Debiopharm Group Wilma Lukas BIO International Convention 2011

Debiopharm Group™ Wilma Lukas BIO International Convention 2011 © Debiopharm Group™ 2011

Debiopharm Group™ : Key Features 2 g Key expertise : drug development g Founded in 1979, privately-owned, financially independent g Headquarters in Lausanne, Switzerland g Operational centres : Debiopharm S. A. (Lausanne, Switzerland) Debio R. P. S. A. (Martigny, Switzerland) Debioclinic S. A. (Paris, France) Debioinnovation S. A. (Lausanne, Switzerland) Debiomanagement S. A. (Lausanne, Switzerland) g Team : staff of more than 300 g Extensive international expert network (over 400 experts) © Debiopharm Group™ 2011
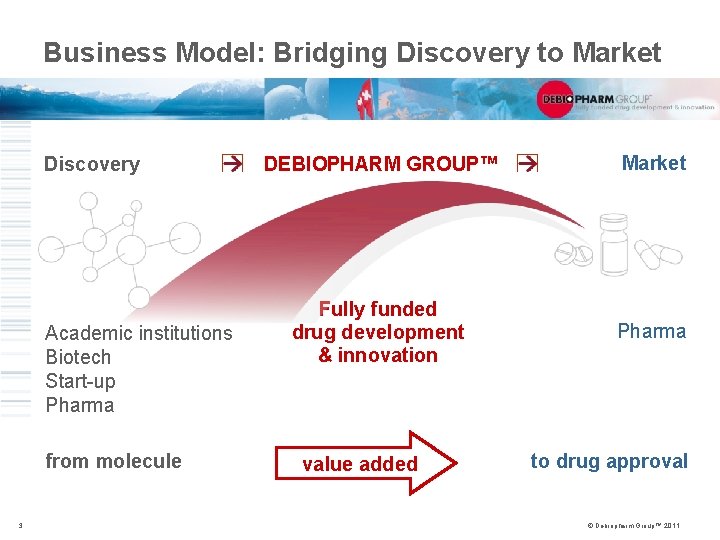
Business Model: Bridging Discovery to Market Discovery Academic institutions Biotech Start-up Pharma from molecule 3 DEBIOPHARM GROUP™ Market Fully funded drug development & innovation Pharma value added to drug approval © Debiopharm Group™ 2011

Debiopharm Group™: Key Features g. Five products commercialised worldwide Eloxatin® / Elplat® / Dacotin® / Dacplat® Decapeptyl® / Trelstar® / Pamorelin® 1 -month Decapeptyl® / Trelstar® / Pamorelin® 3 -month Decapeptyl® / Trelstar® 6 -month Moapar® / Salvacyl® 3 -month 4 © Debiopharm Group™ 2011

Main Current Partners: all over the World Licensors Licensees Sanofi USA u Curis u Mercury Therapeutics u MSM Protein Tech u Tulane University u Yale University Japan u Nagoya City Univ. Novartis Pfizer Inc. Aché Dr Reddy’s Laboratories Ferring Ipsen France u Pharmaleads u Tc. Land India u Aurigene Mepha Technofarma Watson Pharmaceuticals Yakult 5 © Debiopharm Group™ 2011
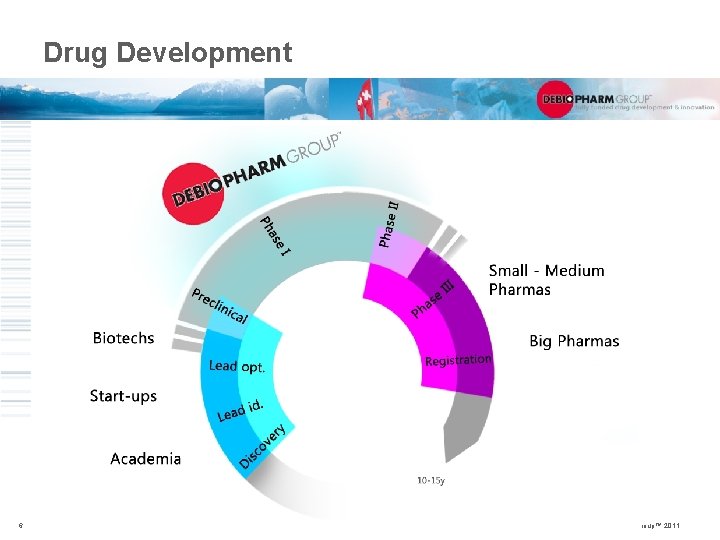
Drug Development 6 © Debiopharm Group™ 2011

