De Broglie wavelengths Contents de Broglie wavelengths Example

De Broglie wavelengths Contents: • de Broglie wavelengths • Example 1 • Whiteboards • Transmission electron microscopes • Scanning tunneling electron microscopes

Louis de Broglie Light is acting as both particle and wave Matter perhaps does also E = hf = hc/ E = mc 2 = hc/ mc = p = h/ • p = momentum (p = mv) • h = Planck’s constant (6. 626 x 10 -34 Js) • = de Broglie wavelength TOC
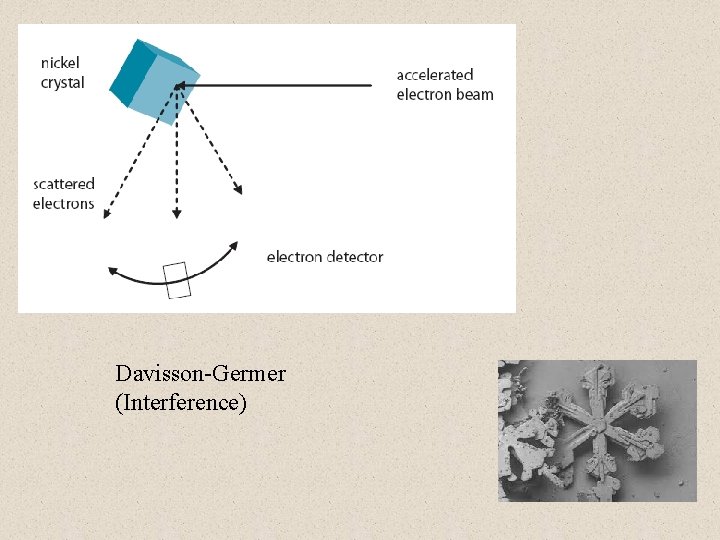
Davisson-Germer (Interference)

• p = momentum (p = mv) • h = Planck’s constant (6. 626 x 10 -34 Js) • = de Broglie wavelength Example 1: What is the de Broglie wavelength of a 0. 145 kg baseball going 40. m/s? p = mv = (0. 145 kg)(40. m/s) = 5. 8 kg m/s p = h/ , = h/p = (6. 626 x 10 -34 Js)/(5. 8 kg m/s) = 1. 1 x 10 -34 m Golly - nothing is that small (atoms are 10 -10 m) How would you observe the wave behaviour of that?

p = h/ • p = momentum (p = mv) • h = Planck’s constant (6. 626 x 10 -34 Js) • = de Broglie wavelength Ve = 1/2 mv 2 • V = accelerating voltage (V) • e = elementary charge (1. 602 x 10 -19 C) • m = particle mass (kg) • v = velocity (m/s) Example 2: Through what potential must you accelerate an electron so that it has a wavelength of 1. 0 nm? p = h/ = (6. 626 E-34 Js)/(1. 0 E-9 m) = 6. 626 E-25 kg m/s p = mv, v = p/m = (6. 626 E-25 kg m/s)/(9. 11 E-31 kg) = 727332. 6 m/s Ve = 1/2 mv 2, V = 1/2 mv 2/e = 1/ (9. 11 E-31 kg)(727332. 6 m/s)2/(1. 602 E-19 C) = 1. 504 V 2

Example 3: A 632. 8. nm photon has what momentum? A 1. 0 m. W laser beam has 3. 2 x 1015 of these photons per second. What net force does this beam exert on a surface where they are absorbed? p = 1. 05 x 10 -27 kgm/s F = 3. 3 x 10 -12 N

Whiteboards: de Broglie Wavelength 1|2|3|4|5

What is the de Broglie wavelength of an electron going 1800 m/s? (3) m = 9. 11 x 10 -31 kg p = mv p = h/ p = (9. 11 x 10 -31 kg)(1800 m/s) = 1. 6398 x 10 -27 kg m/s = h/p = (6. 626 x 10 -34 Js)/(1. 6398 x 10 -27 kg m/s) = 404 nm

What is the momentum of a 600. nm photon? p = h/ p = (6. 626 x 10 -34 Js)/(600. x 10 -9 m) = 1. 10 x 10 -27 kg m/s

Electrons in a microscope are accelerated through 12. 8 V. What de Broglie wavelength will they have? p = h/ Ve = 1/2 mv 2, v 2 = 2 Ve/m, v = √(2(12. 8 V)(1. 602 E-19 C)/(9. 11 E-31 kg)) = 2121739. 443 m/s p = h/ , = h/p = h/mv = (6. 626 E-34 Js)/((9. 11 E-31 kg)(2121739. 443 m/s)) = 3. 428 E-10 m

