 199, 850 Tanzania 129 Data: Cepheid
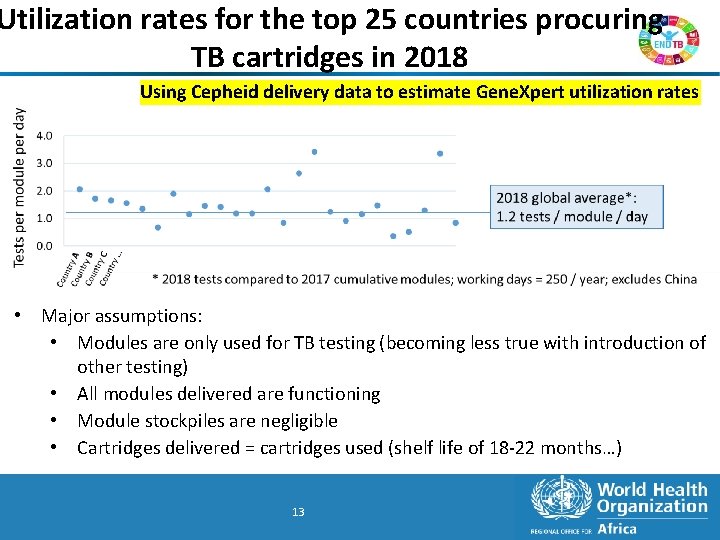
Utilization rates for the top 25 countries procuring TB cartridges in 2018 Using Cepheid delivery data to estimate Gene. Xpert utilization rates • Major assumptions: • Modules are only used for TB testing (becoming less true with introduction of other testing) • All modules delivered are functioning • Module stockpiles are negligible • Cartridges delivered = cartridges used (shelf life of 18 -22 months…) 13 13

Partnership to assist countries in scale-up S Global Laboratory Initiative for Africa (GLIAfrica), 2014 Other partners 14 14
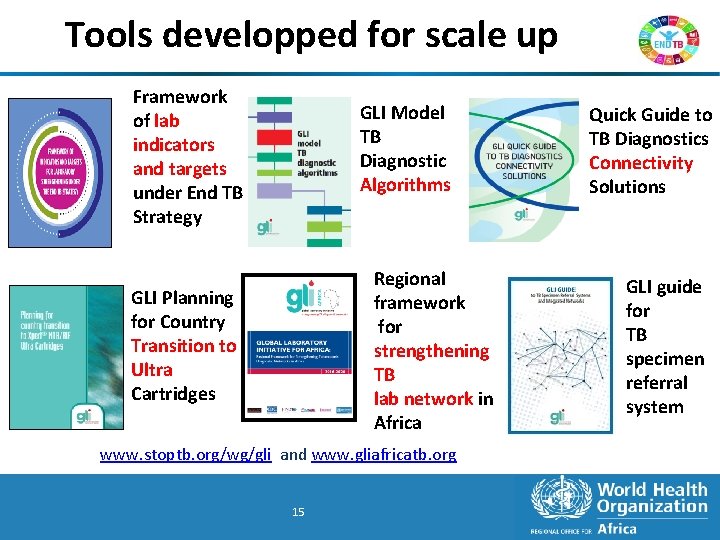
Tools developped for scale up Framework of lab indicators and targets under End TB Strategy GLI Model TB Diagnostic Algorithms Regional framework for strengthening TB lab network in Africa GLI Planning for Country Transition to Ultra Cartridges www. stoptb. org/wg/gli and www. gliafricatb. org 15 15 Quick Guide to TB Diagnostics Connectivity Solutions GLI guide for TB specimen referral system

Gene. Xpert Omni/Edge The Edge is a single module Gene. Xpert instrument connected to a tablet device Evaluation the Omni instrument delayed until early 2020 S S S 16 16 Small and Portable Durable Low Power Consumption Automatic Connectivity Solid State Integrated Battery

Challenges (1) S S S The uptake of Xpert technology has been slow in African countries in decade Diagnostic policies have not been translated into practice at country levels Country ownership for laboratory services is still weak: S S No policy reform at country level, no budget allocated to laboratory services Structures for laboratory services within Ministries of Health are poorly developed, hence HR issues in management Lack of national laboratory strategic and operational plans and clear algorithm for the use of Xpert as the initial diagnostic test S Lack of uninterrupted power supply and sample referral system to optimise use of Gene. Xpert instruments S Lack of training of clinicians, which creates a gap in 17 17 understanding and communication between lab technicians and S

Challenges (2) S S Gene. Xpert instrument maintenance End TB Strategy has very ambitious targets for increasing rapid testing S S 18 there is a considerable gap between the number of instruments that exist in a country and the number of instruments that are actually needed to provide nationwide coverage Currently, efforts are focused on optimizing the too few instruments that are in place without a focus on access 18

Conclusion S Increasing access to rapid detection of TB using Gene. Xpert platform will require major efforts at country level S the goal should be the access for all people in need and the optimal utilisation rate should not be a barreer to ensuring or improving access S Efforts being made by TB programmes to increase utilization of their Gene. Xperts should not be overlooked when planning for introduction of new tests S Multi-disease testing platforms will provide opportunities for laboratory integration and Service and maintenance costs should be shared across disease programmes S Modeling suggests that all TB high burden countries require additional Gene. Xperts and tests to meet the potential demand for TB diagnosis. 19 19

Thank you 20
- Slides: 20