Debiopharm Group™: an alternative for… à Companies that need à Companies that 7 Clinical development expertise Resources Funding … Re-focus therapeutic areas Have too many products Are undergoing M&A … © Debiopharm Group™ 2011

Therapeutic areas and preferred candidates 8 g Oncology Innovative targets g Internal medicine u Selected indications § Infectious diseases § Pain § Metabolic diseases § Immune-mediated diseases § Neurodegenerative disorders g Molecules: synthetics, biologics g Stage: from preclinical to end Phase I © Debiopharm Group™ 2011
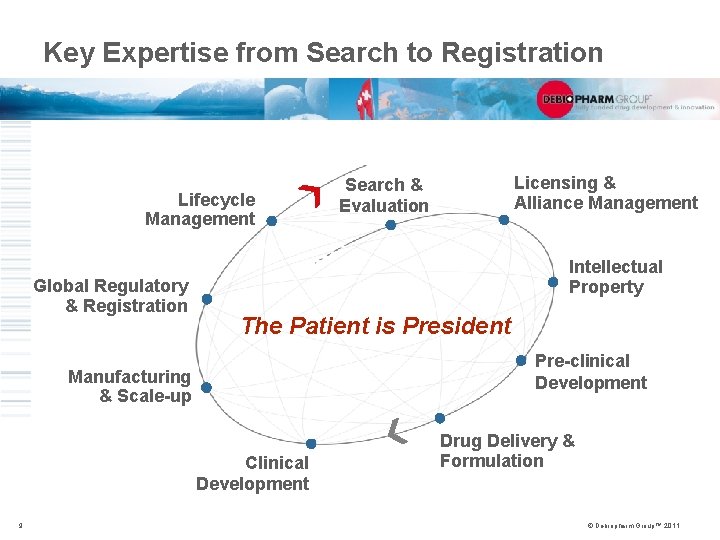
Key Expertise from Search to Registration Lifecycle Management Global Regulatory & Registration Intellectual Property The Patient is President Pre-clinical Development Manufacturing & Scale-up Clinical Development 9 Licensing & Alliance Management Search & Evaluation Drug Delivery & Formulation © Debiopharm Group™ 2011
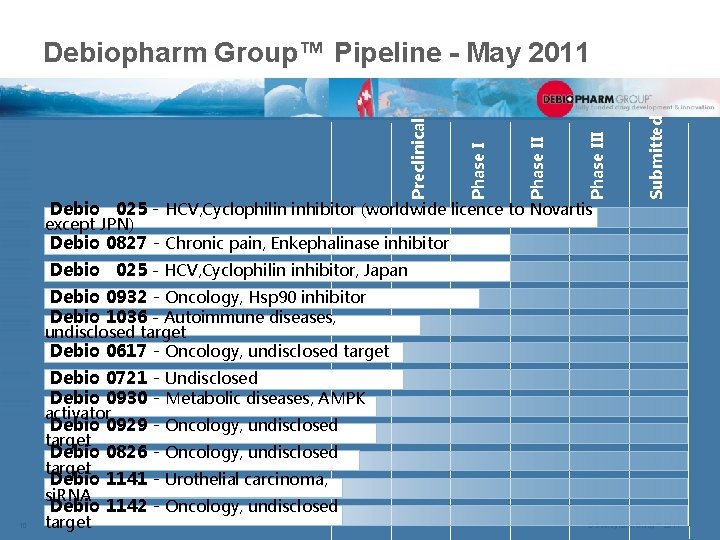
Debio 025 - HCV, Cyclophilin inhibitor (worldwide licence to Novartis Submitted Phase III Phase I Preclinical Debiopharm Group™ Pipeline - May 2011 except JPN) Debio 0827 - Chronic pain, Enkephalinase inhibitor Debio 025 - HCV, Cyclophilin inhibitor, Japan Debio 0932 - Oncology, Hsp 90 inhibitor Debio 1036 - Autoimmune diseases, undisclosed target Debio 0617 - Oncology, undisclosed target Debio 0721 - Undisclosed Debio 0930 - Metabolic diseases, AMPK activator Debio 0929 - Oncology, undisclosed target Debio 0826 - Oncology, undisclosed target Debio 1141 - Urothelial carcinoma, si. RNA Debio 1142 - Oncology, undisclosed 10 target © Debiopharm Group™ 2011