You want to use an electron to have the same wavelength as the waves on a violin string. Through what potential do you accelerate them. (violin strings are about 34 cm long, so the fundamental is about. 68 m long) p = h/ = (6. 626 E-34 Js)/(. 68 m) = 9. 74412 E-34 kg m/s p = mv, v = p/m = (6. 626 E-25 kg m/s)/(9. 11 E-31 kg) = 0. 001069607 m/s Ve = 1/2 mv 2, V = 1/2 mv 2/e = 1/ (9. 11 E-31 kg)(0. 001069607 m/s)2/(1. 602 E-19 C) 2 = 3. 25293 E-18 V

A 300. MW 620. nm laser is putting out 9. 36 x 1026 photons per second. since F = p/ t, and t = 1 second, what is the total thrust of the laser? (2) for 1 photon: p = h/ for 5 photons: 5 p = 5 h/ p = (6. 626 x 10 -34 Js)/(620. x 10 -9 m) = 1. 0687 x 10 -27 kg m/s total p change = (9. 36 x 1026 )(1. 0687 x 10 -27 kg m/s) = 1. 00 N

Applications of “matter” waves Electron Microscopes • Image resolution • = h/p • Electric or magnetic lenses • Gertrude Rempfer (PSU)

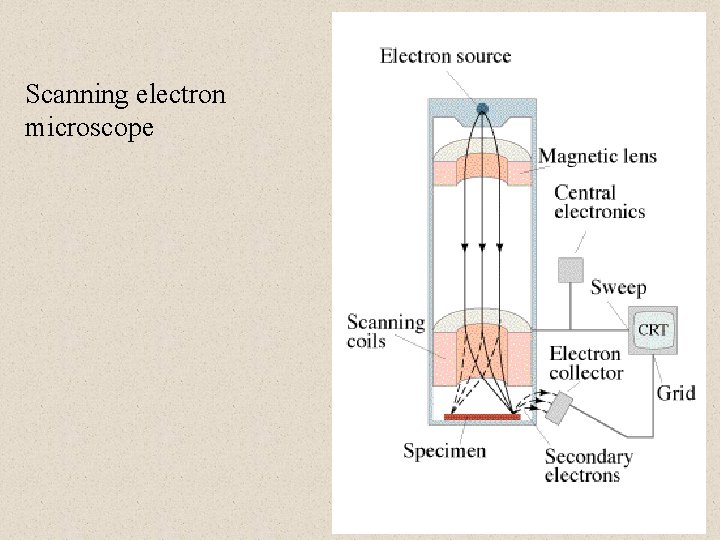
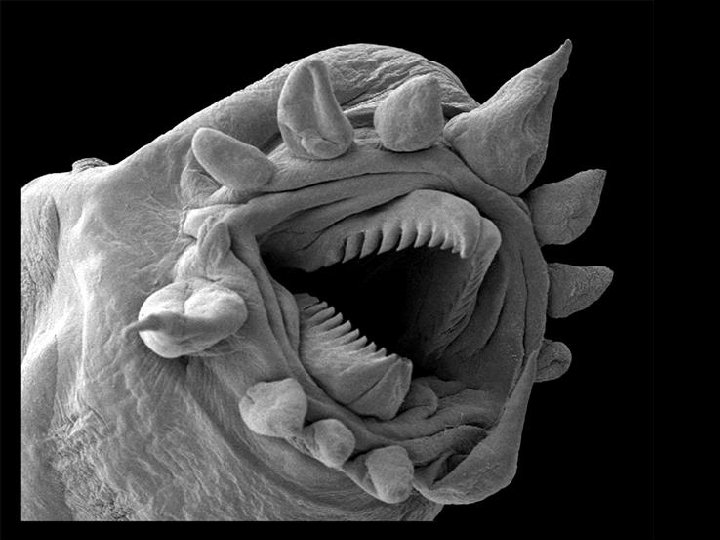
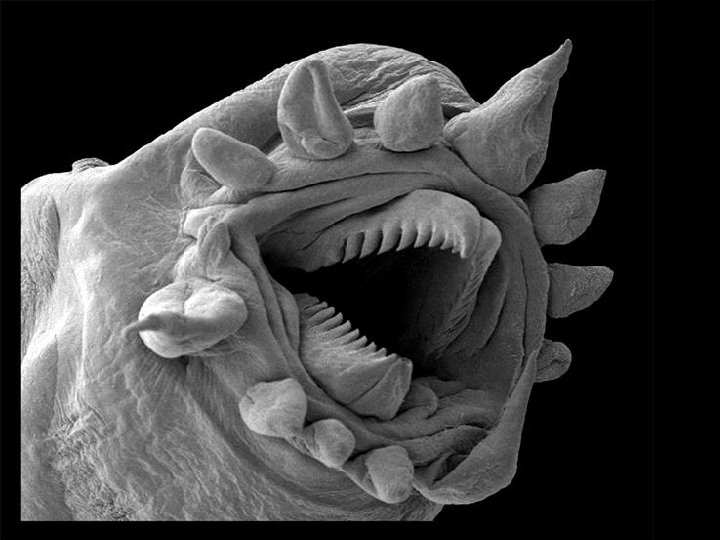
Scanning electron microscope
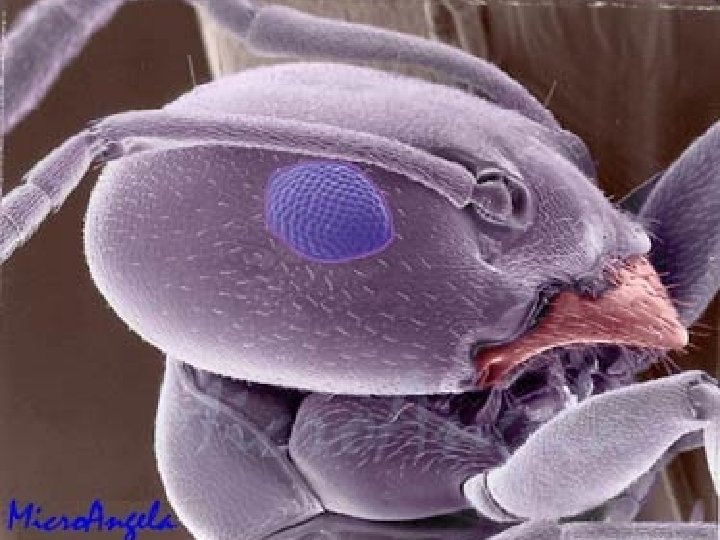
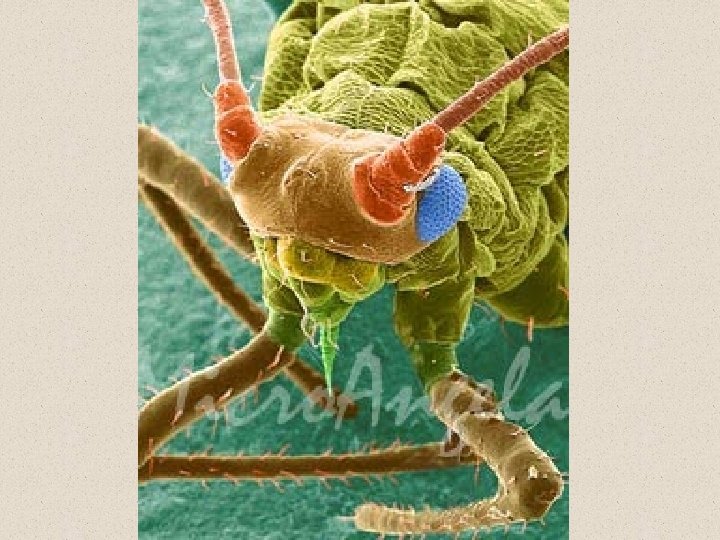
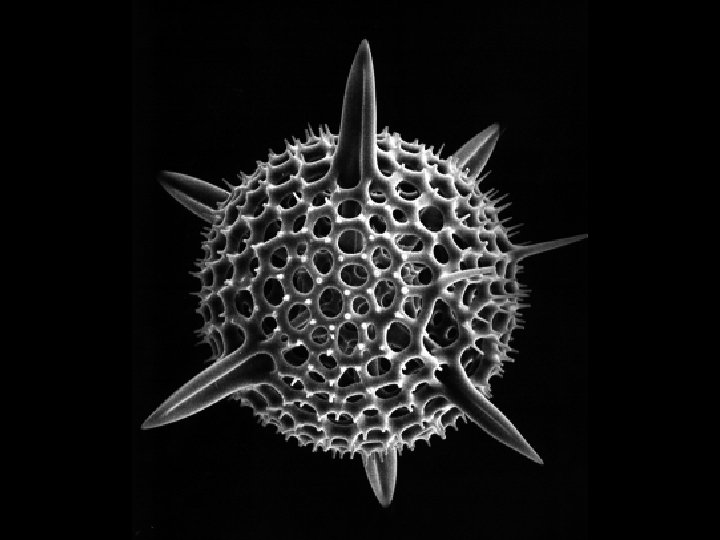
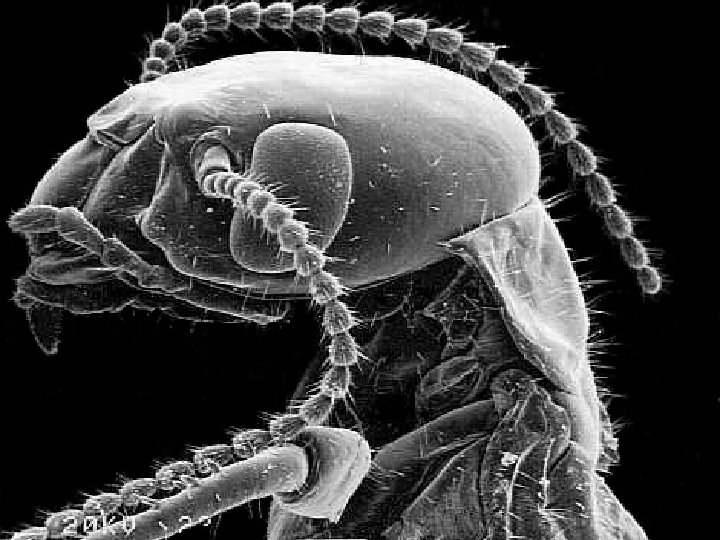