Companion Diagnostics & Personalised Medicine à à à 11 Objective: a more efficient use of drugs Giving the right medicine to the right patient at the right time à Develop molecular diagnostics à Identify and invest in high potential diagnostic start-up companies à Clinical development of Debiopharm Group’s drugs © Debiopharm Group™ 2011
Four strategic priorities : : Pharmaceutical development of molecules : : Production & redesign of molecules Debiopharm Group : : Companion diagnostics & personalised medicine : : Access to medicines in emerging economies 12 © Debiopharm Group™ 2011

Debiopharm Group™: highlights 2010 -2011 13 à Debio 025 / hepatitis C: out-licensing to Novartis à Decapeptyl R 6 month form, launched by Ipsen à Debio 0932 / oncology: initiation of phase I clinical study à Frost & Sullivan’s award: Debiopharm Group elected “European Biopharmaceutical Company of the Year” à Biocartis / diagnostics: partnership in personalized medicine à Diagnoplex / diagnostics: investment in the start-up à Marina Biotech: partnership to develop RNAi-based therapies à Agreements with Aurigen (oncology) and Yale (auto-immune diseases) © Debiopharm Group™ 2011

Debiopharm Group™ à Your long-term first choice partner… Entrepreneurial spirit and stable management Financial strength and independence Creativity, adaptability, flexibility Full development expertise Strong commitment to deliver Significant accomplishments Promising pipeline à 14 …from molecule to approval © Debiopharm Group™ 2011

Photo: Richard Juilliart - 2010 Debiopharm Group’s team builds the future 15 © Debiopharm Group™ 2011

Thank you! Debiopharm Group™ Headquarters Forum “après-demain” Chemin Messidor 5 -7 CP 5911, CH - 1002 Lausanne Switzerland Tel +41 (0)21 321 01 11 Fax +41 (0)21 321 01 69 www. debiopharm. com ll C 023 Ha h 5 t o o Maurice Wagner B Director Corporate Affairs & Communication Wilma Lukas, Ph. D, MBA Senior S&E Officer wlukas@debiopharm. com Photo: Jacques Straesslé - 2009 Contacts: mwagner@debiopharm. com 16 © Debiopharm Group™ 2011

Debiopharm Group™ à Back-up 17 slides © Debiopharm Group™ 2011

Vision, Mission & Values 18 à Vision We provide innovative, relevant and accessible therapies for tomorrow's world, through responsible medicine. à Mission We transform promising molecules into innovative therapies, building on our vision, our scientific excellence and our financial independence. à Values Creativity and pioneering spirit Passion Openness Exceeding excellence High ethical standards © Debiopharm Group™ 2011

Redesign of molecules and Production Debio R. P. à Facilities: FDA inspected and registered Swissmedic certified 19 ANVISA (Brazil) certified à Applied research: Medicinal chemistry for peptides Sustained-release formulations à Triptorelin vials Bulk manufacturing Exported worldwide © Debiopharm Group™ 2011
- Slides: 19