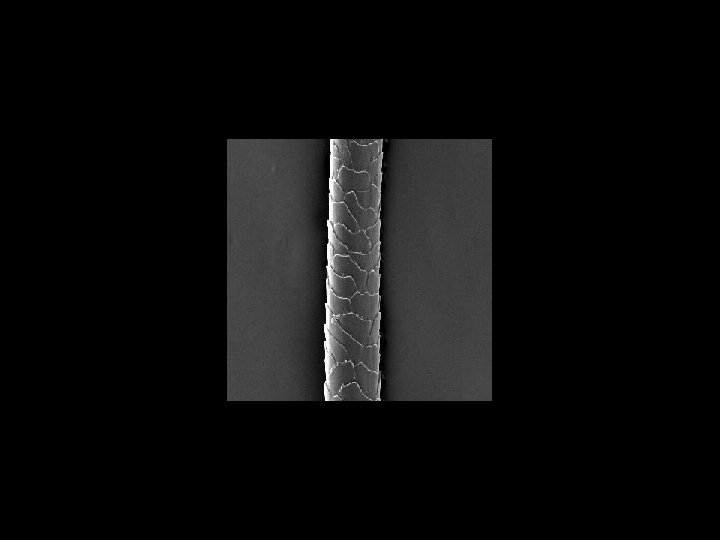
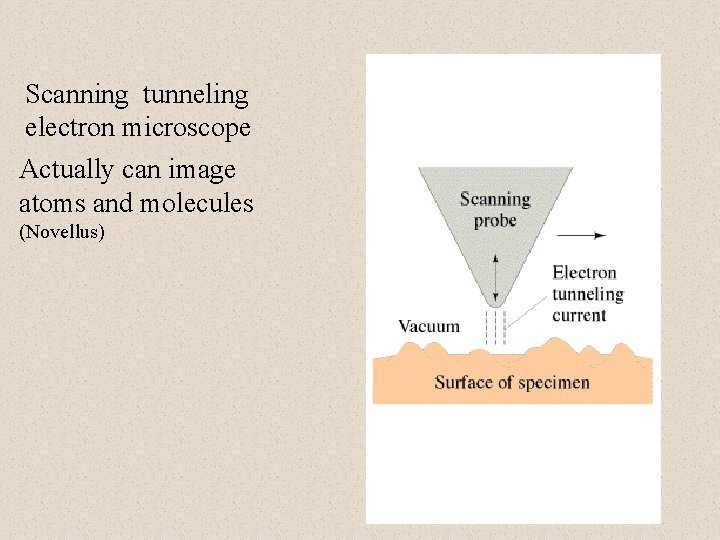
Scanning tunneling electron microscope Actually can image atoms and molecules (Novellus)
Soooo – Is/are Light/electrons a wave or particle? ? ? Wave behaviour Particle behaviour Complementarity/Duality Wave XOR Particle behaviour explains the behavior. Behaviour depends on situation. (Other particle interactions)

- Slides: 29